Abstract
Objective: The purpose of this study was to describe complications associated with the endoscopic transsphenoidal approach and to determine a relation between postoperative central diabetes insipidus (DI) and the characteristics of adenomas.
Methods: 259 patients with adenomas had been selected for our study. All the complications were divided into two groups: surgical complications and DI.
Results: Of the 259 patients 159 had non-secreting adenomas, 83 - growthhormone secreting adenomas, 5 – prolactinomas; 9 patients developed Cushing’s disease, 2 - thyreotropinoma and 1 patient had mixed adenoma (growth-hormone and ACTH-secreting). Most common were macroadenomas, followed by giant adenomas and microadenomas. The overall rate of surgical complications was 13.1%. Some of them had two or more complications. The overall rate of diabetes insipidus (DI) was 42.9% (34.8% - permanent and 8.1% - transient DI).
Conclusions: The complications after endoscopic surgery are rather common and include two big groups: surgical and endocrine complications. In multivariate analysis the predictors of DI were patient’s age, size and consistency of adenomas. The older a patient the greater effect the tumor size had on the risk of developing DI. In young patients the risk of DI was 1.51 times higher with increasing of tumor size by one grade in while in elder patients it was 2.3 times higher. The tight consistency of tumor was another important factor the patients developed DI after surgery. The unsuctionable consistency increased risk of DI in 2.89 times.
Keywords: Endoscopic; Pituitary Adenomas; Complications; Diabetes Insipidus
Introduction
Pituitary adenomas constitute about 10-12% of all intracranial tumors [1,2]. They are benign tumors growing from adenohypophyseal cells. All adenomas are classified according to their hormonal activity as non-secreting and secreting (prolactinoma, growth hormone-secreting, ACTH-secreting and thyroid stimulation hormone-producing) adenomas. The treatment of choice for most patients with secreting adenomas is surgical removal. In case of prolactinomas, dopamine agonists can be prescribed first. Surgery can be an option in this case if the conservative treatment is unsuccessful [1]. Patients with nonsecreting adenomas and any neurological or vision deficit are also candidates for surgery. The most common surgical technique for pituitary adenomas is the endoscopic transnasal transsphenoidal approach. This technique is becoming more popular and refined due to better lighting and endoscope’s superior visualization [3-5]. These factors have significantly reduced surgical complications and improved tumor removal. The aim of this study was to determine the relation between postoperative central diabetes insipidus (DI) and the characteristics of adenomas, tumor removal extent and surgical complications. Central DI has been one of the most common forms of transient or permanent disorder of fluid homeostasis after transsphenoidal surgery. Any disturbance of the posterior pituitary, pituitary stalk or neurons originating in the paraventricular or supraoptic hypothalamic nuclei can lead to DI [6,7].
Material and Methods
This study was a retrospective analysis of 259 patients with pituitary adenoma who were surgically operated at Federal Center of Neurosurgery of Ministry of Public Health of Russian Federation in Novosibirsk from July 2014 to December 2018. Patients who had been operated in our hospital before July 2014 using the microsurgical transsphenoidal technique and patients with DI before operation were excluded from this study. Only histologically verified pituitary adenomas were included. The following patient characteristics were recorded preoperatively: type of adenoma (non-secreting or hormone-producing), age of patient, gender, size of tumor (microadenoma (<10 mm), macroadenoma (10- 39 mm), giant adenoma (≥40 mm)), preoperative visual deficit (loss of visual fields or decreased visual acuity), preoperative hormone insufficiency, number of previous surgeries for pituitary adenoma. At the postoperative period, the following complications were evaluated: postoperative cerebrospinal fluid (CSF) leakage, meningitis, neurological deterioration, new visual deficit, hematoma, epistaxis, deep vein thrombosis (DVT), pulmonary embolism (PE) and DI. Other endocrinological complications were excluded from this study.
Surgical Technique
All the operations had been planned using the data collected from preoperative CT, contrast-enhanced MRI and MR-angiography. In every case intraoperative navigation (Integra, Radionics) was used. All the patients underwent a fully endoscopic endonasal approach. The operations took place with the patient’s head fixed in a three-pin Mayfield holder. Both nostrils were prepared with a topical vasoconstrictor. Between July 2014 and July 2015, we used a mononostril approach. Generally, the approach was through the right nostril, the left one was used only in a case of major septal deviation to the right. The initial step included lateralization of middle turbinate, but not resection. We removed the rostrum and small back part of the septum. At that moment the endoscope irrigating system (ClearVision system, Karl Storz) was additionally used as the monomanual technique was performed. In cases of intraoperative CSF-leakage, a fat-graft and fibrin-glue were applied for skull base reconstruction. After July 2015 we have begun applying a binostril approach, using the bimanual technique with an assistant surgeon holding the endoscope. The middle turbinate was lateralized on the left side and resected on the right side after injection in mucosa a solution of 2% lidocain and adrenalin. On the right side a nasal septal flap was formed using unipolar coagulation.
