ABSTRACT
Circinella, Mucor and Rhizopus all these genera belonging to Order: Mucorales. This review is to demonstrate description, ecology, and secondary metabolites from Circinella, Mucor and Rhizopus and some of their reported biological activities. Besides, describing the importance and potentials of those fungi in order to encourage researchers for further studies to each genus metabolites and purify already known metabolites.
Keywords: Circinella; Mucor; Rhizopus; Secondary Metabolites; Bio transformations
Introduction
Fungi are rich sources of biologically active natural compounds, which are used in the manufacturing of wide range of clinically important drugs. Fungi produce important antibiotics such as the beta-lactam antibiotics members, penicillin and cephalosporin, which and their derivatives are dominating the most important antibiotic market until now [1-5]. Fungi generally and endophytic ones specifically represent future factories and potent biotechnological tools for production of bioactive natural substances which could extend healthy life span of humanity, as done by penicillin from centuries, and considered promising alternatives for some high costly produced chemicals and drugs [6- 12]. The present review highlights some bioactivity of secondary metabolites, produced by Circinella, Mucor, and Rhizopus, involved in medical, pharmaceutical, agricultural, and industrial applications. Zygomycetes constitute a remarkable group of microscopic fungi. These fungi are mainly soil inhabitants living as saprobes and decomposers of organic matter and herbivorous feces. Zygomycota, represent the most basal terrestrial phylum of the kingdom of Fungi [13].
Zygomycetes have an integral role in the development of microbial ecosystems, a property which has the potential to be converted for biotechnological and industrial applications ranging from food technology to drug development [14]. The Mucorales, which is classified into the subphylum Mucormycotina [15], is the largest order of fungi. Members of this group (Circinella, Mucor and Rhizopus) are abundant saprophytes in nature. They are commonly found in soil and decaying vegetation, and can also be found in grains [16,17]. Mucorales members (Example; Circinella, Mucor and Rhizopus) grow and invade quickly on easily digestible substrates, such as those containing starches, sugars, and hemicelluloses. Future studies should investigate Circinella, Mucor and Rhizopus their ability to produce extracellular enzymes and potential applications in biotechnology.
Circinella, Mucor and Rhizopus Description and Ecology
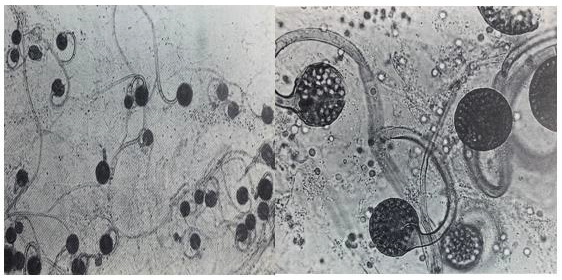
The Genus Circinella belonging to Phylum: Mucoromycota; Class: Zygomycetes; Order: Mucorales; Family: Syncephalastraceae. Colonies mucraceous, usually up to few mm high; sporangiophores erect, branching sympodially and each branch recurved (Circinate) terminated with a sporangium; sporangia globose, columellate, many-spored, covered with non-diffluent wall, incrusted with calcium oxalate, breaking into pieces; columellae globose, subglobose or cylindro-conic; sporangiospores globose or oval, smooth; heterothallic; zygospores when produced, born on erect hyphae, with more or less similar gametangia and suspensors. Colonies fast growing, attaining 7.5 cm after 7 days on malt extract agar at 25C. The most common Circinella species is Circinella muscae. Circinella colonies color is brownish and common present in soil (Figure 1) [11,13]. The Genus Mucor belonging to Phylum: Mucoromycota; Class: Zygomycetes; Order: Mucorales; Family: Mucoraceae. Colonies fast growing, often up to several cm in height, white to yellow becoming dark grey with age; mycelium non-septate, occasionally septa produced in old cultures; sporangiophores erect arising singly from the mycelium without rhizoids, forming a dense mat, unbranched or branched monopodially or sympodially, sometimes the branches recurved, bearing terminal sporangia; sporangia globose, many-spored, columellate, without apophysis; sporangial wall deliquescent, incrusted with needles of calcium oxalate; columellae of various shapes, colourless or coloured; sporangiophores colourless or colored, greyish or brownish, globose or elliptical, wall thin smooth; chlamydospores produced in some species , terminal, hyaline, smooth walled; zygospores produced in the aerial mycelium and arising from copulation of two straight gametangia.
