ABSTRACT
Purpose: To explore the clinical utility of a free-breathing (FB) CMR (FCMR) imaging protocol at 3.0T.
Methods: 148 selected patients with suspected heart diseases were prospectively enrolled. These patients were divided into two groups, (1) group 1, 111 patients with good BH; (2) group 2, 37 patients with bad BH. Patients underwent both CCMR and FCMR protocols on a 3T scanner. The FCMR imaging was performed at the end of the CCMR protocol imaging. Acquisition time and Image quality (IQ) were compared between the two protocols. The consistency of the two methods in diagnosis, left ventricular function (LVF) measurement and scar presence/absence was analyzed.
Results: The acquisition time of the FCMR was significantly shorter than that of CCMR [(10.8±0.6) min vs. (35.5±2.9) min, P<0.001]. There was no difference of IQ between CCMR and FCMR in group 1: 4 (3-5) vs. 4 (3-5), P= 0.623. The IQ of CCMR was significantly lower than that of FCMR in group 2: [2 (2-3) vs.4 (3-5), P <0.001]. In group 1, the consistency was high for the LGE presence/absence (Kappa, 0.87), LVF (ICC, 0.94-0.99) and diagnosis (Kappa, 0.90). In group 2, the consistency was low for the LGE presence (Kappa, 0.32), LVF (ICC, 0.57-0.87) and diagnosis (Kappa, 0.21), the diagnosis rate of FCMR was significantly higher than that of CCMR [94.6% (35/37) vs. 29.7% (11/37), P <0.001].
Conclusions: FCMR could obtain robust and high-quality images in 10 minutes, even in patients with respiratory depression, making CMR imaging more widely available.
Keywords: Compressed Sensing; Cine; Motion-Corrected; Late Gadolinium Enhancement
Abbreviations: BH: Breath-Holding; CCMR: Conventional Cardiac Magnetic Resonance; FB: Free-Breathing; FCMR: Free-Breathing Cardiac Magnetic Resonance; LGE: Late Gadolinium Enhancement; CS: Compressed Sensing; IQ: Image Quality; LVF: Left Ventricular Function; ICC: Intra Class Correlation Coefficient; MOCO: Motion Corrected; FB: Free-Breathing; BSSFP: Balanced Steady-State Free Precession; FLASH: Fast Low-Angle Shot; PSIR: Phase-Sensitive Inversion Recovery; LAX: Long-Axis; SAX: Short-Axis; LV: Left Ventricle; Gd-DTPA: Gadolinium Diethylenetriamine Pentaacetic Acid; EF: Ejection Fraction; EDV and ESV: End-Diastolic And End-Systolic Volumes; SV: Stroke Volume; LVEDM: LV End-Diastolic Mass; ECG: Echocardiography
Introduction
Cardiovascular magnetic resonance (CMR) imaging has become an essential tool for the non-invasive examination of the heart. It has been used for the diagnosis, risk stratification, and prognosis of cardiac diseases [1,2]. Cine and late gadolinium enhancement (LGE) imaging are routinely included in the conventional cardiovascular magnetic resonance (CCMR) protocols in our center. Data acquisitions are typically performed with breath-holding (BH). While they work well in patients that are capable of holding their breath during image acquisition, such CCMR protocols remain challenging in patients with compromised BH capacities. In addition, the relatively long imaging time hinders the efficiency and throughput at a busy medical center like ours where there is a need to scan over 30 cardiac patients per MRI system per day. Real-time compressed sensing (CS) cine has been proved to be able to obtain high-quality images for evaluating cardiac function [3- 7]. Motion corrected (MOCO) single-shot LGE imaging techniques can also produce high-quality images without BH to detect fibrotic myocardial scars [8-9]. The novelty in this work is that both methods (CS cine and MOCO-LGE) are in corporate for a comprehensive FB CMR study. The feasibility and potential clinical utility of the proposed protocol were evaluated in patients that were unable to hold their breath during CMR imaging and in patients who could hold breath well.