The anterior wall of the sphenoid sinus was opened bilateraly. Nasal cavity and sinus were disinfected with Octenisept. The septum and mucosa from sphenoid sinus were removed totally and then the bone of floor of sella turcica was removed rather wide. In all the cases we used a high-speed drill (2 or 4 mm in diameter). The dura was opened with a telescopic knife and microscissors under direct visual control. Before July 2015, the tumor was removed with curettes and after July 2015 - with suction and microforceps or using the two-suction technic. In all the cases the Karl Storz hard endoscopes (180/4 mm) with 0°, 30°, 45° angulations attached to high-definition cameras was used. In the case of unimanual technique the surgeon held the endoscope in his left hand and the instrument - in the right one, while an assistant held a suction tube, but in the case of bimanual technique the assistant held an endoscope and the surgeon - one suction tube in his left hand and in right hand – a second suction tube or any other instrument. In a case of high bleeding the assistant could help, holding one more suction tube. For skull base reconstruction vascularized flaps or mucosa from the resected middle turbinate were used. In presence of middle-flow intraoperative CSF-leakage, a fascia lata graft was applied. In some cases, we used a fat graft or a bone part or suture on dura mater for reconstruction, as well as artificial dura (Duraform), fibrin glue and a Foley catheter. The patients were not routinely given corticosteroids before surgery. Antibiotics administration continued for 24 hours.
Postoperative Evaluation
In 48 hours after surgery a full endocrinological assessment was performed. Within 72 hours postoperatively all the patients underwent MRI brain scans to assess the volume of removal. In cases of non-functional adenomas our main interest was control of cortisol, thyroxin, prolactin and testosterone. In case of growthhormone secreting adenomas, we additionally controlled the patients’ growth hormone, in case of ACTH-hormone secreting - adrenocorticotropic hormone, and in case of TSH-hormon secreting adenomas - thyroid stimulating hormone and threeiodthyronin. For evaluation of DI postoperatively the level of electrolytes, urine-specific gravity and production of excessive volumes of urine were controlled. All the patients underwent postoperative ENT inspection. Every patient with any preoperative visual deficit underwent neuro-ophthalmological evaluation postoperatively.
Statistical Analysis
Data analysis was performed using the R 3.3.3 statistical package [8]. Bivariate analysis was performed to identify the predictive factors of DI among the collected variables. It included Fisher’s exact test (asymptotic implementation) for qualitative variables and Wilcoxon rank sum test for quantitative variables. All the tests were 2-sided. Univariate analysis was carried out to test the covariates predictive of outcome (DI onset). The factors predictive in the univariate analysis (p<0.25) were entered into stepwise logistic regression analysis. Additionally based on some clinical considerations and graphical analysis, we tested a number of hypotheses about the nonlinear entry of variables into the logit function [9]. To select the final model AUC and ANOVA were used.
