ABSTRACT
Cyclic Formazan was invented and prepared for the first time by researcher Dr. Nagham Aljamali was established in April 2021. Therefore, it is considered one of the developed and modern compounds, which is described by a lack of references and researches. Therefore, the researcher, Dr. Nagham prepared and created a series of these compounds and studied several applications, including biological, pharmaceutical studies, also as antifungal, antibacterial, and anticancer agents, and here in this research, cyclic formazan was studied as medical nanomaterials. A number of technical spectroscopic studies have been used to demonstrate their chemical structures which provided clear evidences of their chemical structures via many technical instruments like (FT IR-Spectra , 1H.NMR-Spectra, Mass-Spectra)., Melting points, other studies represented by evolution them as Nano-compounds by {Scanning Electron Microscopy (FESEM), and their Chromatographic behavior}.
Keywords: Cyclic Formazan; Formazan; Imine; Schiff Base; Azo; Aldamine; Anil; Scanning Electron Microscopy (FESEM); Chromatography
Introduction
Cyclo- Formazan is one of the modern compounds in the field of organic chemistry and is considered an innovation by Dr. Nagham Aljamali in April 2021 when it was prepared for the first time globally [1,2]. And because their studies and references are a few for this cause the researcher Dr. Nagham Aljamali prepared and carried out various compounds from Macrocyclic-Formazan by using various conditions and different basic medium [3-7] like (Pyridine ,Pipridine ,5 % Sodium hydroxide, Triethyl amine,…) [3], and linked them with heterocyclic compounds and other compounds with more than two hetero atoms to increase their effectiveness [7-11] , biological [13,16] and industrial applications [17,20]. Cyclic Formazan has cyclic structure of (-N=N=C-N- in cyclic structure) or (-N=N-C-N-NH- in cyclic structure) according to type of amine in reaction [1,2]., They were considered among the organic compounds of importance in organic chemistry because they contain two highly effective groups in several fields of chemistry [1,21-24], especially in coordination chemistry [25-27], as a ligands because they contain free electrons of donor atoms to coordinate with ions to form complexes [28-30]. Formazan also tested in many studies as anti-bacterial [31-34] and anti-fungal compounds [35-36] and types of cancer [37-39], especially breast [40] and laryngeal cancers [41], as anti-bacterial [42] and antifungal [43,44], and other studies [40-44].
Instruments and Experimental Part
All melting points were uncorrected and dignified on an electro-thermal apparatus (Switzerland) in an open capillary tube. FT.IR spectra were detailed on Fourier transform infrared spectrometer (FT-IR) in (FT-IR- 3600) infrared spectrometer via employing KBr Pellet technique., 1H.NMR spectra were recorded in DMSO-d6 as solvent using (TMS) as internal standard and chemical shifts are expressed as (δ ppm)., also Mass– Spectra for some of them other studies represented by evolution them as Nanocompounds by {Scanning Electron Microscopy (FESEM), and their Chromatographic behavior}.
Procedures
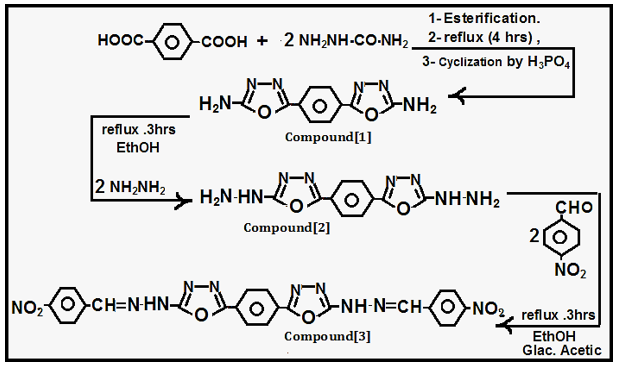
Preparation of Compounds {1, 2, 3}: Terephthalic acid (0.01 mole) was dissolved in (30 ml) absolute ethanol (2ml) of Sulfuric acid with refluxing for (2 hrs) in esterification step, then the ester will cyclize with semicarbazide (0.02 mole) with refluxing for (4 hrs) in presence of phosphoric acid as closing agent in cyclization step, according to procedures [4-7], the product filtered, dried, recrystallized to yield Ox diazole amine Compound [1], which reacted (0.01 mole) with (0.02 mole) hydrazine in refluxing step for (3 hrs) according to procedures [4-7], the product filtered ,dried, recrystallized to yield Oxadiazole hydrazine Compound [2], which refluxed (0.01 mole) with (0.02 mole) of p-nitrobenzaldehyde for (3 hrs) in presence of (3 drops of glacial acetic acid), according to procedure [4-7], the product filtered ,dried ,recrystallized to yield Imine -Compound [3] (Scheme 1).