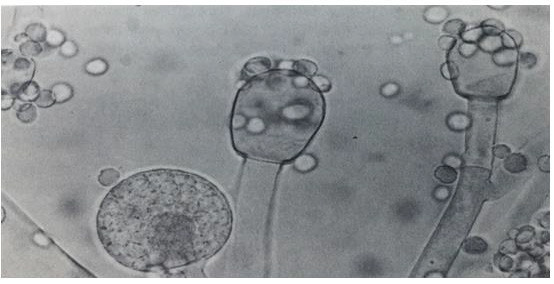
Mucor is one of the largest genera of Order: Mucorales and a number of its species are worldwide. Mucor species are common in soil and also occur organic matter. Colonies fast growing, filling the plate of malt extract agar after 3 days at 25C (Figure 2). The most common Mucor species is Mucor circinelloides, Mucor hiemalis and Mucor racemosus [11,13]. The Genus Rhizopus belonging to Phylum: Mucoromycota; Class: Zygomycetes; Order: Mucorales; Family: Mucoraceae. Colonies fast growing, filling the whole plate after 2 days on malt extract agar at 25C, at first white, later becoming pale or dark grey brown. Mycelium non-septate, occasionally septa produced in old cultures, differentiated into stolons, rhizoids arising from stolon ends and erect unbranched sporangiophores sporouting opposite the rhizoids; sporangia generally globose, apophysate, columellate, many-spored; sporangiospores short ellipsoidal, brownish, striate in many species; most species heterothallic; zygospores naked, dark-coloured; wall strongly roughened, suspensors straight, large and swollen. Rhizopus species are common in soil and soil containing various organic matter. Rhizopus species are impotent spoilage organisms. The most common Rhizopus stolonifer, Rhizopus arrhizus and Rhizopus oryzae (Figure 3) [11,13].
The Genus Circinella as a Biotransformation Agent
Enzymatic reactions and biotransformations catalyzed by fungal enzymes and used in industry, agriculture, food technology, and medicine have increased in importance tremendously in recent years. It was later demonstrated that filamentous fungi from several genera, including Circinella, Mucor and others, are able to hydroxylate dibenzofuran, forming 1-, 2-, 3-, or 4-hydroxybenzofuran and some other more hydrophilic products [18]. Yan et al., [19], mentioned that, nine hydroxylated and glycosylated metabolites, of which two have not been reported previously, were obtained and identified after incubation by Circinella muscae. The specific 7β, 15α, and 21β hydroxylated products yielded from this bio-process by Circinella muscae. In addition, the selectively glycosylation at C-28 was another main reaction type. It was also observed that the 3β-OH group was selectively dehydrogenated into carbonyl group. These reactions may be difficult to achieve by chemical synthetic means. In vitro biological tests indicated that compounds 3β, 7β dihydroxyolean-12-en-28-oic acid; 3β, 7β, 15α-trihydroxyolean- 12-en-28-oic acid, and 3β, 7β, 15α-trihydroxyolean-12-en- 28- oic acid-28-Oβ-D-glucopyranosyl ester showed significant antiinflammatory activities, which suggested that hydroxylation at C-7 and glycosylation at C-28 by Circinella muscae had benefit effects [19]. Macromycete Circinella muscae AS 3.2695, catalyzed the oxidation of Oleanolic Acid [20]. Biotransformation of ursolic acid by Circinella muscae CGMCC 3.2695 was investigated by Chu et al., [21].
Scaled-up biotransformation reactions yielded ten metabolites. Their structures were established based on extensive NMR and HR-ESI-MS data analyses, and four of them are new compounds. Circinella muscae could selectively catalyze hydroxylation, lactonisation, carbonylation and carboxyl reduction reactions. Also, all the identified metabolites were evaluated for their anti-neuroinflammatory activities in LPS-induced BV-2 cells. Most metabolites displayed significant inhibitory effect on nitric oxide (NO) production and the results suggested that biotransformed derivatives of ursolic acid might be served as potential neuroinflammatory inhibitors [21]. Biotransformation of betulinic acid was carried out with Circinella muscae CGMCC 3.2695 and Cunninghamella echinulata CGMCC 3.970 by Chen et al., [22], yielded six previously undescribed hydroxylated metabolites and four known compounds. Circinella muscae could catalyze the regioselecitve hydroxylation and carbonylation at C-3, C-7, C-15 and C-21 to yield seven products. Cunninghamella echinulata could catalyze the C-1, C-7 and C-26 regioselecitve hydroxylation and acetylation to yield five metabolites. The structures of the metabolites were established based on extensive NMR and HR-ESIMS data analyses and most of the metabolites exhibited significant inhibitory activities on lipopolysaccharides-induced NO production in RAW264.7 cells [22].