Materials and Methods
Subject Enrollment
After the institutional review board approval was granted, adult patients scheduled for CCMR imaging were prospectively recruited for this study from February 1, 2017 to June 30, 2019. The inclusion criteria were as follows: in-patient at our hospital scheduled for contrast-enhanced CMR examination, a glomerular filtration rate of ³30 mL/min per 1.7m2, and no contraindications for CMR imaging. All patients who received FCMR protocols signed informed consent. A total of 148 patients were selected to complete both CCMR and FCMR protocols examination. These patients were divided into two groups,
a) Group 1, 111 patients with good BH;
b) Group 2, 37 patients with bad BH. The average age of these patients was 59 ± 16 years, with a male/female ratio of 90/58.
The CMR Imaging Protocol
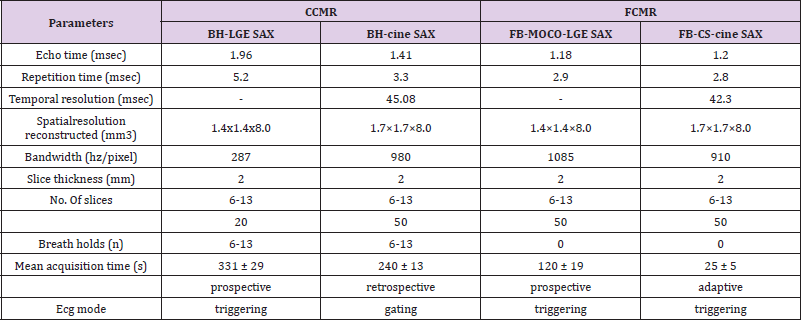
CCMR and FCMR scans were performed on a 3 Tesla (T) clinical magnetic resonance imaging (MRI) scanner (MAGNETOM Skyra, Siemens Healthcare, Erlangen, Germany). The system was equipped with an 18-element body array coil and a 32-element spine array coil. Key sequences for the CCMR included:
a) BH-cine imaging with segmented, balanced steady-state free precession (BSSFP) readout;
b) BH-LGE sequence for viability imaging under breath-hold using segmented, fast low-angle shot (FLASH) readout and phase-sensitive inversion recovery (PSIR) reconstruction.
The primary FCMR protocols included:
a) Single-shot FB-CS-cine imaging with BSSFP readout, featuring a two-dimensional sparse data sampling and iterative reconstruction (SSIR).
b) FB-MOCO-LGE employs non-rigid motion-correction and averaging of multiple single-shot SSFP images with PSIR reconstruction.
c) The BH-cine, FB-CS-cine, BH-LGE, and FB-MOCO-LGE protocols comprised separate 2-, 3-, and 4-chamber long-axis (LAX) acquisitions, and a short-axis (SAX) stack covering the entire left ventricle (LV). All scans were started from running BH CCMR protocols, when the CCMR protocols were successfully finished, FCMR protocols were began. Intravenous gadolinium diethylenetriamine pentaacetic acid (Gd-DTPA) contrast agent was administered at a dose of 0.2 ml/kg of body weight. For all exams, the contrast agent was administered to each patient in one injection.
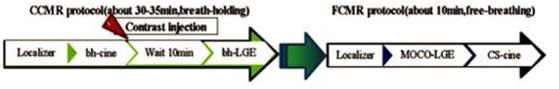
Figure 1: The workflow for the breath-holding (BH) conventional cardiac magnetic resonance imaging (CCMR) protocol and free-breathing cardiac resonance (FCMR) protocol. Abbreviations: HASTE, Half-Fourier-Acquired Single-shot Turbo spin Echo; MOCO, motion-corrected; LGE, late gadolinium enhancement; CS, compressed sensing.