Results
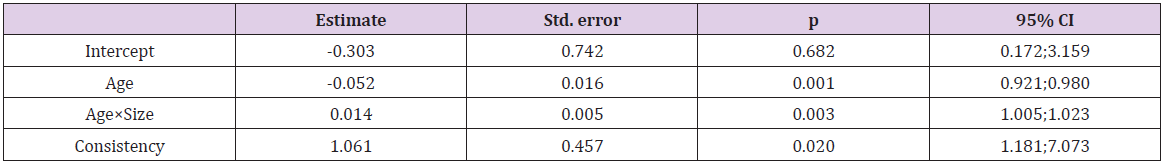
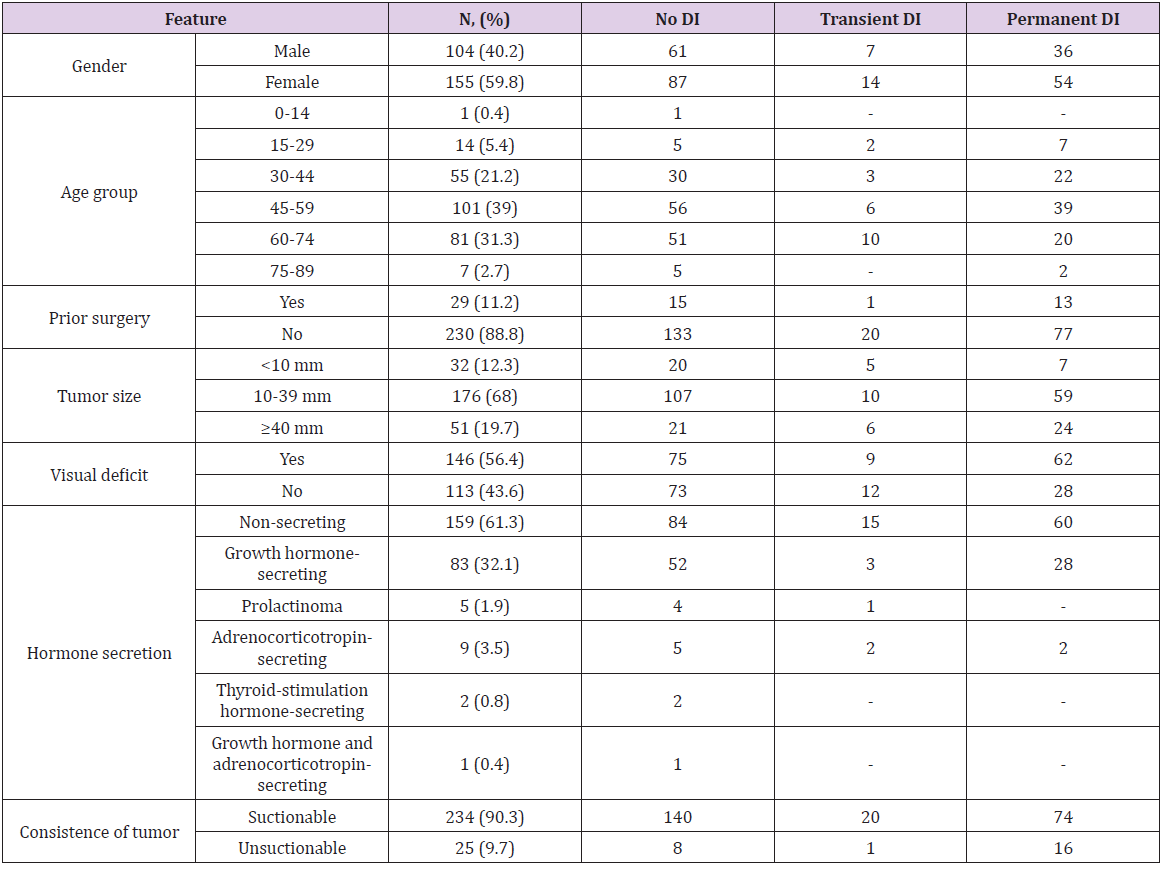
All the 259 patients were operated at the same hospital by the same two surgeons from July 2014 to December 2018. The mean age at the time of surgery was 52.4 years, median - 55 (ranged from 14 to 79). The interquartile range was from 42 to 62. Women prevailed over men (155 versus 104). All the pituitary adenomas were divided by size into microadenomas (32 cases), adenomas of 11-39 mm in size (176 cases) and giant adenomas over 40 mm in size (51 cases). There were 159 non-secreting, 5 prolactinsecreting, 83 growth hormone-secreting, 9 ACTH-secreting, 2 thyroid stimulation hormone-secreting adenomas, and 1 patient had a mixed adenoma (growth-hormone and ACTH-secreting). Preoperatively 172 patients had visual deficit (66.4%). 230 patients were operated for the first time, 29 patients were reoperated due to tumor recurrence. As possible predictors of DI onset, the following factors were considered: gender (p=0.69), age (p=0.10), presence of prior surgery (p=0.53), hormone secretion (p=0.71), adenoma size (p=0.03), its consistency (p=0.01) and extent of tumor removal (p=0.71). The final model of logistic regression included factors interaction (Table 1): In the group of microadenomas, the influence of age was OR=0.963, CI= [0.94; 0.987], in macroadenomas - OR=0.977, CI= [0.957; 0.997], in giant adenomas - OR=0.99, CI= [0.971; 1.01]. The greatest impact the age rendered in increasing DI had in the group of microadenomas, but in the case of giant ones it was about 0.
Estimated OR and 95 CI for Size controlling for Age are shown in (Table 2). The size effect depended on the patients’ age. In young patients (30-year-old) the risk of DI was 1.51 times higher with increasing of tumor size by one grade in while in elder patients (60-year-old) it was 2.3 times higher. The transition from suctionable to unsuctionable increased the risk of DI 2.89 times (CI= [1.18; 7.07], p=0.020). In our opinion, the main cause of DI was changes in the shape and size of the pituitary gland in presence of giant and macroadenomas that determined bad visualization of the gland during surgery (Table 3). One of the most important surgical characteristics of an adenoma is its consistency. More dense and unsuctionable tumors have a higher risk (2.89 times) of post-op DI, because of worse identification of pituitary gland (p=0.020). No significant difference was found between the patients with and without surgical complications. The hormonal activity of adenomas had no effect associated with DI development (p=0.71) as well. In our study patients with prior surgery did not have a higher DI incidence (p=0.53). Smith et al. indicate that percentage of permanent DI after a first operation was 2.6% and while rate of transient DI after second surgery was 37.9% [10]. Also, patients with gross totally, subtotally or partially removed tumors had no differences in DI development (p=0.71).