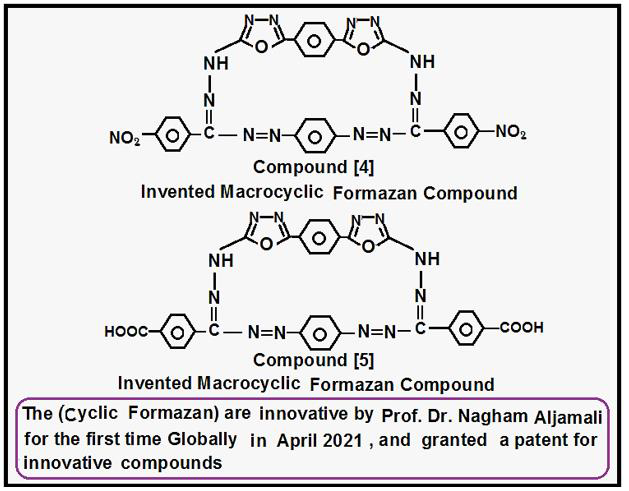
Creation of Inventive Macrocyclic Formazan Compound {4}: Compound [3] was (0.01 mole) reacted in presence of (Pyridine) with (0.01 mole) of diazo salt of p- phenyl diamine via many steps in basic medium to formation Invented Macrocyclic Formazan after (15 hrs), the product filtered, dried, washed by distilled water, recrystallized to yield Invented Macrocyclic Formazan [4] by following literatures [1,2].
Creation of Inventive Macrocyclic Formazan Compound {5}: Compound [2] refluxed (0.01 mole with (0.02 mole) of p-formal benzoic acid in presence of (2-3 drops) of glacial acetic acid for (2 hrs) in absolute ethanol according to procedure [4-7], the product filtered ,dried ,recrystallized to yield Imine -Compound that (0.01 mole) was reacted in presence [1-3] of (Triethyl amine) with (0.01 mole) of diazo salt of p- phenyl diamine via many steps in basic medium to formation Invented Macrocyclic Formazan after (12 hrs), the product filtered, dried, washed by distilled water, recrystallized to yield Invented Macrocyclic Formazan [5] by following literatures [1,2] (Scheme 2).
Creation of Inventive Macrocyclic Formazan Compound{6}: Compound [2] refluxed (0.01 mole with (0.02 mole) of p-formal phenol in presence of (2-3 drops) of glacial acetic acid for (3 hrs) in absolute ethanol according to procedure [3-7], the product filtered ,dried ,recrystallized to yield Imine -Compound that (0.01 mole) was reacted in presence [1,2] of (Pipyridine) with (0.01 mole) of diazo salt of p- phenyl diamine via many steps in basic medium to formation Invented Macrocyclic Formazan after (10 hrs), the product filtered ,dried ,washed by distilled water, recrystallized to yield Invented Macrocyclic Formazan [6] by following literatures [1,2].
