ABSTRACT
Introduction: In India majority of the neonatal transfusions are often prescribed on expert clinical opinion rather than concrete documented guidelines. Paucity of National Clinical Transfusion guidelines put neonatal transfusion practices in controversy. Due to the lack of sufficient data from India focusing on transfusion needs, patterns, indications, and short-term outcomes in preterm neonates prompted the requisite for this study.
Methods: It was a retrospective study conducted by department of transfusion medicine & blood bank, AIIMS, Jodhpur, a tertiary care center in western part of India. The patients were included all those who were admitted in neonatal intensive care unit (NICU), whether term or preterm for whom any blood component requested from blood bank, over a 19 months period from June 2019 to December 2020. Each transfusion episode was assessed as per BSH guidelines by reviewing blood requisition form. Data was analyzed via descriptive statistics.
Results: Out of the total 303 requisition of blood component transfusions, 127 requests were appropriate and 176 requests were inappropriate. Among these, FFP had highest inappropriate (76.4%) ordering component followed by PRBC (63.6%) , reconstituted whole blood (50%) and RDP (41.75%).
Conclusion: Almost 58.08%% of blood and blood components (FFP, platelets and PRBC, reconstituted whole blood, cryo) were ordered inappropriately. Judicious use of various blood products by following recommended guidelines may help in decreasing the inappropriate use of blood components.
Keywords: Transfusion Practices; Blood Components; Neonatal Intensive Care Unit
Abbreviations: PRBC: Packed Red Cell; RDP: Random Donor Platelet; FFP: Fresh Frozen Plasma; ELBW: Extremely Low Birth Weight; VLBW: Very Low Birth Weight; LBW: Low Birth Weight; NBW: Normal Birth Weight; BSW: British Society of Hematology; NICU: Neonatal Intensive Care Unit; RCC: Red Cell Transfusion; Hb: Hemoglobin; TRNEC: Transfusion Related Necrotizing Enterocolitis; NEC: Necrotizing Enterocolitis; PBM: Patient Blood Management; SGA: Short for Gestational Age; AGA: Appropriate for Gestational Age; INR: International Normalization Ratio; PT: Prothrombin Time; APTT: Activated Partial Thrombin Time; DIC: Disseminated Intravascular Coagulation
Introduction
Appropriate transfusion of neonates is vital to balance the transfusion benefits against risk. Sick neonates are heavily transfused groups of patients in critical care medicine. The rational utilization of blood components is very consequential in this age group. It, consequently, remains an important tool to continuously improve, amend and implement the most appropriate transfusion protocols for blood components utilized in neonates within the constraint of the evidence-based guidelines. Because of the length of their stay in the neonatal intensive care unit (NICU) and the frequent monitoring of parameters through blood sampling neonates experience iatrogenic blood loss and may require multiple transfusions. Majority of the neonatal transfusions are often prescribed on expert clinical opinion rather than concrete documented guidelines [1]. Lack of perspective patient blood management (PBM) program in neonates accentuates the blood management and best transfusion practices in the neonatal intensive care unit (NICU). Due to the lack of sufficient data from India focusing on transfusion needs, patterns, indications, and short-term outcomes in preterm neonates prompted the requisite for this study.
Materials & Methods
Study data were collected retrospectively over 19 months from June 2019 to December 2020. All preterm and term neonates admitted to the Neonatal Intensive Care Unit (NICU) at All India Institute of Medical Sciences (AIIMS) Jodhpur, Rajasthan, India, for whom blood requisition form was sent for component transfusion, i.e., Packed Red Blood Cells (PRBC), Random Donor Platelets (RDP), Fresh Frozen Plasma (FFP), Cryoprecipitate, Reconstituted Whole Blood, were included in the study. Data was collected from NICU medical records and review of the NICU blood order forms at the AIIMS Jodhpur blood center. According to British Society for Hematology (BSH) guidelines [2]. Those blood order forms were considered appropriate and satisfied all the transfusion criteria required to evaluate transfusion needs according to the BSH guidelines [2]. Inappropriate blood order forms are those which deviate from the guidelines and include incomplete documentation (i) for PRBC transfusion missing Hemoglobin (Hb), Weight of newborn (wt.), Saturation status (SpO2), (ii) for RDP transfusion missing platelet count, comorbidity or surgical or invasive procedure patient is undergoing, and (iii) for FFP transfusion coagulation parameter, bleeding at present and patient is undergoing some invasive procedure. Data analysis was done with descriptive statistics. Perpetual variables will be summarized as mean & standard deviation. Nominal/categorical variables will be summarized as the proportion (percentage) and analyzed by utilizing the Chi-Square test/Fisher exact test. P-value < 0.05 was considered as significant (Tables 1-4).