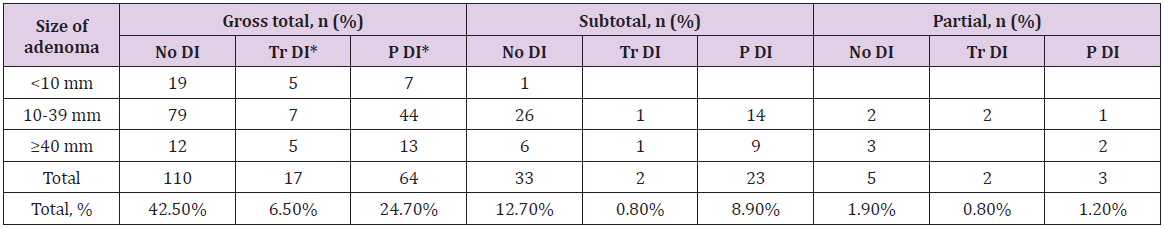
Table 3: Extent of resection.
Note: *Tr DI – Transient Diabetes Insipidus; P DI – Permanent Diabetes Insipidus
Extent of Tumor Removal
The radicality of tumor removal was evaluated using contrastenhanced MRI within the first 72 hours. The resection extent is presented in (Table 4). Gross total resection was achieved in 74% (191/259) of cases. This group included all microadenomas and a part of macro- and giant adenomas, which were easily suctionable. A group of patients with subtotal (22%, 58/259) or partial removal (4%, 10/259) had unsuctionable macro- or giant adenomas, and giant adenomas with big antesellar part. The radicality of tumor removal depended on a tumor’s consistency (р=0.009) and size (р <0.001) (Table 3).
Visual Outcome
Of the 146 patients with preoperative visual deficit, impaired visual acuity improved postoperatively in 61%, visual fields normalized in 61.6% Postoperative deterioration of visual function was detected in 6 patients including 3 cases of visual acuity deterioration and 3 cases of deterioration of both visual acuity and fields. These patients had macro and giant adenomas with a large suprasellar part and had already been suffering from severe visual impairment. Fifty-three patients did not have any changes in their visual fields. The visual acuity stayed the same after surgery in 51 patients.
Overall Complication Rate
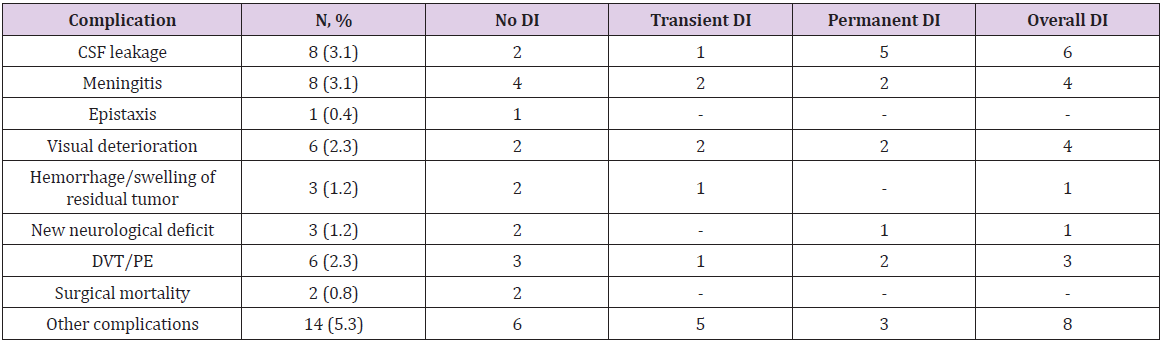
34 patients developed different complications in the early postoperative period, while the remaining 225 patients had no complications whatsoever (Table 4).
Postoperative CSF leakage
In 8 patients (3.1%) a postoperative CSF leakage occurred. One of the patients had microadenoma, 4 patients had macroadenomas and 3 patients – giant adenomas. In 4 cases we did not use vascularized or mucosal flaps. Artificial dura or a fat graft was used instead. In two cases necrotization of the vascularized flap was observed. One patient developed CSF leakage after skull base reconstruction due to necrosis of the vascularized flap. Another patient developed hydrocephalus of normal pressure. In that case, we first performed a ventriculoperitoneal shunting and then did a reoperation.