The CMR protocol workflows is illustrated in Figure 1. Detailed information regarding the sequence parameters of both protocols is shown in Table 1. Both protocols were conducted using semiautomated cardiac day optimizing throughput (DOT) engine software including Auto Align feature to automatically prescribe the 2-, 3-, and 4-chamber views as well as the short axis stack [4,10]. Scan parameters like trigger delay were automatically adapted to patient physiology such as patient heart rate. The scanning time of every sequence and the total time of every protocol was write down, every two sequence intervals time was not included in the total time. Acquisition time, IQ were compared between the two protocols. The consistency of the two methods in diagnosis, LVF measurement and scar presence/absence was analyzed.
Image Analysis
Image Quality: All CMR images were transferred to a workstation (cmr42, Version 5.12.1, Circle Cardiovascular Imaging, Calgary, Canada) for evaluations. For those 148 patients where both FCMR and CCMR images were acquired, FCMR and CCMR were randomly assigned to two senior doctors, Reader 1 and Reader 2, both with more than 5 years of experience in CMR reading, for double-blind evaluation respectively. Image quality scores were evaluated based on a five-point Likert score (5 = excellent, 4 = good, 3 = the presence of artifacts but acceptable, 2 = poor, and 1 = non diagnostic) [6-8].
Imaging Analyses: LVF measurements were assessed with cmr42 software. Endocardial and epicardial contours were automatically delineated on the short-axis cine images using the cmr42 software and manually adjusted as needed [11]. Papillary muscles and trabeculations of the left ventricle (LV) were included in the ventricular cavity volume measurements. Ejection fraction (EF), end-diastolic and end-systolic volumes (EDV and ESV), stroke volume (SV), and LV end-diastolic mass (LVEDM) measurements were accessed from the cine images acquired in both protocols, and the consistency of measurements between both protocols was analyzed by using linear regression analyses and Bland-Altman plots. If LGE involves the sub endocardial distribution of coronary artery, it can be identified as ischemic LGE type; otherwise, it can be identified as non-ischemic LGE [12,13].
Statistical Analyses: Statistical analyses were performed using dedicated SPSS (version 20.0, SPSS Inc., Chicago, USA) and MedCalc10.0 (Med Calc Software, Ostend, Belgium) software. Continuous data were checked for normality using the Shapiro-Wilk test and presented as the mean ± standard deviation or median (interquartile range, Q1–Q3), and compared using the T test or Mann-Whitney test. The FB-CS-cine and BH-cine imaging were compared using Bland-Altman. Intra class Correlation Coefficient (ICC) were used to evaluate the consistency of quantitative data. The Kappa statistics were used to evaluate consistency of qualitative variable. Agreement strengths for kappa values were classified as follows: <0.1, poor; 0.1 to 0.20, slight; 0.21 to 0.40, fair; 0.41 to 0.60, moderate; 0.61 to 0.80, substantial; and 0.81 to 1.00, almost perfect. P <0.05 was considered statistically significant.
Results
Image Acquisition Times
The total time of the FB-CS-cine SAX (25±5)s, FB-CS-cine LAX (8±2)s, FB-MOCO-LGE SAX (120±19)s, FB-MOCO-LGE LAX (37±6) s was significantly shorter than that of the BH-cine SAX (340±30) s, BH-cine LAX (75±16)s, BH-LGE SAX (331±29)s, BH-LGE LAX (100±9)s respectively, (all P-values < 0.001).The total time of the FCMR was significantly shorter than that of the CCMR protocol [(10.8 ± 0.6) minutes vs. (35.5 ± 2.9) minutes, P<0.001].