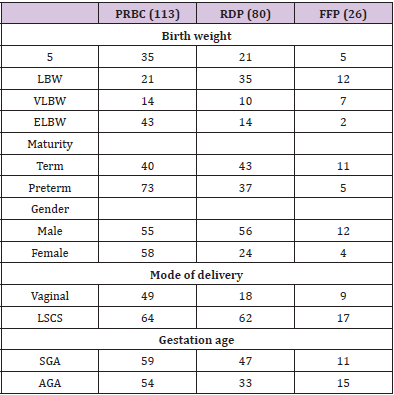
Table 1: Blood component issued as per Birth weight, maturity, gender, mode of delivery & gestational age.
Results
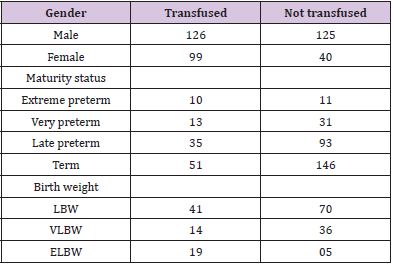
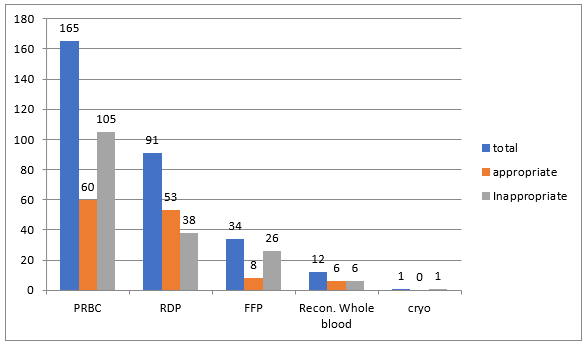
Data were analyzed retrospectively from June 2019 to December 2020. There were 390 NICU admissions during the study period, and 303 blood order forms were received. PRBC was the most frequently ordered blood component (165), followed by RDP (91), FFP (34), reconstituted whole blood (12), and cryoprecipitate (1) (Figure 1). Out of 303 received blood order forms, blood was issued for 225 requisitions (74%). Most frequent blood component issued was PRBC (113/225, 50%) followed by RDP (80/225, 35.5%), FFP (26/225, 11.5%) and reconstituted whole blood (6/225, 0.02%). NICU’s overall crossmatch to transfusion ratio (CT ratio) was 1.46 suggesting a significant utilization of blood components. During the study period, male admissions outnumber female admissions (251 vs. 139 respectively), and male requisitions received outnumber females (166 vs. 137 respectively). Females received more transfusions (p-value 0.000, chi-square 16.2), with PRBC accounting for the majority of transfusions in terms of gender predisposition. Out of the 303 requisitions received, the majority of the neonates were born through lower segment cesarean section (LSCS) compared with vaginal delivery and required frequent PRBC transfusions (166 v/s 137).
Out of the 390 NICU admissions, 187 were preterm (<37 weeks), and the rest were term. Requisitions received were higher for preterm neonates than term neonates (163 vs. 140 respectively). Out of the 163 preterm requisitions, 125 were issued and transfused (125/163; 77%), while for term babies, out of 140 requisitions, 94 were issued and transfused (94/140; 67%). Out of all the preterm transfusions, PRBC was the most frequent blood component transfused (73/125; 58%), followed by RDP (37/125; 30%) and FFP (15/125; 12%). While in term babies, RDP was the most frequent blood component transfused (43/94; 46%), followed by PRBC (40/94; 43%) and FFP (11/94; 12%). However, no significant association between transfusion requirement and maturity status (extreme preterm, very preterm, late preterm, and term patients) was seen (p-value 0.210).
In terms of birth weight, out of the 390 NICU admissions, LBW (birth weight 2500 to 1500 gm) were 111, VLBW (birth weight 1500 to 1000 gm) was 50, while ELBW (birth weight <1000 gm) were 24. Out of the 303 requisitions 101 (33%) were received from LBW neonates, 94 (31%) form NBW, 68 (23%) ELBW and 40 (13%) from VLBW neonates. In our study, ELBW neonates were more transfusion-dependent (Chi-square 18.8, p-value 0.000), and the most frequent blood component transfused was PRBC.