Creation of Inventive Macrocyclic Formazan Compound{7}: Compound [2] refluxed (0.01 mole with (0.02 mole) of 4-N,Ndimethylamine benzaldehyde in presence of (2-3 drops) of glacial acetic acid for (2 hrs) in absolute ethanol according to procedure[3-7], the product filtered ,dried ,recrystallized to yield Imine -Compound that (0.01 mole) was reacted in presence [1-3] of (5 % NaOH) with (0.01 mole) of diazo salt of p- phenyl diamine via many steps in basic medium to formation Invented Macrocyclic Formazan after (20 hrs), the product filtered, dried, washed by distilled water, recrystallized to yield Invented Macrocyclic Formazan [7] by following literatures [1,2] (Scheme 3).
Results and Discussion
In recently scientific paper, various of Invented Macrocyclic Formazan Compounds have been created in same procedure that followed and invented [1,2] by Dr. Nagham in April 2021 that got a patent to invention of Macrocyclic Formazan compounds, then several studies were carried out to improve these innovative compounds by the using of spectral identification like : 1H.NMR spectra, FT.IR- Spectra, Mass- Spectra., other studies represented by (Melting points, other studies represented by evolution them as Nano-compounds via {Scanning Electron Microscopy (FESEM) ,and their Chromatographic behavior}., all the results are shown in tables and figures.
Spectral Investigation
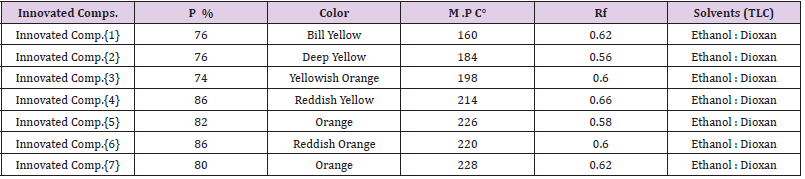
FT.IR- Spectral Indication of Invented Macrocyclic Formazan Compounds: The first characterization of innovative compounds by shifting of frequencies of Imine group (CH=N) in starting compounds (Imine compounds) that were about at (1615 , 1610, 1618, 1620) Cm-1 respectively in all starting materials of imine compounds that were shifted to (1630 , 1627 , 1631, 1642) Cm-1 for (-C=N-) due to formation of Macrocyclic Formazan, also appearance of three bands due to partitions of azo group of Formazan in Macrocycle (-N=N-) are (1429 ,1451, 1476) Cm-1 for (-N=N-C-) in compound {4}., and other compound like this., all frequencies clarified according to reference [1,33].
1H.NMR- Spectral Indication of Invented Macrocyclic Formazan Compounds: The second characterization of innovative compounds by disappearance of peak for imine group (CH=N) in starting compound (Imine compound) that were at δ (8.13) in Compound {3} (starting compound) due to formation of (N=CN= N) for (Formazan groups) in innovated compounds [4, 5, 6, 7], also in compound [5] appeared peak at δ (12.31) due to proton of carboxyl group (COOH), while compound [6] appeared peak at δ (10. 82) due to proton of hydroxyl group (OH), all peaks explained according to reference [33].
