ABSTRACT
Background and Objective: Over the years, the use of herbal products globally either as food supplement or curative medicine has witnessed a phenomenal growth. This is partly related to the fact that, herbal products are cheaper than the synthetic drugs and the erroneous impression by the general populace that, herbal products are natural and thus less harmful to the body. Therefore, the objective of this study was to carry out biochemical study on the effects of Ruzu Herbal Bitters formulation on wistar albino rats.
Materials and Methods: Twenty (20) adult albino rats were used for this research. Ruzu Herbal Bitters (treated groups, n=5) was administered to each rat at 5, 10 and 15mg/kg body weight dosages. Administration of Ruzu Herbal Bitter formulation to the animals was done for 14 days after the 7 days acclimatization before sacrificing them through cervical dislocation. Blood was collected by cardiac puncture, using 5ml syringes and 23G needles into blood sample containers for biochemical analysis using the standard biochemical methods. Biochemical indices analyzed were creatinine, urea, albumin, total protein, transaminases (AST, ALT), alkaline phosphatase (ALP), total cholesterol and total bilirubin.
Results: The result of the study shows that, all the mean values of total protein and creatinine levels analyzed were significantly higher while albumin levels were lower when compared with the control. Higher concentrations (10 and 15mg/kg) of the serum levels of ALT, AST, ALP and total cholesterol were significantly higher when compared with the control. Whereas lower concentration (5mg/kg) of serum levels of urea and total bilirubin in the experimental animals were observed to be higher when compared with the controls. Statistically, there were no significant differences of the analyzed parameters at 95% confidence level (P < 0.05).
Conclusion: From this study, Ruzu Herbal Bitters may be relatively safe at lower therapeutic dosage but at higher therapeutic dosages and prolong administration may result in renal, hepatic and cardiac toxicity. Therefore, public health awareness on the safety and the impending health risk associated with the administration of the polyherbal products becomes very imperative.
Keywords: Ruzu Herbal Bitters; Toxicity; Renal; Hepatic; Cardiac; Biochemical Parameter; Albino Rats
Abbreviations: ALP: Alkaline Phosphatase; WHO: World Health Organization; AST: Aspartate Aminotransferase; ALT: Alanine Aminotransferase; CREA: Plasma Creatinine; BUN: Blood Urea Nitrogen; TP: Total Serum Protein; TC: Total Serum cholesterol; SEM: Standard Error of Mean; ANOVA: Analysis of Variance; SPSS: Statistical Analysis Program for Social Sciences
Introduction
The development and use of traditional herbal medicine have come a long way and it corresponds to the Stone Age. In Africa, the practice of traditional healing and magic is much older than some of the other traditional medical sciences and seems to be more prevalent than conventional medicine [1]. World Health Organization (WHO) has defined Herbal medicine as an authorized medicinal herb or herbal materials or herbal preparations and finished herbal products which contain whole plants, parts of plants, or other plant materials such as: barks, leaves, flowers, berries and roots, and/or their extracts as active ingredients intended for human therapeutic use and sometimes animals [2]. It has become a common traditional practice in some cases, that a combination of plants or their extracts is used in the treatment of certain ailments with the believe by the herbalists that the individual plants contain different therapeutic agents in which when combined together will give a better therapeutic efficacy for a particular disease or multiple diseases than that of a single plant.
Therefore, most herbal preparations work in synergy, hence the emergence of polyherbal mixtures. Ruzu herbal bitters (Figure 1) are polyherbal mixtures, produced by Ruzu Natural Health Product and Services, Nigeria. The polyherbal mixture is made up of three different plants: 20% Uvaria chamae (bush banana), 40% Citrullus colocynthis (bitter apple) and 40% Curculigo pilosa (squirrel groundnut). Ruzu herbal bitters are commercially available and the manufacturers concluded that the product has the following medicinal functions amongst others, as indicated in the leaflet of the product: management/treatment of diabetes, typhoid and malaria, high blood pressure, waist and back pains, fibroid, infertility (male and female), gonorrhea/staphylococcus, syphilis, pile, obesity/ stomach troubles, detoxifies the kidney and tones the liver [3]. Based on the medicinal claims of the manufacturer of Ruzu herbal bitters with paucity scientific proofs, there was need to carry out scientific investigations to ascertain its effects on wistar albino rats by assessing biochemical indices.