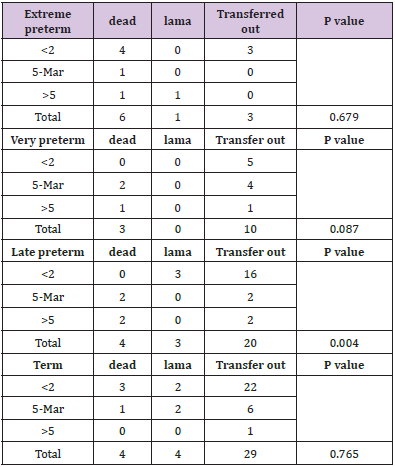
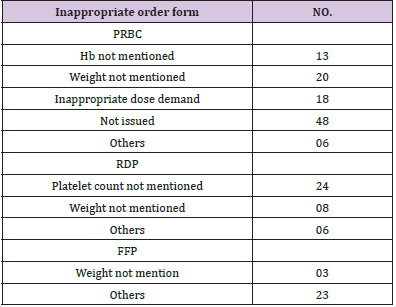
The outcome was assessed only for 87 patients (Chi-square 12.7, p-value 0.005), and the extremely premature neonate mortality was higher. In terms of maturity, mortality was higher in late preterm neonates when the number of transfusions increased by more than 2 (p-value =0.004). As per the gestational age, out of 303 requisitions, 140 (46%) were from small for gestational age neonates (SGA) while 163 (54%) were from appropriate for gestational age neonates (AGA). PRBC and RDP transfusions requirement were more in SGA neonates compared with AGA with mean number of transfusions being 2.92 for preterm neonate and 3.45 for SGA. Out of 303 blood requisitions 127 were found to be appropriate while rest 176 were inappropriate. Most frequent inappropriate component requested was FFP 76.4% (26/34), followed by PRBC 63.6% (105/165), reconstituted whole blood 50% (6/12) and RDP 41.75% (38/91) (Figure 1) Chi square =17.8, p value 0.001. A single request was received for cryoprecipitate for deranged coagulopathy during study period but component was not issued. Most frequent reason for inappropriate blood order forms demanding FFP was missing indication for transfusion (n=23;88%) and information against dose directed transfusion (n=3;12%). Similarly for inappropriate blood order forms received for RDP, majority (n=24;63%) do not have pre transfusion platelet count (needed to calculate percentage recovery and corrected count increment), 21% (n=8) do not have Weight mentioned and for rest 16% (n=6) order forms patient had no indication for transfusion.
Most common indication for reconstituted whole blood was exchange transfusion for the management of hyperbilirubinemia. Exchange transfusion was done only when serum bilirubin exceeds its target limit based on maturity status of newborn and live days. Out of 12 blood order forms received, 50% required the transfusion support and in rest jaundice was subsided by use of phototherapy. Out of 105 inappropriate blood order forms received for PRBC transfusion 12% (n=13) have missing Hb, 19% (n=20) have missing Weight, 17% (n=18) demand for inappropriate volume and 6% (n=6) can’t justify the transfusion need because of missing other parameters like comorbidity and saturation status. Forty six percent (n=48) blood order forms were those which were not issued and where transfusion was not needed. Most common indication for PRBC transfusion was anemia (63/113: 56%), for RDP transfusion thrombocytopenia and for FFP transfusion deranged coagulopathy with bleeding tendencies. Other indication for PRBC transfusion were during surgery (21/113:19%), shock (12/113:11%), blood loss (11/113: 10%), pathological jaundice (6/113: 0.05%) in decreasing order.