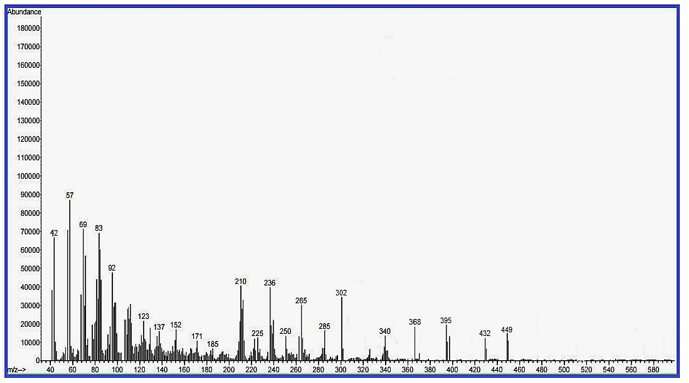
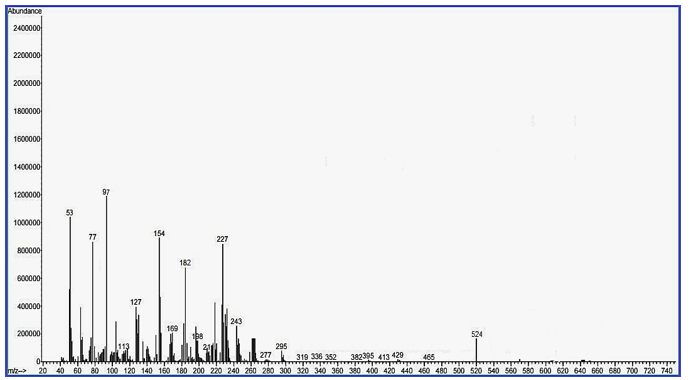
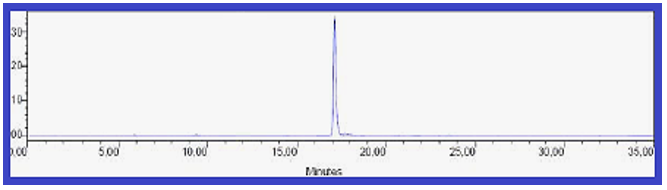
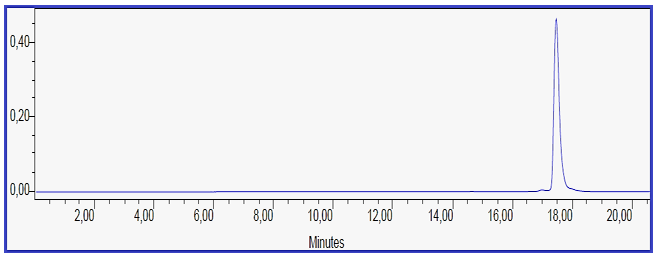
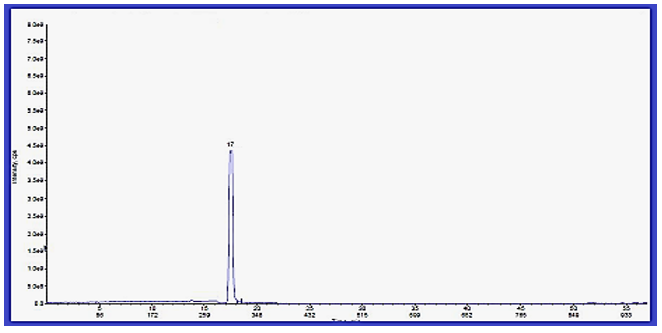
Mass– Spectral Indication of Invented Macrocyclic Formazan Compounds: The third characterization of inventive compounds by partition of innovative cyclic compounds via appearance of fragments in spectra in (Figures 1 & 2).
Other Characterization: All Invented Macrocyclic Formazan derivatives were studied to collect all the chemical and physical properties, in (Table 1).
Scanning Electron Microscopy (FESEM): Scanning Electron Microscopy (FESEM) of the Innovated Cyclic Formazan compounds (for morphological properties) that revealed in this research that they have a spherical shape and have granular sizes within the Nano-scale they have an average size of (44. 23 , 40. 12 , 46. 61, 55. 97) nanometers for Cyclic Formazan Compounds [4, 5, 6, 7] respectively, so the surface area increases and this characteristic makes it eligible for medical uses because it has a small granular size , spherical shape within the nano-scale that is used in medical fields as a treatment for many types of cancers as well as in the industrial field, (Figures 3-6).