Imaging Analyses in the Group 1
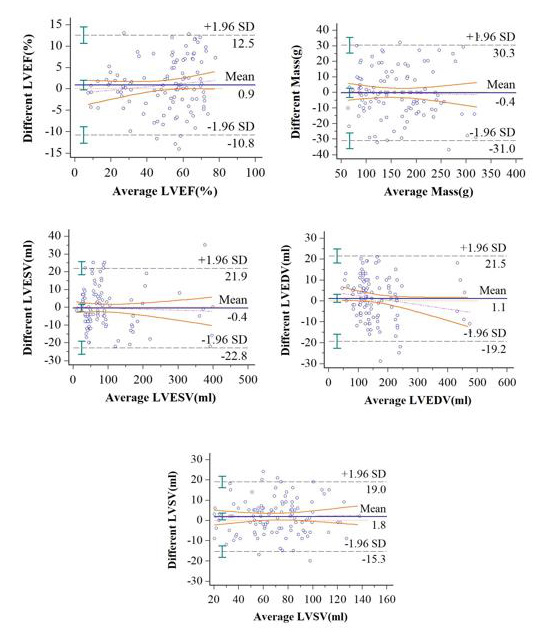
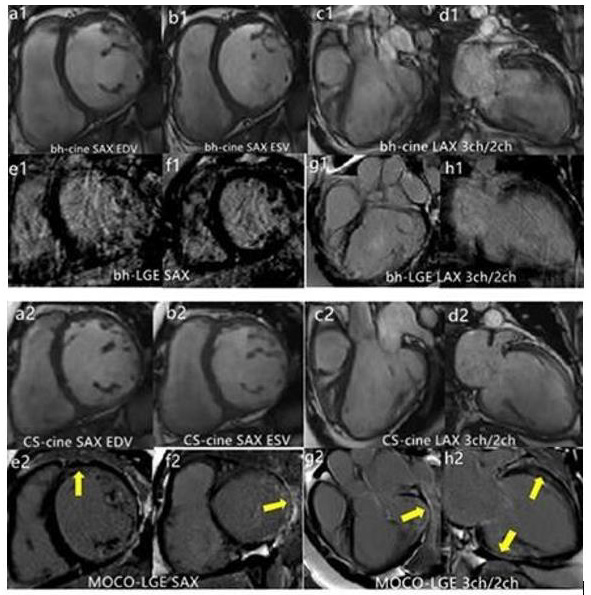
IQ was significantly better in the BH-cine images compared to the FB-CS-cine images [5 (4-5) vs. 4 (3-4), P<0.001]. However, IQ was significantly better with FB-MOCO-LGE compared to BH-LGE [5 (4-5) vs. 3 (3-4), P <0.001]. When the cine and LGE scores were averaged, they were 4 (3-5) for the CCMR protocols, and 4 (3-5) for the FCMR protocols, no differences were seen in IQ between the CCMR and FCMR protocols (P = 0.623). Figure 2 showed images from patients with good BH cooperation acquired with both excellent CCMR and FCMR images. The comparison of LVF parameters between BH-cine and FB-CS-cine is as follows: LVEDV (ml) [ 161.7 ± 88.5 vs. 160.5 ± 90.4, P = 0.828], LVESV(ml) [ 90.8 (74.5 ± 107.1) vs. 91.6 (75.2 ± 107.9), P = 0.962], LVSV(ml) [ 70.8 ± 25.9 vs. 69.8 ± 25.7, P = 0.920], LVEDM(g) [ 161.6 ± 64.8 vs. 162.0 ± 65.3, P = 0.939], LVEF(%) [50.2 ± 18.6 vs. 49.3 ± 18.1, P = 0.685]. There was high consistency (ICC, 0.94-0.98) between BH-cine and FB-CS-cine for LVF evaluations. The Bland-Altman statistical method was used for intergroup bias analysis Figure 3.