Discussion
Blood transfusion has a pivotal role in modern medicine. Despite being a lifesaving drug, it has deleterious effects also. This becomes important in neonates who have small blood volume and immature organ system. Both restrictive and liberal transfusion are well described in literature. Because of the lack of National Clinical Transfusion guidelines and varied transfusion practices among clinicians, most transfusions are based on expert clinical opinion. We advocate that neonate transfusions should be critically analyzed in terms of maturity, gestational age, disease effect, blood volume, appropriateness and clinician’s transfusion practice which is neglected in majority of the times. Between June 2019 and December 2020, 390 neonates were hospitalized with 225 requiring transfusions and 303 blood order forms received. In our study, the incidence of transfusion was 58.4 % (225/384) with PRBC being the most frequently requested blood component (113/303; 37.29 percent). Several studies have found that the incidence of neonatal transfusion ranges from 20% to 90% [3,4]. The fact that some studies included only extremely low birth weight newborns while others included term neonates explains the wide variance in transfusion. The majority of preterm and extremely low birth weight neonates in the NICU are transfused based on laboratory criteria. It should be emphasized that while transfusions offer an instant benefit in tissue oxygenation, it also inhibits the neonate’s immature bone marrow, delay newborn neurodevelopment outcomes, and lengthen hospital stays [5-9]. In our study, we observed that ELBW neonates are an extensively transfused patient population, with PRBC being the most often requested component. Thus, it may be concluded that frequent transfusion to maintain a higher hemoglobin level in ELBW infants gives no further benefit [5-9]. As a result, a restricted transfusion approach may be adopted. In our study, we found that the mortality was more in extremely preterm newborns than the term neonates (p value 0.005). The association is significant when frequency of transfusion is more than 2 and mortality was higher in late preterm neonates (p value 0.004). Thus, RBC transfusions may have a negative impact on neonatal survival.
In our study, we found that SGA neonates required more transfusions than AGA neonates (mean of 3.45). Conti et al. found similar findings, with the majority of transfused newborns being between 24 and 29 weeks of gestation and weighing less than 1000g [10]. Iatrogenic anemia occurring due to repeated blood sampling in the NICU is a leading cause of anemia [11,12]. It is estimated that in NICU mean sample volume range between 0.8-3.1 ml/kg/day which corresponds to 30%-300% of neonates blood volume [11]. Phlebotomy loss alone in first 24 hours of NICU admission is around 3-10ml corresponding up to 25% of the blood volume [12]. For us establishing a temporal relationship between iatrogenic blood loss and requirement for PRBC transfusion is difficult because no documentation ever made regarding the number and amount of neonatal blood samples collected.
Necrotizing Enterocolitis (NEC), a complication of red cell transfusion, is one of the leading causes of newborn death. Marin et al. found that alterations in mesenteric oxygenation during PRBC transfusion might be linked to transfusion-related NEC. In our study we report NEC in 3 patients, one having single episode of transfusion and rest had 7 and 19 transfusion episodes. All the patients were preterm and mortality was found only in one patient received multiple transfusions and had long hospital stay. Though we haven’t found any relation of NEC with transfusion but several other studies have found that PRBC transfusions are an independent risk factor for NEC with VLBW neonates being more vulnerable as early as 48 hours after the transfusion [13]. As a result, it is necessary to explore pharmacological agents to reduce the need for transfusions. The use of erythroid stimulating agents, such as recombinant erythropoietin or longer acting darbepoetin, might potentially reduce the need for transfusions. Recently, the PENUT study shown that erythropoietin treatment can maintain a much higher Hb level, reduce the need for transfusions, and expose fewer donors [13]. Our findings are consistent with other research that show anemia is the most prevalent reason for PRBC transfusion. A review of the literature reveals that a greater blood transfusion/ unit volume ratio in newborns puts them at risk of metabolic derangement. This is due to immature liver’s decreased ability to metabolize anticoagulant and sense hypoxia, and the immature kidney’s inability to handle electrolytes, leads to hypocalcemia, hyperkalemia, acidosis, and hypomagnesaemia. With multiple or large transfusions, these potential hazards are compounded. Though it is uncommon with top-up transfusions, hyperkalemia can develop if the neonate receives more than 20 ml/kg of transfusion volume, has underlying hyperkalemia, or has renal disease.
Potassium levels in these patients should be regularly checked. Cardiac arrest has been observed after receiving a large blood volume transfusion due to hyperkalemia, particularly in neonates receiving exchange transfusion. Because of the stored blood and high body surface area, exchange transfusion can potentially cause hypothermia. Hypothermia can aggravate coagulopathy and result in a fatal triad [14]. We found no negative effects from transfusion in our study. This might be because patient blood management is tightly enforced at our center, and every transfusion is scrutinized. The amount of coagulation protein in the neonate varies with gestational age. Premature babies have prolonged prothrombin time and activated partial prothrombin time (PT/aPTT) due to underdeveloped hepatic function. Unless there are bleeding symptoms, FFP is not recommended to restore a prolonged international normalized ratio (INR). The development of disseminated intravascular coagulation (DIC) in the absence of current bleeding does not warrant the use of FFP. The use of FFP in liver disease is also debatable, because FFP may not always result in a complete correction of the coagulation abnormality [15]. In our study we analyzed that FFP was the most inappropriately requested component. This can be explained by the fact that parameters required for assessing the FFP transfusions were missing on the requisition form. Majority of the platelet transfusions in neonates were done prophylactically as neonatal guideline have higher platelet transfusion threshold as compared to adult. Improved outcome was seen with platelet transfusions for hemorrhage secondary to thrombocytopenia [15].