The Genus Mucor Source of Biologically Active Compounds
Species of Mucor ramosissimus produce extracellular enzymes, such as endopolygalacturonase and lipase, and secondary metabolites, such as phytoalexin elicitor [23,24]. Lately, several studies have focused on applying Mucorales members to produce ethanol and biomass by-product. Particularly, Mucor ramosissimus has been reported as a potential ethanol-producing mold [25]. Mucor circinelloides has a great industrial potential since it started producing bioactive compounds of interest, such as microbial lipids, carotenoids or sterols [26,27]. Mucor circinelloides is able to produce microbial lipids which could be useful for the production of biodiesel using the common way to produce fatty acid methyl esters, due to the beneficial properties of these compounds, new sustainable and more environmentally friendly methods have emerged in order to increase their production [28]. Three important strains of Mucor circinelloides grown in specific media for specified period (72 h, 120 h and 168 h) under submerged fermentation conditions were investigated by Hameed et al., [29], for their potential antioxidants/secondary metabolite production and found that all mycelial extracts demonstrated effective antioxidant activities in terms of β-carotene/linoleic acid bleaching, radical scavenging, reduction of metal ions and chelating abilities against ferrous ions [29].
Antioxidant property of all three important strains of Mucor circinelloides extracts were duo to their phenolic and condensed tannin contents. Strains Mucor circinelloides MC277.49 was found to be the biggest producer of secondary metabolites under nutritional stress condition in late exponential phase. These Mucor strains prove to be rich sources of antioxidants and secondary metabolites, which could be used in the development of nutraceuticals and natural antioxidants [29]. The new cyclic heptapeptide unguisin F and the known congener unguisin E, were obtained from the endophytic fungus Mucor irregularis, isolated from the medicinal plant Moringa stenopetala, collected in Cameroon studied by [30]. The structure of the new compound was determined on the basis of one- and two-dimensional NMR spectroscopy as well as by high-resolution mass spectrometry. Heptapeptide unguisin F and congener unguisin E were evaluated for their antibacterial and antifungal potential, but failed to display significant activities [30]. Biotransformation is an important tool for the structural modification of organic compounds, especially natural products with complex structures, which are difficult to achieve using ordinary methods [31].
Mucor genus is widespread in nature and some species are used extensively in biotechnology for enzyme and useful compound production. Biotransformation can be used as a very convenient way of producing compounds, particularly when the structure is complex and they can neither be isolated as metabolites nor chemically synthesized [31]. It is safe to conclude that biotransformation by Mucor species is of great importance due to their wide-ranging use in the stereospecific production of compounds of commercial interest, and because they simplify the study of the metabolism of such compounds in order to obtain novel agents with many interesting biological activities [31]. The biotransformation of 1R-(−)-camphorquinone, achieved by growing cells of Mucor plumbeus isolated from soil. Results were found for M. plumbeus, which was only able to perform monoreduction of camphorquinone when cultivated on a glucose– peptone–yeast extract medium. Large-scale experiments were set up and the camphorquinone biotransformation products formed by Mucor plumbeus were purified by column chromatography and identified by 1H and 13C nuclear magnetic resonance (NMR). Mucor plumbeus could be of great use for the selective reduction of camphorquinone and related compounds [32].
The Genus Rhizopus Source of Biologically Active Compounds
Secondary metabolites is one of the characteristic features of microorganisms. More than 50,000 bioactive compounds have been isolated from the extracts of microorganisms with a diversified arrangement of chemical structures, which showed antimicrobial, antitumor and agrochemical activity and others [33]. The ethyl acetate extract of Rhizopus stolonifer has potent cytotoxic activity against brine shrimps, suggesting that it could serve as a lead compound for anticancer compounds. Its antifungal activity also indicates its potential for development into an antifungal product. Furthermore, its phytotoxicity against Lemna minor weed demonstrates that it contains compounds with herbicidal activity. More studies on Rhizopus stolonifer is needed to isolate and structural characterization of its constituents which could lead to the development of pharmaceutical and agricultural products [34].