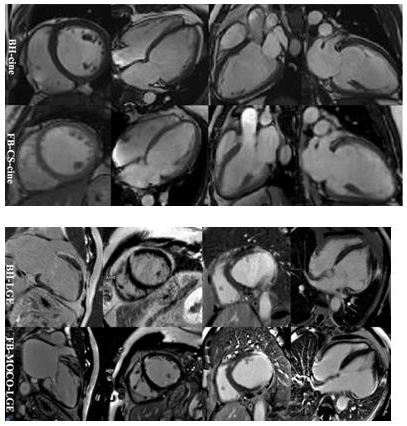
Figure 2: Both conventional cardiovascular magnetic resonance (CCMR) late gadolinium enhancement (BH-LGE) images including breathing hold (BH) cine and BH and late gadolinium enhancement (LGE) sequences corresponding free-breathing cardiac magnetic resonance (FCMR) protocol including free-breathing (FB) cine and motion-corrected (MOCO) LGE sequences were excellent in patients with good breathing hold (BH).
Figure 3: Bland-Altman plots for left ventricle (LV) functional parameters in breath-hold cine MRI and free-breathing compressed-sensing cine MRI derived a cohort of 111 patients. A, LV ejection fraction (LVEF); B, LV end-diastolic volume (LVEDV); C, LV end-systolic volume (LVESV); D,) LV stroke volume (LVSV); and E, LV end-diastolic myocardial mass (LVEDM).SD= standard deviation.
The mean differences in LVF measurements between BH-cine and FB-CS-cine were as follows: LVEDV, 1.1 ml (95 % CI: -24.7 to 22.5 ml); LVESV, -0.8 ml (95% CI: -23.7 to 22.1ml);LVSV,1.8ml(95% CI: -5.0 to 8.7 ml); LVED mass, -0.4 g (95% CI: -17.5 to 16.8 g); LVEF, 0.9% (95 %CI: -5.7 to 4. 0 %). There was high consistency (kappa, 0.89) between BH-LGE presence 55.9% (62/111) and FBMOCO- LGE 54.1 % (60/111). All the 111 patients with suspected heart disease were diagnosed by CCMR as following: 18 patients were diagnosed as myocardial infarction; 40 patients were dilated cardiomyopathy; 23 patients were hypertrophic cardiomyopathy; 6 patients were hypertensive cardiomyopathy; 5 patients were restrictive cardiomyopathy; 1 patients were right ventricular cardiomyopathy; 1 patients were myocardial amyloidosis; 2 patients were non-compaction of the ventricular myocardium; 4 patients were myocarditis; 1 patient was pericarditis; 5 patients were atrial or ventricular septal defect; 1 patient was myxoma; In addition, 65 patients had some of the above cardiomyopathy combined with valve insufficiency.
There were 6 patients without any heart disease. All the 111 patients with suspected heart disease were diagnosed by FCMR as following: Among the 5 cases of CCMR trial or ventricular septal defect, 2 cases was positive and 3 cases were negative by FCMR. Among 65 cases with valvular insufficiency by CCMR, 39 cases were positive and 26 cases were negative by CCMR. The two diagnostic methods were completely consistent in the remaining patients. The diagnostic consistency of the two protocols was 0.56 for atrial or ventricular septal defect, 0.55 for valvular insufficiency, and 0.86 for all patients. Figures 4 & 5 show some cases with bad BH cooperation got both non-diagnostic CCMR but diagnostic FCMR.
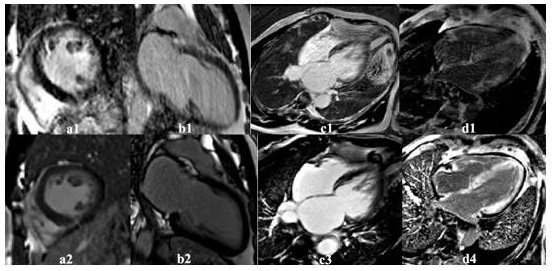
Figure 4: Images of a patient with an uncertain diagnosis on conventional cardiovascular magnetic resonance (CCMR) imaging (a1-h1). IQ of 2-chamber short axis (LAX) breath-holding (BH)-cine was excellent, however, 2 chamber and 3 chamber long axis (LAX) BH-cine images (c1, d1) and BH-late gadolinium enhancement (BH-LGE) images (e1-h1) were poor. On free-breathing cardiac magnetic resonance (FCMR) imaging, FB-CS-cine images (a2-d2) and motion-corrected (MOCO)-LGE images (e2-h2) were all excellent. LGE appeared at Left ventricular free wall on the MOCO-LGE images (e2-h2).