Materials and Methods
Materials and Chemicals
Syringes and needles, hand Gloves, incubator, glucometer, Aucku check active strip micropipette, stop watch, oven, centrifuge Model 800, cotton wool, HPLC, and GCMS. The chemicals included 10% Chloroform, xylene, hemotoxylin and eosin stains.
Collection of Herbal Formulation Samples
Ruzu Herbal Bitters a Nigerian Herbal formulation was purchased from a registered pharmaceutical shop (Cynflac Pharmacy Yenagoa, Bayelsa State). The product is a combination of several medicinal plants. As an inclusion criterion, the product was ascertained to have been registered with the National Agency for Food, Drug Administration and Control (NAFDAC number: A7- 1102L). The manufactured and expiry dates of the product were inspected and all were confirmed to be within the acceptable time frame. The Manufacturer’s seal, inspected to ascertain the authenticity of the product was intact in the bottles of the syrup purchased for the analysis and was taken to the Research Laboratory, Department of Pharmacology, Faculty of Basic Medical Sciences, College of Health Sciences, Niger Delta University, Wilberforce Island, Bayelsa State and was stored under room temperature prior to the experiment.
Experimental Animals
Animal Handling: Twenty (20) adult albino rats used for this study were purchased from the animal house of the Faculty of Basic Medical Sciences, College of Health Sciences, Niger Delta University, Wilberforce Island, Bayelsa State. The animals were kept in standard plastic rat cages in the research laboratory of the Department of Pharmacology, Faculty of Basic Medical Sciences, College of Health Sciences, Niger Delta University, Wilberforce Island, Bayelsa State. The animals were allowed to acclimatize for 7 days under standard laboratory conditions with free access to commercial grower’s mash (Delta Feeds), water ad libitum and 12h/ 12h light/darkness cycle and fresh air prior to the inception of this study. The animal experiment was conducted in accordance with internationally accepted practice for laboratory animals and approved by the Animal Ethics Committee of the Faculty of Basic Medical Sciences, College of Health Sciences, Niger Delta University, Wilberforce Island, Bayelsa State.
Administration of Poly-Herbal Formulation: The herbal formulation was administered using 5ml syringe, the corresponding dose was given to each rat based at 5, 10 and 15 mg/kg body weight dose of the herbal formulation was selected.
Experimental Design
After the period of acclimatization, the animals were randomly divided into experimental and control groups. The albino rats were grouped and herbal formulation administered as follows;
• Group 1 (n = 5) Control: Albino rats received 2ml of distilled water daily within the period of the study before sacrificing.
• Group 2 (n = 5) Albino rats were treated with 5 mg/kg body weight of Ruzu Herbal Bitters
• Group 3 (n = 5) Albino rats were treated with 10 mg/kg body weight of Ruzu Herbal Bitters
• Group 4 (n = 5) Albino rats were treated with 15 mg/kg body weight of Ruzu Herbal Bitters.
Blood Sample Collection: The animals were observed in their cages for clinical symptoms daily and at the end of the 14 days treatment, the rats were sacrificed under chloroform anesthesia and blood was collected by cardiac puncture, using 5ml syringes and 23G needles into blood sample containers. The blood was allowed to stand for 2 hours to coagulate and was centrifuged for 10 minutes at 2000 rpm and the supernatant (Serum) carefully collected for biochemical analysis.
Biochemical Analysis: Serum levels of aspartate aminotransferase (AST) and alanine aminotransferase (ALT) were determined following the principle described by [4] while the alkaline phosphates (ALP) were carried out according to the method described by [5] to assess liver function. Renal function was assessed by measuring plasma creatinine (CREA) levels and blood urea nitrogen (BUN) was assayed following the method of [6,7]. In order to assess the synthetic function of the liver, total serum protein (TP), Total bilirubin and albumin (ALB) concentrations were determined according to the principles based on the Biuret reaction [8] and bromocresol green reaction [9] respectively. Total Serum cholesterol (TC) concentrations were estimated following the method described by [10].
Statistical Analysis of Data: All data were expressed as Mean ± Standard Error of Mean (SEM). Significant differences among the groups were determined by One-Way Analysis of Variance (ANOVA) using the statistical analysis program for social sciences (SPSS 17.0).