Meningitis
8 patients (3.1%) developed meningitis. All of them were patients with intraoperative CSF leakage. Six of them had CSF leakage postoperatively. They were treated with ceftriaxone (4.0 per day) or vancomycin (2.0 per day) and meropenem (6.0 per day). The treatment was successful and no neurological deficit after meningitis was observed.
Epistaxis
One patient (0.4%) developed postoperative epistaxis in 6 hours after surgery and required reoperation. This patient was operated using a unonostril approach. The sphenoidal sinus was opened on both sides but the mucosa on the opposite side was left intact. The bleeding originated from the opposite - side ramus of sphenopalatina artery.
New Visual Deficit
6 patients (2.3%) (2 had a macroadenoma and 4 - giant adenomas) developed a new visual deficit after surgery. In 3 cases of giant adenoma, the patients had both vision and visual fields impairment. The other patients had only visual acuity deterioration. In 5 cases, these new symptoms were gone after treatment. One patient did not have any improvement despite of the treatment.
Hemorrhage/Swelling of Residual Tumor
There was one case of thalamus hemorrhage that required external ventricular drainage. This patient developed a new neurological deficit (diencephalon syndrome), which regressed after the treatment. One more patient developed an asymptomatic ventricular hemorrhage after total removal of the macroadenoma that did not require any additional treatment. A third patient had swelling of residual tumor after partial resection and was reoperated.
Surgical Mortality
The surgical mortality rate was 0.8% (2 patients). The first patient had macroadenoma and Cushing’s disease. The tumor had an invasive growth. It was almost unsuctionable and therefore was not totally removed. After the operation, the patient developed hemiparesis followed in 8 days by deep vein thrombosis and cavafilter implantation. Within the next 7 days, the patient developed pulmonary embolism and on the 34th day after the operation he died. The second patient was a 54-year-old woman with a nonfunctional macroadenoma. During the operation we had no difficulties, and the tumor was removed gross totally. The first day after the surgery she was verticalized and developed pulmonary embolism. A cavafilter was implanted, and thrombus fragmentation was performed. The patient died on the 4th day after the surgery.
Diabetes Insipidus
The DI diagnostics was based on the level of electrolytes (serum Na >145 mmol/L), urine-specific gravity (1.004 or less) and excessive volumes of urine (quantified as more than 300 mL of urine per hour for 3 consecutive hours). In this study there were no patients with preoperative DI. After DI was diagnosed all the patients were treated with desmopressin (the dose was titrated by the level of diuresis). In this series of 259 patients, 111 cases of postoperative DI (42.9%) were diagnosed. 21 patients (8.1%) had transient DI and 90 (34.8%) – permanent DI. Since only the early postoperative period had been analyzed we considered transient DI to be the one that regressed during hospital admission. The patients’ data and tumor characteristics are summarized in (Table 5).
Table 5: Demographic characteristics and tumor characteristics in patients with or without postoperative DI.
Discussion
According to the literature, the rate of surgical complications after removing pituitary adenomas varies between 9% [11] and 35.8% [12] (in average 12.1%). The incidence of CSF leakage is about 3.1-4.7% [3,11-20] (minimal is 1.8% [20], maximal is 19.5% [21]). Smith et al. report 0% of CSF leakage in surgery of microadenomas. Contrariwise, in the study of giant adenomas (Koutourousiou M. et al.) rate of CSF leakage is 16.7%. Based on these data, we can conclude that percentage of postoperative CSF leakage depends on a tumor’s size. Another possible complication after endoscopic surgery is meningitis whose mean rate is 2.0-2.7% [21-24]. In most cases, it has been successfully treated by antibacterial therapy [25- 27]. Hemorrhage inside the residual tumor or its swelling occurs in 0.4-4.0% of patients [28,29] and can lead to new visual (0.9-4.4%) [30] or neurological deficit (0.4-2.3%) [31]. The most common are cranial nerve (III, IV, VI) dysfunction, hemiparesis (0.2-1.0%) and diencephalon syndrome [32]. The incidence of epistaxis in the studies rates from 0.4 to 2.9% [32]. In most cases nasal cavity tamponade is effective, but some cases require a revision surgery.