From previous studies, it was concluded that the extracts of Rhizopus species have compounds which have ability to inhibit the growth of fungal and bacterial strains [35]. Chitosan was isolated from the fungus Rhizopus oryzae by using yeast peptone glucose broth medium, showed antimicrobial activity against Escherichia coli, Staphylococcus aureus and Candida albicans and reach to 20, 15 and n 15 mm as inhibition zone respectively [36]. Nanotechnology is a field that is increasing day by day, making an impact in all spheres of human life. Biological methods of synthesis nanoparticles called “greener synthesis” of nanoparticles and these have proven to be better methods due to slower kinetics, they offer better manipulation and control over crystal growth and their stabilization [37]. The extracellular synthesis of silver nanoparticles by Rhizopus stolonifer and its efficacy against multidrug resistant (MDR) strains isolated from burnt cases from hospitals at Gulbarga region, Karnataka, India is reported by Rathod and Ranganath, [38], the biosynthesized nanosilver showed excellent antibacterial activity against multidrug resistant Pseudomonas aeruginosa isolated from burnt infections. Biologically synthesized nanosilver showed zone of inhibition (mm) of two isolates about 33mm and 30.5mm in diameter strains of Pseudomonas aeruginosa respectively [38].
This study reports the extracellular synthesis of silver nanoparticles by Rhizopus stolonifer and its efficacy against multidrug resistant (MDR) Escherichia coli and Staphylococcus aureus isolated from Khwaja Bande Nawas Hospital, Gulbarga, Karnataka was carried by Banu and Rathod, [39] and synthesis of silver nanoparticles (AgNPs) was carried out by using fungal filtrate of Rhizopus stolonifer and an aqueous solution of AgNO3. Biotransformations of sesquiterpenoids by Rhizopus species have been used to provide new derivatives with potential biological activities. Fungal transformation processes by Rhizopus species, has been the introduction of hydroxyl groups into remote positions of the molecules, which is difficult to achieve by chemical means. Other reactions carried out with stereo-selectivity by these microorganisms have been: epoxidations of double bonds, hydrogenations of the exocyclic C-C double bond of the lactone rings, and reductions of carbonyl groups giving Salcohols. Several reactions have also been performed by these Rhizopus species with regio-selectivity, such as oxidations of hydroxyl groups and deacetylations [40]. The enttrachyloban diterpene ent-18, 19-dihydroxytrachylobane was biotransformed by Rhizopus stolonifer, and produced the new ent- 11β, 18, 19-trihydroxytrachylobane, and the new ent-kaurene diterpenes ent-16α, 18, 19-trihydroxykaur-11-ene and ent- 18, 19-dihydroxy-16α-methoxykaur-11-ene. The formation of derivative ent-11β,18,19-trihydroxytrachylobane, involved the first hydroxylation of C-11 of ent-trachyloban diterpene skeleton by fungus, and compounds ent-kaurene diterpenes ent-16α, 18, 19-trihydroxykaur-11-ene and ent-18,19-dihydroxy-16α- methoxykaur-11-ene were probably produced by backbone rearrangement of ent-11β,18,19-trihydroxytrachylobane, these results, verified the potential application of microbial transformation (Especially By Mucorales Rhizopus species) of natural products for the formation of new compounds [41].
Conclusion
Fungi in general are important source of unique natural products with a high level of biodiversity and also yield several compounds having different industrial applications and pharmaceutical activities, which is currently attracting scientific researches. Every study conducted on Circinella, Mucor and Rhizopus resulted in discovery of new metabolites that may have an important applications, which made these genera potential source of pharmaceuticals and attracted attention for further investigations of their important bioactivities properties. Circinella, Mucor and Rhizopus are known for their capability of producing various bioactive compounds with medical applications as antineuroinflammatory, antiinflammatory, and enzymes producers and as biotransformation agents.
References
- Elkhateeb WA, Daba GM (2018) Where to Find? A Report for Some Terrestrial Fungal Isolates, and Selected Applications Using Fungal Secondary Metabolites. Biomed Journal Science &Technology Research 4(4): 1-4.
- Elkhateeb WA, Daba GM (2019) The amazing potential of fungi in human life. ARC J Pharma Sci AJPS 5(3): 12-16.
- Elkhateeb WA, Daba GM (2019) Epicoccum species as potent factories for the production of compounds of industrial, medical, and biological control applications. Biomedical Journal of Scientific and Technical Research 14(3): 10616-10620.
- Elkhateeb WA, Daba GM (2019) Myrothecium as promising model for biotechnological applications, potentials and challenges. J Sci Res 16(3): 12126-12131.