Results
Biochemical Parameters
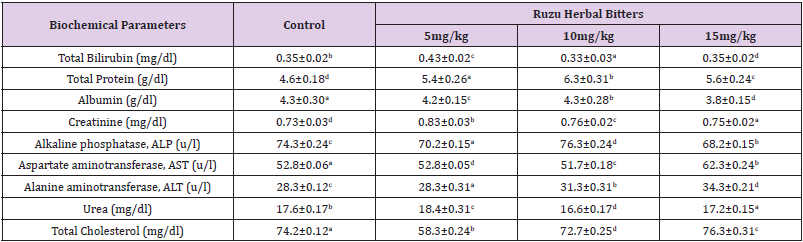
The Body Weight of wistar albino rats administered with Ruzu herbal bitters formulation is shown in Table 1. Table 2 shows the effects of Ruzu herbal Bitters formulation on biochemical indices of wistar albino rats at 5, 10 and 15mg/kg doses. The renal, hepatic, cardiac and lipid profile indices analyzed were albumin, total protein, urea, creatinine, Alkaline Phosphatase (ALP), Alanine aminotransferase (ALT), total bilirubin, Aspartate aminotransferase (AST) and total cholesterol and were compared with the control groups. All mean values of total protein and creatinine levels analyzed were significantly higher while albumin levels were lower when compared with the control. Higher concentrations (10 and 15mg/kg) of ALT, AST, ALP and total cholesterol were significantly higher when compared with the control. Whereas lower (5mg/kg) concentration of urea and total bilirubin levels in the experimental animals were observed to be higher when compared with the controls. Statistically, there were no significant differences of the analyzed parameters at 95% confidence level (P < 0.05).
Table 1: Body weight of wistar albino rats administered with Ruzu Herbal Bitters.
Note: (Mean ± SEM, n = 5)
Table 2: Effects of Ruzu Herbal Bitters on Biochemical indices of wistar albino rats.
Note: Data are Means ± SEM; n=5. Means of the same superscript alphabets in the same column shows no significant difference at 95% confidence levels (p<0.05).
Discussion
Herbal medicines are now receiving greater attention as an alternative to clinical therapy leading to increase in their demands [11]. In the rural communities of developing countries, the exclusive use of herbal drugs to treat various diseases is still very common and is prepared most often and dispensed by herbalists without formal training. Experimental screening method is therefore important in order to establish the active components present, ascertain the efficacy and safety of the herbal products [12]. The evaluation of the safety of these poly-herbal products is presently carried out in animals. In risk assessments, there are better correlations between rats and humans, whereas it is less predictive when mice are used for experimental [13]. Therefore, the aim of this study was to assess the effects of Ruzu Herbal Bitters formulation on wistar albino rats by carrying out biochemical parameters as marker enzymes. Study of the renal profile such as total protein, urea, creatinine and albumin levels give useful information about the poly-herbal formulation-induced renal toxicities. Animals which received herbal formulation in 5, 10 and 15mg/kg body weight doses did not show any significant changes in albumin levels. Decreased serum levels of albumin occur when there is impairment in the synthesizing function of the liver [14] therefore; the significant decrease in serum levels of albumin observed in this study may be an indication of liver impairment. However, animals at 5, 10 and 15mg/kg body weight doses showed highly significant increase in creatinine and total protein levels when compared to the control group. Creatinine levels are used as marker of kidney function.