The rate of surgical mortality varies from 0.6 to 1.8% [33]. The main reasons of mortality are hemorrhage, somatic pathology (cardio-vascular disease, acute pancreatitis etc.), meningitis, ischemic stroke due to the damage of internal carotid artery, hard electrolytes irregularities (because of involvement of diencephalon region), pulmonary embolism, subarachnoid hemorrhage (in case of undiagnosed aneurism) [33]. In our study we did not find any difference between complications and DI postoperatively. DI belongs to a group of hereditary or acquired polyuria and polydipsia diseases. It is associated with inadequate arginine vasopressin (AVP) or antidiuretic hormone (ADH) secretion or renal response to AVP, resulting in hypotonic polyuria and a compensatory/ underlying polydipsia. The characteristics of DI polyuria include polyuria (>50 mL/kg), diluteurine (osmolality <300 mOsm/L), and increased thirst (water intake of up to 20 L/day). Untreated DI can cause hypovolemia, dehydration, and electrolyte imbalances. There are different types of DI such are central, nephrogenic, gestational and primary polydypsya. This paper refers only to central DI (CDI) [34].
CDI or neurogenic DI is the most common form of DI, occurring equally in both sexes and at any age. It is caused by inadequate synthesis/release of AVP, often secondary to surgery or to a head injury causing traumatic injury to the hypothalamus or posterior pituitary gland and destruction/degeneration of neurons originating in the supraoptic and paraventricular nuclei of the hypothalamus. DI Symptoms manifest themselves after 80–90% of the magnocellular neurons in the hypothalamus are damaged. Damage to the proximal part of the hypothalamo-neurohypophyseal region kills more neurons than do injuries to the distal region. However, proximal injuries account for 30–40% of posttraumatic and postoperative CDI, while distal injuries account for 50–60% of cases. Acquired CDI is more common than congenital one, and about 25% of adult CDI cases are idiopathic. Children with acute injury to the nervous system and CDI have a high mortality rate. Traumatic brain injury is associated with high mortality and acute and chronic morbidity [34]. Pituitary surgery results in CDI with a wide range of incidence (1–67%). Minimally invasive surgery has a low incidence of postoperative CDI (transient CDI 13.6% and permanent CDI 2.7%). Abnormalities in the secretion of AVP usually begin during the intraoperative period. The incidence of immediate postoperative DI is higher among the patient’s undergoing surgery by the traditional method than by the endoscopic transsphenoidal method (36% vs. 15%). The incidence of long-term DI did not vary significantly between the groups [34].
The history of desmopressin started from the first use of vasopressin to treat CDI in 1913. That time it was pituitary extracts containing oxytocin and vasopressin. Pitressin (vasopressin tannate in oil) became available for clinical use in the 1930s and was the principal drug for DI treatment almost until the 1970s when desmopressin was introduced. Substitution of the 8th residue of vasopressin with the D-isomer of arginine diminished the pressor effect of vasopressin. This synthetic analog of vasopressin, 1‑deamino‑8‑D‑AVP (DDAVP), or desmopressin, has increased and prolonged antidiuretic effect, decreased vasoconstrictive and oxytocic effects, reduced side effects (hyponatremia and convulsions), and enhanced resistance to vasopressinase. It can be administered in the form of intranasal spray, oral tablets, and orally disintegrating tablets (ODT) [34]. In our study, the patients received only the oral tables. The minimal initial dose of desmopessin is 0.1 mg. The titration control included blood electrolytes, urine-specific gravity and production of excessive volumes of urine. The maximum dose is 0.2 mg twice a day. In this study an attempt was made to determine the relation between postoperative DI frequency and different characteristics of adenomas, surgical complications and extent of resection.
In the literature, the rate of DI is estimated to be between 8.7% and 18.3%. In their study of giant adenomas M. Koutourousiou et al. have reported 9.6% rate of permanent DI, and PG Campbell et al. in their study of growth hormone secreting adenomas have detected no cases of permanent DI while transient DI have occurred in 7.7% of patients. Multivariate analysis of our cases showed the predictors of DI were patient’s age, size and consistence of tumor. In our study, we revealed the influence of adenomas size relative to a patient’s age. For older patients the size of the adenoma was a stronger factor than for the younger ones. In other words, in the elderly patients the tumor size has a greater effect on the risk of DI development. These data contradict to the results obtained by Sigounas et al. in 2008 who has not revealed any correlation between DI and tumor size. In our opinion our findings are related to the anatomy of a pituitary gland. The older a patient the more prone the gland is to hypoplasia while the risk its injury is lower, especially in case of microadenomas. Macroadenomas and giant adenomas are often found in close proximity to the pituitary gland that increases the risk of its injury and DI development after surgery. In case of giant adenomas, the last squeeze the gland a lot, but in case of microadenomas, when we usually observe healthy hypophysis tissue, the hypoplasia will play a major role.