- Daba GM, Mostafa FA, Elkhateeb WA (2021) The ancient koji mold (Aspergillus oryzae) as a modern biotechnological tool. Bioresources and Bioprocessing 8(1): 1-17.
- Daba GM, Elkhateeb WA, Thomas PW (2018) This era of biotechnological tools: an insight into endophytic mycobiota. Egyptian Pharmaceu J 17(3): 121-128.
- Elkhateeb WA, EL Ghwas DE, AL Kolaibe AG, Akram M, Daba GM (2021) Yeast the Present and Future Cell Facture. Open Access Journal of Mycology & Mycological Sciences 4(2): 1-5.
- Elkhateeb WA, Elnahas MO, Daba GM, Zohri AN (2021) Biotechnology and Environmental applications of TrichodermaResearch Journal of Pharmacognosy and Phytochemistry 13(3): 149-157.
- Elkhateeb WA, Kolaibe AG, Daba GM (2021) Cochliobolus, Drechslera, Bipolaris, Curvularia different nomenclature for one potent fungus. Journal of Pharmaceutics and Pharmacology Research 4(1): 1-6.
- Elkhateeb WA, Kolaibe AG, Elnahas MO, Daba GM (2021) Highlights on Chaetomium morphology, secondary metabolites and biological activates. Journal of Pharmaceutics and Pharmacology Research 4(1): 1-5.
- Elkhateeb WA (2005) Some mycological, phytopathological and physiological studies on mycobiota of selected newly reclaimed soils in Assiut Governorate, Egypt (M. Sc. Thesis, Faculty of Science, Assuit University, Egypt, pp. 238.
- Elkhateeb WA, Zohri AA, Mazen M, Hashem M, Daba GM (2016) Investigation of diversity of endophytic, phylloplane and phyllosphere mycobiota isolated from different cultivated plants in new reclaimed soil, Upper Egypt with potential biological applications. Inter J MediPharm Res 2(1): 23-31.
- Voigt K (2012) Zygomycota. Syllabus of plant families – A. Engler’s Syllabus der Pflanzenfamilien. Part 1/1: Blue-green algae, Myxomycetes and Myxomycete-like organisms, Phytoparasitic protists, Heterotrophic Heterokontobionta and Fungi. In: Frey W (Edt.)., Borntraeger Verlag, Germany, pp. 130-162.
- Voigt K, Wolf T, Ochsenreiter K, Nagy G, Kaerger K, et al. (2016) 15 Genetic and Metabolic Aspects of Primary and Secondary Metabolism of the Zygomycetes. In Biochemistry and molecular biology. Springer, Cham, pp. 361-385.
- Hibbett DS, Binder M, Bischoff JF (2007) A higher-level phylogenetic classification of the fungi. Mycol Res 111(5): 509-547.
- Hoffmann K, Pawłowska J, Walther G (2013) The family structure of the Mucorales: a synoptic revision based on comprehensive multigene-genealogies. Persoonia 30: 57-76.
- Walther G, Pawłowska J, Alastruey Izquierdo A (2013) DNA barcoding in Mucorales: an inventory of biodiversity. Persoonia 30: 11-47.
- Hammer E, Krowas D, Schafer A, Specht M, Francke W, et al. (1998) Isolation and characterization of a dibenzofuran-degrading yeast: Identification of oxidation and ring cleavage products. Appl Environ Microbiol 64(6): 2215-2219.
- Yan S, Lin H, Huang H, Yang M, Xu B, et al. (2019) Microbial hydroxylation and glycosidation of Oleanolic acid by Circinella muscae and their anti-inflammatory activities. Natural product research 33(13): 1849-1855.
- Luchnikova NA, Grishko VV, Ivshina IB (2020) Biotransformation of oleanane and ursane triterpenic acids. Molecules 25(23): 5526.
- Chu C, Song K, Zhang Y, Yang M, Fan B, et al. (2021) Biotransformation of ursolic acid by Circinella muscae and their anti-neuroinflammatory activities of metabolites. Natural Product Research 12: 1-6.
- Chen C, Song K, Zhang Y, Chu C, Fan B, et al. (2021) Biotransformation of betulinic acid by Circinella muscae and Cunninghamella echinulata to discover anti-inflammatory derivatives. Phytochemistry 182: 112608.