Previous studies have shown that an apparently minor increase in serum creatinine can reflect a marked decrease in glomerular filtration rate [15]. Thus, elevated serum levels of creatinine may indicate kidney injury, with resultant reduced glomerular filtration. The results are synonymous with the findings of [16]. Urea is formed in the liver, representing the principal waste product of protein catabolism and is excreted by the kidney. In this study, urea levels at 10 and 15mg/kg doses shows significant decrease. However, the minimal increase in serum levels of urea at 5mg/kg dosage observed in the poly-herbal formulation group may be due to nephrotoxic effect of the herbal formulation, leading to reduced renal function. Liver damage tempted by herbal formulations or synthetic drugs may consist of hepatocellular necrosis, cholestasis, or a mixture of biochemical and histopathological patterns [17]. The estimation of AST, ALT and ALP is suitable in the early diagnosis of viral or toxic hepatitis and thus patients exposed to hepatotoxic drugs [18]. Generally, perturbation of parenchymal cells of the liver by xenobiotics or drugs results in elevation of both transaminases (AST and ALT) in the blood [19]. AST has both mitochondrial and cytoplasmic origin and any elevation could be taken as a first sign of cell damage that leads to the appearance of these enzymes in the serum [20]. Hence animals were tested for ALP, ALT, AST and total bilirubin levels to check for hepatic and cardiac toxicity. Therefore the increases observed in AST (15mg/kg) and ALT (10 and 15mg/ kg) activities in this study suggest that the administration of Ruzu herbal bitters did interfere with the integrity of the parenchymal cell. However the increase was only significant in the high dosage groups. The findings of this study were in agreement with the works of [21].
One of the major enzymes involved in hepatobiliary evaluation is alanine amino phosphatase (ALP). ALP levels above normal are mainly associated with blocked bile duct although this enzyme is also concentrated in the kidney and bone. The liver, via bile excretes ALP, whenever liver function is compromised the excretion of bile by the hepatocytes diminishes and this results in the increased levels in the serum ALP [22]. There was no significant increase in the ALP values at 5 and 15mg/kg body weight dosages, suggesting that Ruzu herbal bitters at the above doses did not obstruct bile excretion nor caused congestion and therefore Ruzu herbal bitters has no tendency to cause cholestasis. However, a minimal increase was observed at 10mg/kg dosage for ALP and 5 mg/kg dosage for total bilirubin when compared with the control groups. The non-significant and minimal increase observed in ALP and total bilirubin levels in this study is an indication that Ruzu herbal bitters did not impair the capacity of the liver to excrete bilirubin. Liver disease is characterized by an elevation in the serum levels of alkaline phosphatase (ALP), while hyperbilirubinaemia is seen in conditions causing hepatic liver diseases that impair the excretion of bilirubin [23]. This result is in accord with the works of [24,25]. The minimal increase in 15mg/kg dose observed in serum total cholesterol in the animal group may be due to the effect of the herbal formulation. The increase in the serum levels of total cholesterol may be attributed to the toxic effect of the formulation, leading to hepatobiliary disorders and impaired cholesterol metabolism. However the increase was only significant in the high dose group. These findings were in agreement with the works of [26].
Conclusion
In conclusion, this study has provided an insight and data on the Nigerian Poly-herbal formulation (Ruzu Herbal Bitters) which may be relatively safe at lower therapeutic dosage. However, at higher therapeutic dosages and prolong administration may result in renal, hepatic, cardiac and hepatobiliary disorders. Therefore, the creation of public health awareness becomes imperative on the safety and the impending health risk associated with the administration of the poly-herbal products.
References
- Ezekwesili Ofili JO, Okaka ANC (2019) Herbal Medicines in African Traditional Medicine. In: Ezekwesili Ofili JO, Okaka ANC (Eds.)., Intechopen Book, pp. 191-204.
- Falodun A, Imieje V (2013) Herbal Medicine in Nigeria: Holistic Overview. Nigerian Journal of Science and Environment 12(1): 1-13.
- Kale OE, Akinpelu OB, Bakare AA, Yusuf FO, Gomba R, et al. (2018) Five traditional Nigerian polyherbal remedics protect against high fructose fed, Streptozotocin-inuced type 2 diabetes in male Wistar rats MBC Complement Altern Med 18: 160.
- Reitman S, Frankel SA (1970) Colorimetric method for the determination of serum glutamate-oxaloacetate and pyruvate transaminases. Am J Clin Pathol 28(1): 56-63.
- Roy AV (1970) Rapid method for determining alkaline phosphatase activity in serum with thymolphthalein monophosphate. Clin Chem 16(5): 431-436.
- Fossati P, Prencipe L, Bert G (1980) Use of 3,5-dichloro- 2-hydroxybenzenesulfonic acid/4-aminophenazone chromogenic system in indirect enzymatic assay of uric acid in serum and urine. Clin Chem 26(2): 227-231.
- Skeggs LT (1957) An automatic method for colorimetric analysis. American J Clin. Pathol 28(3): 311-322.