Figure 5: Images show a few cases with various cardiac diseases acquired with breath-holding (BH)-LGE and corresponding free-breathing motion-corrected (MOCO)-late gadolinium (LGE). Some patients with bad BH cooperation had non-diagnostic BH LGE images (a1-d1) but diagnostic FB-MOCO-LGE images (a2-d2).
Imaging Analyses in the Group 2
In group 2, of those 37 patients with BH limitation, IQ was significantly worse in the BH-cine images compared to the FB-CScine images [3 (2-4) vs. 4 (3-4), P <0.001]. IQ was also significantly worse with BH-LGE compared to FB-MOCO-LGE [2 (1-2) vs. 5 (4- 5), P <0.001]. When the cine and LGE scores were averaged, the IQ of CCMR was significantly lower than that of FCMR [2 (2-3) vs.4 (3-5), P <0.001]. The consistency was low for the LGE presence (Kappa, 0.32), LVF assessment (ICC, 0.57-0.87) and the diagnosis (Kappa, 0.047). There were 24 patients with limited BH capabilities had inconclusive results with the CCMR protocol, but definitive diagnoses were made with the FCMR protocol. The diagnosis rate of FCMR was significantly higher than that of CCMR [94.6% (35/37) vs. 29.7% (11/37), P <0.001].
Discussion
The purpose of the study was to explore the feasibility of clinical utility of a FCMR imaging protocol at 3.0T. The results showed that FCMR and CCMR protocols had comparable image quality ratings, left ventricular function assessment, myocardial scar detection and heart disease diagnosis when both protocols were successfully obtained. The total acquisition time of FCMR including FB-CS-cine and FB-MOCO-LGE was significantly shorter than that of the CCMR including BH-cine and BH-LGE. Furthermore, our results showed that the FCMR protocols could get robust images even in patients with limited BH capabilities. In our study, the total acquisition time of FCMR was significantly shorter than that of the CCMR protocol. The CCMR imaging protocol requires multiple breath-holds to provide diagnostic image quality [14,15]. Generally, each BH takes 8-15 seconds per slice, with an additional pause that lasts 10 seconds before the next breath-hold session. So, the CCMR protocol needs longer scanning time than the FCMR protocol. Such repeated and long BH requirements can be challenging for patients who cannot hold their breath for extended periods.
Also, to achieve sufficiently high spatial and/or temporal resolutions during CCMR imaging, segmented k-space data are acquired over multiple heartbeats. Such segmented acquisition is prone to motion artifacts that could lead to repeated scans in case of suboptimal breath-holding. In our clinical setting, a few of the patients were unable to complete the CCMR examinations due to impaired BH capacity. The FCMR protocol not only removes the BH barrier which is particularly important for scanning most vulnerable patients with compromised BH capability, it also improves the scan efficiency. In addition, single-shot readout effectively eliminates breathing motion artifacts in both FB-CS-cine and FB-MOCO-LGE images [14-16]. High quality images were acquired for cine with CS acceleration, the high image quality of the CS technique translated into high agreement for left ventricular function. Also, high quality images were acquired for LGE by combining non-rigid MOCO and averaging of multiple single-shot measurements.
For patients with good BH, they can well cooperate with breath holding, high agreement between the BH and FB MOCO technique was also achieved for LGE, with a non-significant difference of LGE presence or types. Our study found that there was no difference in LVF calculation and LGE detection between CCMR and FCMR images obtained from 111 patients without BH impairment, which was consistent with previous studies [3-5,7-9]. Moreover, the consistency of the two protocols for the diagnosis of myocardial infarction and cardiomyopathy was excellent, although the consistency of the two protocols for detecting the atrial or ventricular septal defect and valvular insufficiency was moderate. However, we observed that FB-CS-cine scans sometimes lead to a little of image blurring and low spatial resolution, and the IQ in FB CS cine was lower than that of BH cine. In addition, CS cine of the FCMR protocol was worse than the segment cine of the CCMR protocol in the recognition of valve insufficiency and atrial septal or ventricular septal defect. There were some reasons for these phenomena as following [17].