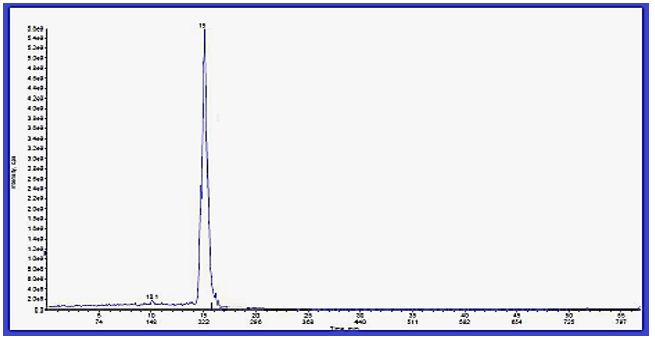
Chromatographic Study for Invented Cyclic Formazan Compounds: This section of the study involved a study of the chromatographic separation of the invented cyclic formazan compounds to know the effect of the effective groups in the chemical composition on the separation according to procedures [39-43], such as polar groups. In this work, Cyclic Formazan Compound {5} is the slowest compound in the separation because it contains two polar carboxyl groups (OH) that are affected when descending during the season and followed by Cyclic Formazan compound {6} according to polarity, then compound [4], and last one is compound [7], for this reason the compound [7] separated faster than other compounds due to its structure is less polarity and less interaction with column, (Figures 7-10).
Conclusion
All Invented Macrocyclic Formazan compounds gave good evidences for their structures via various spectral techniques, also some of them studied like scanning microscope appeared Nanoproperties for these compounds, which means that the cyclic Formazan compounds can be good drug delivery to treatment and medical applications.
Conflict of Interest
The authors declare that there is no conflict of interest.
Funding Source
None.
References
- Nagham Mahmood Aljamali (2021) Inventing of Macrocyclic Formazan Compounds and Studying Them against Breast Cancer for The first Time Globally. Annals of pharma research, pp. 525-533.
- Nagham Mahmood Aljamali (2021) Creation of Innovated Macrocyclic Sulfazan-Formazan Compounds and Linear Sulfazan-Formazan for the first Time Globally with their Assay as Antifungal. Biomedical Journal of Scientific & Technical Research 40(3): 32266-32272.
- Nagham Mahmood Aljamali (2018) Experimental Methods for Preparation of Mannich Bases, Formazan, Normal and Cyclic Sulfur Compounds. (1st)., Evince pub Publishing House.
- Nagham Mahmood Aljamali (2019) Review on (Azo, Formazane, Sulfazane)-Compounds. International Journal of Innovations in Scientific Engineering 10: 19-45.
- Nagham Mahmood Aljamali (2020) Alternative Methods in Organic Synthesis (1st )., Eliva Press SRL.
- Nagham Mahmood Aljamali (2021) Effect of Conditions and Catalysis on Products. 2021; (1st )., Eliva Press SRL.
- Nagham Mahmood Aljamali (2019) The Various Preparation Methods in Synthetic Chemistry. Evince pub Publishing house.
- Nagham Mahmood Aljamali, Imd Kam (2020) Development of Trimethoprim Drug and Innovation of Sulfazane-Trimethoprim Derivatives as Anticancer Agents. Biomedical & Pharmacology Journal 13(2): 613-625.
- Imd Kam, Hasaneen Kudhair Abdullabass, Nagham Mahmood Aljamali (2020) Invention of (Gluta.Sulfazane-Cefixime) Compounds as Inhibitors of Cancerous Tumors. Journal of Cardiovascular Disease Research, p. 44-55.
- Nagham Mahmood Aljamali (2018) Reactions and Mechanisms. IJMRA Publication.
- Matheus ME, de Almeida Violante F, Garden SJ (2007) Isatins inhibit cyclooxygenase-2 and inducible nitric oxide synthase in a mouse macrophage cell line. Eur J Pharmacol 556: 200-206.
- Nagham Mahmood Aljamali (2016) Synthesis and Biological Study of Hetero (Atoms and Cycles) Compounds. Der Pharma Chemica, p. 40-48.
- Nagham Mahmood Aljamali (2015) Synthesis and Chemical Identification of Macro Compounds of (Thiazol and Imidazol). Research J Pharm and Tech 8(1): 78-84.
- Mestaf M, Nawfel Muhammed Baqer Muhsin (2019) Neuro Quantology. 17(11): 11-16.