- Gornall AG, Bardawill CJ, David MM (1949) Determination of serum proteins by means of the Biuret reaction. J Biol Chem 177(2): 751-766.
- Doumas BT, Watson WA, Biggs HG (1971) Albumin standards and the measurement of serum albumin with bromocresol green reaction. Clin Chem 31(1): 87-96.
- Trinder P (1969) Enzymatic Calorimetric determination of triglyceride using GPO-PAP method. Annals Clin Biochem 6: 24-27.
- Mythilypriya R, Shanthi P, Sachdanandam P (2007) Oral acute and subacute toxicity studies with Kalpaamruthaa, a modified indigenous preparation, on rats. J Health Sci 53(4): 351-358.
- Sushruta K, Satyanarayana S, Srinivas N, Sekhar RJ (2006) Evaluation of the blood–glucose reducing effects of aqueous extracts of the selected Umbellifereous fruits used in culinary practice. Trop J Pharmaceutical Res 5(2): 613-617.
- Olson H, Betton G, Robinson D, Thomas K, Monro A, et al. ( 2000) Concordance of the toxicity of Pharmaceuticals in humans and animals. Reg Tox and Pharmacol 32(1): 56-67.
- Omotosho IO, Olusanya T, Onyeaghala AO (2018) Biochemical Studies on Toxicity of Yoyo Bitters (A Polyherbal Preparation) on Liver and Kidney Functions of Wistar Rats Arch. Bas App Med 6: 177-182.
- Dhanvijay P, Misra AK, Varma SK (2013) Diclofenac-induced acute renal failure in a decompensated elderly patient. J Pharmacol Pharmacother 4(2): 155-157.
- David OC, Vincent ON (2020) Effect of Ruzu Herbal Bitters on the kidney Function and Hematological Parameters of Alloxan-Induced Diabetic Rats. International Journal of Scientific & Engineering Research 11(5): 63-86.
- Bonadonna G (1988) Chemotherapy Induced Complications. In: Bonadonna, G, Robustelli Della Cuna G (Eds.)., Handbook of Medical Oncology (3rd)., Masson, Milano, Parigi, Barecellona and Mexico, pp. 963-975.
- Zimmerman HJ (1984) Function and Integrity of the Liver. In: Henry JB (Edt.)., Clinical Diagnosis and Management by Laboratory Methods (17th)., WB Saunders, Philadelphia, pp. 217-250.
- Wolf PL, Williams D, Tsudaka T, Acosta L (1972) Methods and techniques in clinical chemistry. In: Wolf PL, Williams D, Tsudaka T, Acosta L (Eds.)., John Wiley and Sons, USA.
- Mdhluli M (2003) Toxicological and antifertility investigations of oleanolic acid in male vervet monkeys (Chlorocebus aethiops). Discipline of Physiological Sciences, University of the Western Cape, Cape Town, South Africa.
- Igweze ZN, Ezejiofor AN, Orisakwe OE, Obianime A (2019) Hepatic and heamatological effects of smart herbal purifier: a Nigerian poly herbal remedy in male sprague-dawley rats. Medical Research Archives 7(1): 1-13
- Rajesh MG, Latha MS (2004) Preliminary evaluation of the antihepatotoxic activity of Kamilari, a polyherbal formulation. J Ethnopharmacol 91(1): 99-104.
- Nsirim N (1999) Clinical Biochemistry for Students of Pathology (1st Edn)., In: Nsirim N (Edt.)., Longman Nigeria Plc. Lagos Nigeria, pp. 42-126.
- Omotosho IO, Olusanya T, Onyeaghala AO (2018) Biochemical Studies on Toxicity of Yoyo Bitters (A Polyherbal Preparation) on Liver and Kidney Functions of Wistar Rats Arch. Bas App Med 6: 177-182.
- Khan RA, Aslam M, Ahmed S (2016) Evaluation of Toxicological Profile of a Polyherbal Formulation Pharmacology & Pharmacy 7(1): 56-63.
- Odangowei IO, Timipa RO, Chiemeziem ON, Eruom EC, Eladebi BA, et al. (2020) Toxicity Studies on the Effects of Non-Steroidal Anti-Inflammatory Drugs in Wistar Albino Rats. Elixir Pharmacy 149: 55010-55014.

 Research Article
Research Article