First, FB-CS-cine was susceptibility for fold over artifacts, therefore, the field of view must cover the entire anatomy, and thus, some penalty in spatial resolution may occur in relation to the patient’s anatomy. Second, in some scans, flow-related artifacts occurred in the phase-encoding direction during systole because the sparsity in the temporal domain may be limited in anatomic regions of very high flow. Lastly, the contrast between myocardium and blood pool in FB CS cine images was not as good as that of the BH cine images.
Overall, FCMR imaging leads to consistent images for diagnosis in all patients, regardless of whether they could hold their breath or not. In comparison, the IQ of CCMR depends on the BH capability of a patient during data acquisition. For patients with BH impairment, CCMR images suffer from severe motion artifacts, interfering the radiologist’ ability to interpret morphologic cardiac structures, cardiac function calculations, and LGE detection. FCMR has been shown to be an effective alternative to CCMR in this study, expanding the application range of CMR imaging. There were several limitations to this study. First, the current study assessed FCMR and CCMR scans in patients with various cardiac diseases, complicating the comparison of the two protocols. Secondly, no advanced MRI sequences, such as mapping, perfusion, and flow quantification were performed in the study since they are not part of the standard CMR protocols at our institution. Finally, the consistent of myocardial strain between BH-cine and FB-CS-cine, and scar volume between BH-LGE and FB-MOCO-LGE was not assessment. The encouraging results from this study warrants future study with a larger sample size to demonstrate the clinical utility of free-breathing.
Conclusion
In conclusion, we demonstrated that FCMR imaging could be used as an alternative technique in patients with BH impairment to obtain high-quality images. FCMR significantly shortens the time needed for CMR imaging and resulted in improved image quality. We believe that the FCMR protocol will allow the fast screen of cardiac diseases in clinical practice, with the potential to increase both the throughput and robustness of CMR.
Declarations
Ethics Approval and Consent to Participate
Approval was obtained from the First Affiliated Hospital of Zhengzhou University ethics committee. All subjects/ legal guardians gave written consent and assent as appropriate.
Consent to Publish
The local ethics committee approved the use of images and content from this study. Consent and assent were signed for all subjects prior to publication.
Availability of Data and Materials
The datasets generated for this study are available on reasonable request to the corresponding author.
Competing Interests
The authors declare that they have no competing interests.
Funding
Not applicable.
Authors’ Contribution
KYW (MD) conceived the study, performed the statistical analyses, and drafted the manuscript. Both WBZ (MD) and SML (MD) quantitatively measured cardiac function and infarction size, and analyzed image quality. MS (PhD) contributed to the sequence development and scanner implementation and helped to revise the manuscript. JA (MD) participated in the study design and scanner implementation and helped to revise the manuscript. JLC (MD) assisted in the interpretation of the results and helped to revise the manuscript. All authors have read and approved the final manuscript.
Acknowledgement
None.
References
- Pennell DJ (2010) Cardio vascular magnetic resonance. Circulation 121(5): 692-705.
- Miller CA, Pearce K, Jordan P, Rachel Argyle, David Clark, et al. (2012) Comparison of real-time three-dimensional echocardiography with cardiovascular magnetic resonance for left ventricular volumetric assessment in unselected patients. Eur Heart J Cardiovasc Imaging 13(2): 187-195.
- Lustig M, Donoho D, Pauly JM Sparse (2007) MRI: The application of compressed sensing for rapid MR imaging. MagnReson Med 58(6): 1182-1195.