Conclusion
Evaluation of clinical transfusion practices is intended to bring a realization among clinicians’ expert in their field about the need to evaluate their own practices regularly. The two primary risk factors identified that lead to numerous blood transfusions are low birth weight and preterm. Transfusion has been linked to an increase in mortality, therefore studies towards reducing newborn transfusion needs, particularly PRBC, which is commonly prescribed for anaemia correction, is necessary. The most inappropriately requested component was fresh frozen plasma, and the indication was abnormal coagulation profile; however, there is no basis for using FFP for coagulation profile correction in neonates except bleeding.
Conflict of Interest
There is no Conflict of Interest between authors. No economic or financial interest/ aid exists.
References
- Valentine SL, Bateman ST (2012) Identifying factors to minimize phlebotomy – induced blood loss in the pediatric intensive care unit. Pediatr Crit Care Med 13(1): 22-27.
- New HV, Berryman J, Bolton-Maggs PHB, Cantwell C, Chalmers EA, et al. (2016) Guidelines on transfusion for fetuses, neonates and older children. Bjh 175(5): 784-828.
- Dogra K, Kaur G, Basu S, Chawla D (2018) Red Cell Transfusion Practices in Neonatal Intensive Care Unit: An Exerience from Tertiary Care Centre. Indian J Hematol Blood Transfus 34(4): 671-676.
- Santos AM, Guinsburg R, de Almeida MF, Procianoy RS, Marba ST, et al. (2015) Factors associated with red blood cell transfusions in very-low-birth-weight preterm infants in Brazilian neonatal units. BMC pediatrics 15(1): 113.
- Whyte RK, Kriplani H, Asztalos EV, Andersen C, Blajchman M, et al. (2009) Neurodevelopmental outcome of extremely low birth weight infants randomly assigned to restrictive or liberal hemoglobin thresholds for blood transfusion. Pediatr 123(1): 207-213.
- Wang YC, Chan OW, Chiang MC, Yang PH, Chu SM, et al. (2017) Red blood cell transfusion and clinical outcomes in extremely low birth weight preterm infants. Pediatr Neonatol 58(3): 216-222.
- O'Riordan JM, Fitzgerald J, Smith OP, Bonnar J, Gorman WA (2007) Transfusion of Blood Components to Infants under Four Months: Review and Guidelines. Ir Med J 100(6): 1-21.
- Valieva OA, Strandjord TP, Mayock DE, Juul SE (2009) Effects of transfusions in extremely low birth weight infants: A retrospective study. J Pediatr 155(3): 331-337.
- Sanmugha Priya RA, Krishnamoorthy R, Panicker VK, Ninan B (2018) Transfusion support in preterm neonate and < 32 weeks in a tertiary care center: A descriptive study, Asian J Transfus Sci 12(1): 34-41.
- Said Conti V, Azzopardi E, Parascandalo R, Soler P, Attard Montalto S (2013) Overview of the blood transfusion policy in preterms on the Neonatal Intensive Care Unit. Malta medical journal 25(3): 46-50.
- Lubon NLC (1994) Review of neonatal red cell transfusion practices. Stood Rev 8(3): 148-153.
- Agrawal A, Goyal S (2014) Analysis of phlebotomy blood losses in neonates in a tertiary care hospital. Indian J Child Health 1(1): 7-11.
- Sandra E juul, Bryan A Comstock, Wadhawan R, Dennis E Mayock, Courtney SE, et al. (2020) A Randomized trial of erythropoietin for neuroprotection in preterm infants. N Eng J Med 382(3): 233-243.
- Palmieri TL (2017) Children are not little adults: blood transfusion in children with burn injury. Burns Trauma 5: 24.
- Galel SA, Fontaine MJ, Viele MK, Gonzalez L, Goodnough LT (2013) Wintrobe's Clinical Hematology: Thirteenth Edition. The Netherlands: Wolters Kluwer Health Adis (ESP); Transfusion medicine, pp. 547-586.

 Research Article
Research Article