- Nguyen TT, Lee HB (2018) Isolation and characterization of three Zygomycetous fungi in Korea: Backusella circina, Circinella muscae, and Mucor ramosissimus. Mycobiology 46(4): 317-327.
- Marques MR, Buckeridge MS, Braga MR (2006) Characterization of an extracellular endopolygalacturonase from the saprobe Mucor ramosissimus Samutsevitsch and its action as trigger of defensive response in tropical plants. Mycopathologia 162(5): 337-346.
- Satari B, Karimi K (2018) Mucoralean fungi for sustainable production of bioethanol and biologically active molecules. Appl Microbiol Biotechnol 102(3):1097-1117.
- Nicolás FE, Calo S, Murcia Flores L, Garre V, Ruiz-Vázquez RM, et al. (2008) A RING-finger photocarotenogenic repressor involved in asexual sporulation in Mucor circinelloides. FEMS Microbiol Lett 280(1): 81-88.
- Rodríguez Frómeta RA, Gutiérrez A, Torres Martínez S, Garre V (2013) Malic enzyme activity is not the only bottleneck for lipid accumulation in the oleaginous fungus Mucor circinelloides. Appl Microbiol Biotechnol 97(7): 3063-3072.
- Vicente G, Bautista LF, Rodríguez R, Gutiérrez FJ, Sádaba I, et al. (2009) Biodiesel production from biomass of an oleaginous fungus. Biochem Eng J 48(1): 22-27.
- Hameed A, Hussain SA, Yang J, Ijaz MU, Liu Q, et al. (2017) Antioxidants potential of the filamentous fungi (Mucor circinelloides). Nutrients 9(10): 1101.
- Akone SH, Daletos G, Lin W, Proksch P (2016) Unguisin F, a new cyclic peptide from the endophytic fungus Mucor irregularis. Zeitschrift für Naturforschung C J 71(1-2): 15-19.
- De Oliveira Silva E, Furtado NA, Aleu J, Collado IG (2015) Non-terpenoid biotransformations by MucorPhytochemistry Reviews 14(5): 745-764.
- De Souza GG, Anconi CP, Cornelissen S, De Almeida WB, Dos Santos HF, et al. (2009) Selective activity of Mucor plumbeus reductase towards (-)-camphorquinone. Journal of Industrial Microbiology and Biotechnology 36(8): 1023-1027.
- Nikapitiya C (2012) Bioactive Secondary Metabolites from Marine Microbes for Drug Discovery. Marine Medicinal Foods: Implications and Applications. Animals and Microbes 65: 363-387.
- Iqbal M, Amin M, Iqbal Z, Bibi H, Iqbal A, et al. (2014) Antimicrobial, cytotoxic and phytotoxic potency of ethyl acetate extract of Rhizopus stoloniferTropical Journal of Pharmaceutical Research 13(1): 87-92.
- Rahman WK, Asghar A, Ullah I, Numan M (2014) Antimicrobial activity of mycelial extracts of Rhizopus stolonifer against different fungal and bacterial pathogenic strains. International Journal of Biosciences 4(7): 276-281.
- Khalaf KT (2016) Antimicrobial activity of chitosan isolated from fungus Rhizopus oryzae. Al-Qadisiyah Journal of Pure Science 21(1): 41-48.
- Hiremath J, Rathod V, Ninganagouda S, Singh D, Prema K (2014) Studies on biosynthesis of silver nanoparticles using Rhizopus and its antibacterial efficacy on E. coli MDR strains. International Letters of Natural Sciences 18: 1-10.
- Rathod V, Ranganath E (2011) Synthesis of monodispersed silver nanoparticles by Rhizopus stolonifer and its antibacterial activity against MDR strains of Pseudomonas aeruginosa from burnt patients. International Journal of Environmental Sciences 1(7): 1830-1840.
- Banu A, Rathod V (2011) Synthesis and characterization of silver nanoparticles by Rhizopus stolonifer. Int j biomed adv Res 2(5): 148-58.
- García-Granados A, Rivas F, Martínez A, Parra A (2009) Biotransformations of Sesquiterpenoids by Rhizopus Species. Current Organic Chemistry 13(12): 1182-1193.
- Vasconcelos DH, Mafezoli J, Uchôa PK, Saraiva NN, Lima MA, et al. (2015) Biotransformation of the Diterpene Ent-18, 19-dihydroxytrachylobane by Rhizopus stolonifer.Journal of the Brazilian Chemical Society 26: 1043-1047.

 Review Article
Review Article