- Nagham Mahmood Aljamali (2015) Review in Azo Compounds and its Biological Activity. Biochem Anal Biochem 4: 169.
- Nagham Mahmood Aljamali, SF Jawad, (2022) Preparation, Spectral Characterization, Thermal Study, and Antifungal Assay of (Formazane -Mefenamic acid) - Derivatives. Egypt J Chem 411 65(2).
- Nagham Mahmood Aljamali (2017) Synthesis of Antifungal Chemical Compounds from Fluconazole with (Pharma-Chemical) Studying. Research journal of Pharmaceutical, biological and chemical sciences 8(3): 564 -573.
- MN Abdmajed, Nagham Mahmood Aljamali (2021) Preparation of Benzothiazole-Formazane Reagents and Studying of (Spectral, Thermal, Scanning Microscopy, Biological Evaluation). International Journal of Pharmaceutical Research 13(1): 4290-4300.
- Mad M, Nagham Mahmood Aljamali, Nadheema AA (2018) Preparation, Spectral Investigation, Thermal Analysis, Biochemical Studying of New (Oxadiazole -Five Membered Ring)-Ligands. Journal of Global Pharmacy Technology 10(1): 20-29.
- Nagham Mahmood Aljamali (2020) Survey on Methods of Preparation and Cyclization of Heterocycles. International Journal of Chemical and Molecular Engineering 6(2): 19-36.
- Mid M, Nagham Mahmood Aljamali, Wassan Ala Shubber, Sabreen Ali Abdalrahman (2018) New Azomethine- Azo Heterocyclic Ligands Via Cyclization of Ester. Research Journal of Pharmacy and Technology 11(6): 2555-2560.
- Hasaneen Kudhair Abdullabass, Aseel Mahmood Jawad, Nagham Mahmood Aljamali (2020) Synthesis of drugs derivatives as inhibitors of cancerous cells. Biochem Cell Arch 20(2): 5315-5322.
- Aseel Mahmood Jawad, Nagham Mahmood Aljamali (2020) Innovation, Preparation of Cephalexin Drug Derivatives and Studying of (Toxicity & Resistance of Infection). International Journal of Psychosocial Rehabilitation 24(04): 3754-3767.
- Husen Ali Ahmd, Nagham Mahmood Aljamali (2021) Preparation, Characterization, Antibacterial Study, Toxicity Study of New Phenylene diamine- Formazan Derivatives. Indian Journal of Forensic Medicine & Toxicology 15(2): 3102-3112.
- Nagham Mahmood Aljamali, Hussein Mejbel Azeez (2021) Synthesis and Characterization of Some New Formazan - Cefixime and Study of Against Breast Cancer Cells. Annals of RSCB 25(3): 8562-8578.
- Nagham Mahmood Aljamali, Asmaa Kefah Mahdi (2021) Synthesis, Identification and Anticancer Studying of Heterocyclic- Mefenamic Drug via Thiosemicarbazide. Annals of RSCB 25(3): 8521-8537.
- F Jawad, Nagham Mahmood Aljamali (2021) Preparation, Investigation and Study of Biological Applications of Tyrosine Derivatives against Breast Cancer Cells. NeuroQuantology 19(9): 117-125.
- Nagham Mahmood Aljamali, Intisar Obaid Alfatlawi (2015) Synthesis of Sulfur Heterocyclic Compounds and Study of Expected Biological Activity. Research J Pharm and Tech 8(9): 1225-1242.
- Intisar Obaid Alfatlawi, Nuha SS, Zainab MJ, Nagham Mahmood Aljamali (2017) Synthesis of New Organic Compounds Via Three Components Reaction with Studying of (Identification, Thermal Behavior, Bioactivity on Bacteria of Teeth). Journal of Global Pharma Technology 11(9): 157-164.