The second important factor was consistence of tumor. The unsuctionable adenomas increased the risk of DI 2.89 times. This dependency also can be related to the tumor’s intimacy to the gland making the last more prone to injury and increasing the risk of DI development. The rate of all complications also depends on the surgeon’s experience. Ciric et al. analyzed complications in three groups of patients who were operated by surgeons with experience less than 200, 200-500 and more than 500 cases respectively. It has been demonstrated that those patients who were operated by surgeons with experience less than 200 surgeries (as in our study) had higher incidence of not only surgical complications, but also higher rate of DI (19%).
Conclusion
The endoscopic transsphenoidal surgery is the treatment of choice for patients with pituitary adenomas. Complications are rather common after endoscopic surgery and include two big groups that are surgical and endocrine complications. Further studies into the incidence and causes of postoperative diabetes insipidus and other complications can contribute to the reduction of both types. We can conclude that the predictors of DI include the patient’s age, size and consistence of adenomas. The influence of adenomas size depends on the age of a patient. The risk of DI in 30-year-old patients has been 1.51 times higher with increasing of the size group by one grade (micro, macro, giant), while in the elder patients (60-year-old) it has increased 2.3 times. Nevertheless, in case of giant adenomas the age has not been the impact factor to trigger DI after surgery. The tight consistence of tumor has been another important factor to provoke DI after surgery. The unsustainable consistency increases the risk of DI 2.89 times. The hormone activity of adenomas, prior surgery, extent of surgery have not been associated with DI development. The DI rate after endoscopic resection is 42.9% (in 8.1% of cases the patients developed transient DI and 34.8% - its permanent form).
References
- Halvorsen H, Ramm-Petersen J, Josefsen R, Pål Rønning, Sissel Reinlie, et al. (2014) Surgical complications after transsphenoidal microscopic and endoscopic surgery for pituitary adenoma: a consecutive series of 506 procedures. Acta Neurochir 156(3): 441-449.
- In: B A Kadashev (Eds.)., (2007) Pituitary adenomas: clinical symptoms, diagnostics, treatment. Moscow.
- Dehdashti AR, Ganna A, Karabatsou K (2008) Pure Endoscopic Endonasal Approach for Pituitary Adenomas: Early Surgical Results in 200 Patients and Comparison with Previous Microsurgical Series. Neurosurgery 62(5): 1006-1015.
- Kassam AB, Prevedello DM, Carrau RL, Carl H Snyderman, Ajith Thomas, et al. (2011) Endoscopic endonasal skull base surgery: analysis of complications in the authors initial 800 patients. J Neurosurg 114(6): 1544-1556.
- Yang Gao, Chunlong Zhong, Yu Wang, Siyi Xu, Yang Guo, et al. (2014) Endoscopic versus microscopic transsphenoidal pituitary adenoma surgery: a meta-analysis. World J Surg Oncol 12: 94.
- Smith TR, Hulou MM, Huang KT, Breno Nery, Samuel Miranda de Moura, et al. (2015) Complications after transsphenoidal surgery for patients with Cushing's disease and silent corticotroph adenomas. Neurosurg Focus 38(2): E12.
- Sigounas DG, Sharpless JL, Cheng DML, Tiffany G Johnson, Brent A Senior, et al. (2008) Predictors and incidence of central diabetes insipidus after endoscopic pituitary surgery. Neurosurgery 61(1): 71-79.
- R Core Team (2017) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria.
- David W (2000) Hosmer Stanley Lemeshow. Applied Logistic Regression (2nd)., 2000 John Wiley & Sons, Inc.
- Koutourousiou M, Paluzzi A, Tormenti MJ (2012) Giant pituitary adenomas: advantages and limitations of endoscopic endonasal surgery. J Neurol Surg B 73-A119: 2012.
- In: Kassam AB, Gardner PA (Eds.)., (2012) Endoscopic approaches to the skull base. Basel; Karger.
- Koutourousiou M, Filho FVG, Fernandez-Miranda JC, et al. (2014) Endoscopic endonasal surgery for pituitary adenomas with cavernous sinus invasion: experience with 175 cases. J Neurol Surg B, 75-A148.
- Hae-Dong Jho, Ricardo L Carrau (1997) Endoscopic endonasal transsphenoidal surgery: experience with 50 patients. J.Neurosurg 87(1): 44-51.
- Campbell PG, Kenning E, Andrews DW, Sanjay Yadla, Marc Rosen, et al. (2010) Outcomes after a purely endoscopic transsphenoidal resection of growth hormone-secreting pituitary adenoma. Neurosurg Focus 29 (4): E5.
- Thomas JG, Gadgil N, Samson SL, Masayoshi Takashima, Daniel Yoshor, et al. (2014) Prospective Trial of a Short Stay Protocol After Endoscopic Pituitary Adenoma Surgery. Word Neurosurgery 3/4: 576-583.