- Kido T, Kido T, Nakamura M, Kouki Watanabe, Michaela Schmidt, et al. (2017) Assessment of Left Ventricular Function and Mass on Free-Breathing Compressed Sensing Real-Time Cine Imaging. Circ J 81(10): 1463-1468.
- Pednekar Amol S, WangHui, Flamm S, Scott Flamm, Benjamin Y Cheong, et al. (2018) Two-center clinical validation and quantitative assessment of respiratory triggered retrospectively cardiac gated balanced-SSFP cine cardiovascular magnetic resonance imaging in adults. J Cardiovasc Magn Reson 20(1): 44.
- Sudarski S, Henzler T, Haubenreisser H, Christina Dösch, Michael O Zenge, et al. (2017) Free-breathing Sparse Sampling Cine MR Imaging with Iterative Reconstruction for the Assessment of Left Ventricular Function and Mass at 3.0 Radiology 282(1): 74-83.
- Kocaoglu M, Pednekar AS, Wang H, Tarek Alsaied, Michael D Taylor, et al. (2020) Breath-hold and free-breathing quantitative assessment of biventricular volume and function using compressed SENSE: a clinical validation in children and young adults. J Cardiovasc MagnReson 22: 54.
- Piehler KM, Wong TC, Puntil KS, Karolina M Zareba, David M Harris, et al. (2013) Free-breathing, motion-corrected late gadolinium enhancement is robust and extends risk stratification to vulnerable patients. Circ Cardiovasc Imaging 6(3): 423-432.
- Captur G, Lobascio I, Ye Y, Veronica Culotta, Redha Boubertakh, et al. (2019) Motion-corrected free-breathing LGE delivers high quality imaging and reduces scan time by half: an independent validation Int J Cardiovasc Imaging 35(10): 1893-1901.
- XG L, Marie-Pierre J, Georgescu B, Edgar Mueller, Jens Guehring, et al. (2011) Automatic view planning for cardiac MRI acquisition. Med Image Comput Assist Interv 14: 479-486.
- Van Geuns RJ, Baks T, Gronenschild EH, Filippo Cademartiri, Pim J de Feyter, et al. (2006) Automatic quantitative left ventricularanalysis of cine MR images by using three-dimensional information for contour detection. Radiology 240(1): 215-221.
- Mahrholdt H, Wagner A, Judd RM, Udo Sechtem, Raymond J Kim (2005) Delayed enhancement cardiovascular magnetic resonance assessment of non-ischaemic cardiomyopathies. Eur Heart J 26(15): 1461-1474.
- Desroche LM, Milleron O, Safar B, Phalla Ou, Eric Garbarz, et al. (2020) Cardiovascular Magnetic Resonance May Avoid Unnecessary Coronary Angiography in Patients with Unexplained Left Ventricular Systolic Dysfunction: A Retrospective Diagnostic Pilot Study J Card Fail 26(12): 1067-1074.
- Yang AC, Kretzler M, Sudarski S, Vikas Gulani, Nicole Seiberlich (2016) Sparse Reconstruction Techniques in Magnetic Resonance Imaging: Methods, Applications, and Challenges to Clinical Adoption. Invest Radiol 51(6): 349-364.
- Usman M, Atkinson D, Odille F, Christoph Kolbitsch, Ghislain Vaillant, et al. (2013) Motion corrected compressed sensing for free-breathing dynamic cardiac MRI. MagnReson Med 70(2): 504-516.
- Lin ACW, Strugnell W, Riley R, Benjamin Schmitt, Michael Zenge, et al. (2017) Higher resolution cine imaging with compressed sensing for accelerated clinical left ventricular evaluation. J MagnReson Imaging 45(6): 1693-1699.
- Vincenti G, Monney P, Chaptinel J, Tobias Rutz, Simone Coppo, et al. (2014) Compressed sensing single-breath-hold CMR for fast quantification of LV function, volumes, and mass. JACC Cardiovasc Imaging 7(9): 882-892.

 Research Article
Research Article