- Nagham Mahmood Aljamali, Saher Mahmood Jawd, Zainab MJ, Intisar Obaid Alfatlawi (2017) Inhibition activity of (Azo–acetyl acetone) on bacteria of mouth. Research Journal of Pharmacy and Technology 10(6): 1683-1686.
- Aseel Mahmood Jawad, Nagham Mahmood Aljamali, Saher Mahmood Jawd (2020) Development and Preparation of ciprofloxacin Drug Derivatives for Treatment of Microbial Contamination in Hospitals and Environment. Indian Journal of Forensic Medicine & Toxicology 14(2): 1115-1122.
- Nagham Mahmood Aljamali (2022) Spectral and Laboratory Diagnostics of Compounds. (1st)., Eliva Press SRL.
- Shireen R Rasool, Nagham Mahmood Aljamali, Ali Jassim Al-Zuhairi (2020) Guanine substituted heterocyclic derivatives as bioactive compounds. Biochem Cell Arch 20(2): 3651-3655.
- Deniz Ar, Alexander FR Kilpatrick, Beatrice Cula, Christian Herwig, Christian Limberg (2021) Transformation of Formazan ate at Nickel(II) Centers to Give a Singly Reduced Nickel Complex with Azoiminate Radical Ligands and Its Reactivity toward Dioxygen. Inorganic Chemistry 60(18): 13844-13853.
- Francesca Milocco, Folkert de Vries, Harmke S Siebe, Silène Engbers, Serhiy Demeshko, et al. (2021) Widening the Window of Spin-Crossover Temperatures in Bis(formazanate)iron(II) Complexes via Steric and Noncovalent Interactions. Inorganic Chemistry 60(3): 2045-2055.
- Abd Ali H, Nagham Mahmood Aljamali (2021) Chalcone-Heterocyclic Derivatives (Synthesis, Spectral Identification, Microbial Evaluation). International Journal of Pharmaceutical Research 13(1): 4234-4242.
- Nor A, Sud S, Nagham Mahmood Aljamali (2019) Synthesis, Characterization and Thermal Analysis for New Amoxil Ligands. Asian Journal of Chemistry 31(5): 1022-1026.
- Rajaa Abdul Ameer Ghafil, Nor A Alrab, Nagham Mahmood Aljamali (2020) Synthesis of Triazole Derivatives via Multi Components Reaction and Studying of (Organic Characterization, Chromatographic Behavior, Chem-Physical Properties). Egypt J Chem 63(11): 4163-4174.
- Nagham Mahmood Aljamali (2018) Synthesis, Investigation, Chromatography, Thermal) - Behavior of (Five, Seven) - Membered Ring with Azo and Anil Compounds. Pak J Biotechnol 15(1): 219-239.
- Mhammd Abdul, Abd Ali H, Nagham Mahmood Aljamali (2021) Synthesis, Spectral, Bio Assay, Chromatographic - Studying of New Imidazole Reagents Via Three Components Reaction. NeuroQuantology 19(7): 115-122.
- Nagham Mahmood Aljamali (2019) Separations of Samples Contents (Vitality, Environmental, Chemical) By Instrumental & Laboratory Methods. 1 (Eds.)., Evince pub Publishing.
- Nagham Mahmood Aljamali, Tabark Emad Al-Faham (2021) Synthesis, Identification, Chromatographic Studying of Formazane –Phenylenediamine Derivatives. Annals of RSCB 25(4).
- Nagham Mahmood Aljamali, Jad F (2021) Preparation, Diagnosis and Evaluation of Cyclic-Tryptophan Derivatives as Anti Breast Cancer Agents. Biomed Pharmacol J 14(4).
- Mid Mohmd, Nagham Mahmood Aljamali, Sabreen Ali Abdalrahman, Wassan Ala Shubber (2018) Formation of Oxadiazole Derivatives Ligands from Condensation and Imination Reaction with References To Spectral Investigation, Thermal and Microbial Assay. Biochem Cell Arch 18(1): 847-853.

 Research Article
Research Article