- Gondim JA, Almeida JP, De Albuquerque LA, Erika Gomes, Michele Schops, et al. (2015) Endoscopic endonasal transsphenoidal surgery in elderly patients with pituitary adenomas. J Neurosurg 123(1): 31-38.
- Chin-Hsun Lee, Shu-Mei Chen, Tai-Ngar Lui (2015) Posterior Cerebral Artery Pceudoaneurism, a Rare Complication of Pituitary Tumor Transsphenoidal Surgery: Case Report and Literature Review. World Neurosurgery 84(5): 1493.
- Dusick JR, Esposito F, Mattozo CA, Charlene Chaloner, David L McArthur, et al. (2006) Endonasal Transsphenoidal surgery: the patient’s perspective – survey results from 259 patients. Surgical Neurology 65(4): 332-342.
- De Divitiis E, Cappabianca P, Cavallo LM (2003) Endoscopic endonasal transsphenoidal approach to the sellar region. Vienna: Springer Vienna.
- Thorp BD, Sreenath SB, Ebert CS (2014) Endoscopic skull base reconstruction: a review and clinical case series of 152 vascularized flaps used for surgical skul base defects in the setting of intraoperative cerebrospinal fluid leak. Neurosurg Focus 37(4): E4.
- Mascarenhas L, Moshel YA, Bayad F, Oszkar Szentirmai, Al Amin Salek et al. (2014) The transplanum transtuberculum approaches for suprasellar and sellar-suprasellar lesions: avoidance of cerebrospinal fluid leak and lessons learned. World Neurosurg 82(1-2): 186-195.
- Goudakos JK, Markou KD, Georgalas C (2011) Endoscopic versus microscopic trans-sphenoidal pituitary surgery: a systematic review and meta-analysis. Clin Otolaryngol 36(3): 212-220.
- Cappabianca P, Cavallo LM, Colao AM (2002) Surgical complications associated with the endoscopic endonasal transsphenoidal approach for pituitary adenomas. Journal Neurosurgery 97(2): 293-298.
- Kono Y, Prevedello DM, Snyderman CH, Paul A Gardner, Amin B Kassam, et al. (2011) One thousand endoscopic skull base surgical procedures demystifying the infection potential: incidence and description of postoperative meningitis and brain abscess. Infection control and hospital epidemiology, January 32(1): 77-83.
- Ivan MI, Iorgulescu JB, El-Slayed I, Michael W McDermott, Andrew T Parsa, et al. (2015) Risk factors for postoperative cerebrospinal fluid leak and meningitis after expanded endoscopic endonasal surgery. Journal of clinical neuroscience 22(1): 48-54.
- Smith TR, Hulou MM, Huang KT, Breno Nery, Samuel Miranda de Moura, et al. (2015) Complications after transsphenoidal surgery for Cushung disease. J Neurol Surg B 76-A010.
- McCoul ED, Bedrosian JC, Akselrod O, Vijay K Anand, Theodore H Schwartz, et al. (2015) Preservation of multidimentional quality of life after endoscopic pituitary adenoma resection. J Neurosurg 123(3): 813-280.
- Wilson D, Jin DL, Wen T (2015) Demographic factors, outcomes, and patient access to transsphenoidal surgery for Cushing’s disease: analysis of the Nationwide Inpatient Sample from 2002 to 2010. Neurosurg Focus 38.
- Dallapiazza RF, Grober Y, Starke RM, Edward R Laws Jr, John A Jane Jr, et al. (2015) Long-tern Results of Endonasal Endoscopic Transsphenoidal Resection of Nonfunctioning Pituitary Macroadenomas. Materials of Congress Neurological Surgeons. Neurosurgery 76(1): 42-52.
- Oruckaptan HH, Senmevsim O, Ozcan OE (2000) Pituitary Adenomas: results of 684 Surgically treated Patients and Review of the Literature. Surgical Neurology 53(3): 211-219.
- Zolli M, Mazzatenta D, Valuzzi A (2015) Endoscopic endonasal treatment of pituitary adenomas invading the cavernous sinus: surgical outcome in 374 patients. J Neurol Surg B 76-A107.
- Pashaev BYu, Vagapova GR, Bochkarev DV (2012) Transnasal surgery of pituitary adenomas in Republic of Tatarstan. Practical medicine 09.
- Matsuno A, Yoshida S, Basugi N (1993) Severe Subarachoid Hemorrhage During Transsphenoidal Surgery for Pituitary Adenoma. Surg Neurol 39(4): 279-288.
- Kalra S, Zargar AH, Jain SM, Bipin Sethi, Subhankar Chowdhury, et al. (2016) Diabetes insipidus: the other diabetes. Indian Journal of Endocrinology and Metabolism 20(1): 9-21.

 Research Article
Research Article