Abstract
Shortly after the SARS-CoV-2 virus, which caused Corona Virus Disease 2019 (COVID-19) infection, spread all over the world, many countries started vaccine trials. One of these vaccines, the Pfizer/BioNTech Covid-19 (BNT162b2) vaccine, was granted Emergency Use Authorizations (EUAs) for the Advisory Committee on Immunization Practices (ACIP) by the Food and Drug Administration (FDA) in December 2020. It has been determined that these vaccines, which have entered medical applications, may require follow-up and treatment due to their simple and serious side effects. Acute myocardial infarction is a life-threatening, serious condition requiring early diagnosis and treatment, which can manifest itself with many clinical manifestations such as chest pain, arm, jaw, neck pain, shortness of breath, and syncope. In this case, we aimed to emphasize the serious side effects such as acute ST-elevation myocardial infarction in a patient who applied to the emergency department with chest pain after the Pfizer/ BioNTech Covid-19 vaccine, in the accompanied by of the literature.
Keywords: Myocardial İnfarction; Covid-19; Pfizer/Biontech Vaccine
Abbreviations: AMI: Acute Myocardial İnfarction; ECG: Electrocardiography; PCR: Polymerase Chain Reaction; RCA : Right Coronary Artery; PCI: Percutaneous Coronary İntervention; VAERS: Vaccine Adverse Event Reporting System; AEs: Suspected Adverse Events
Introduction
Corona-type viruses have been known for years. In the
2019/2020 period, a new type of SARS-Corona virus-2 (SARS-CoV-
2=COVID-19) started to spread all over the world. Several vaccines
have been approved against COVID-19. Each of these sought to
respond to the pandemic by providing individual protection against
COVID-19. One of these vaccines is the messenger ribonucleic acid
(mRNA) COVID-19 (Pfizer/BioNTech) vaccine that is based on new
gene-based technology. It was recommended that the vaccine be
administered in two doses, with a 21-day interval between them.
Based on current knowledge, COVID-19 mRNA vaccines have a high
efficacy rate of approximately 95%. Current research data indicate
that the odds of contracting COVID-19 infection are approximately
95% lower in fully vaccinated persons than in unvaccinated
persons. Its effectiveness in preventing severe COVID-19 disease
(for example, hospitalization) is about 85%. It is not yet known how
long the protection of this vaccine will last. The Pfizer/BioNTech
vaccine is approved for people 12 years of age and older. However,
some unexpected side effects may occur after vaccinations for
COVID-19 infection, the future of which is still unknown. After
one of the mRNA vaccines, local and general reactions can be seen
as a result of the interaction of the vaccine with the body. These
reactions are generally expected to disappear within 2 days after
vaccination [1].
Although rare cases of myocarditis and pericarditis have been
reported among cardiac side effects after the mRNA covid-19
vaccine, no significant heart attack was observed in vaccinated patients. It is thought that other medical conditions that develop
during or after vaccination may not always be related to the vaccine,
since large-scale vaccination is performed [2]. Diagnosing postvaccine
Acute Myocardial İnfarction (AMI) can be difficult. Because
muscle pain at the injection site may cause ischemic symptoms to
be overlooked and delayed admission. As far as we can search the
literature, we know of two cases of acute myocardial infarction in
the United States that started less than 24 hours after the first dose
of the COVID-19 vaccine [3]. In our case, we aimed to emphasize
acute myocardial infarction and other possible side effects that
developed within hours after the second dose of Pfizer/BioNTech
COVID-19 vaccine, in the light of the literature.
Case Report
A 39-year-old male patient said he had shoulder, arm, and chest
pain lasting more than three days after receiving the first dose of
the Pfizer/BioNTech COVID-19 vaccine. There was a relief after
taking paracetamol and nonsteroidal anti-inflammatory tablets
for his pain. He attributed all these pains to possible post-vaccine
reasons. After 21 days, the patient applied to the emergency service
with complaints of discomfort and tightness in the chest, which
started 1.5-2 hours after the second dose of vaccine, continued
for more than 10 minutes. Also, the patient could not describe
the nature of the pain exactly. He stated that the pain was not like
in the first dose, but was suppressive, restless, starting from the
scaphoid and spreading to the jaw and neck. The patient had both
vaccines done on the left arm. The patient did not have the fever
or respiratory distress at the time of admission and did not report
any drug-vaccine allergy. There was no cardiac history in his own
and first-degree relatives. In his examination; general condition
was good. Vital signs are blood pressure; arterial 130/70mmHg,
temperature: 36.5 °C, oxygen saturation parameter (SpO2): 99%,
heart rate: 70/minute. When the heart was listened to, there was no
additional sound and no murmur. Peripheral pulses were palpable.
Other system examinations were normal. Electrocardiography
(ECG), complete blood count, biochemistry, and cardiac biomarkers
were requested from the patient. Polymerase Chain Reaction (PCR)
was requested for COVID-19 in the nasopharyngeal aspirate in the
emergency department, the test result was negative.
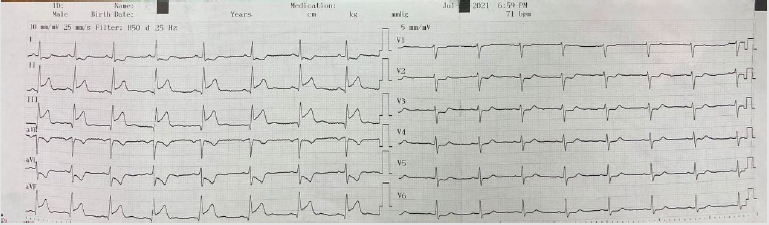
In the ECG taken in the fifth minute of the patient’s admission
to the emergency department; ST-segment elevation in inferior
leads (II, III, and aVF) and reciprocal ST-segment depression in I
and aVL were detected (Figure 1). The patient’s cardiac troponin
T value at admission; 8.83pg/dl (range: 0-14pg/ml), 5097pg/dl
at the 6th hour and 2689pg/dl at the 24th hour. ECG monitoring,
300 mg acetylsalicylic acid, 300 mg clopidogrel orally, 5000 IU
heparin intravenously were administered to the patient with the
diagnosis of acute inferior myocardial infarction. Emergency
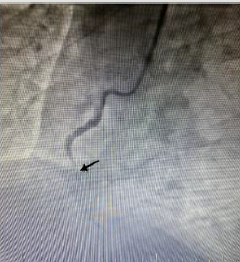
coronary angiography showed globular thrombus and occluded
proximal segment of the Right Coronary Artery (RCA) with
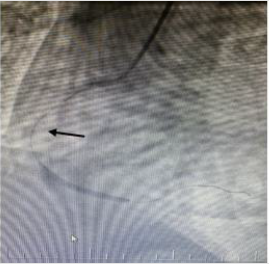
TIMI 0 flow (Figure 2). RCA emergency Percutaneous Coronary
İntervention (PCI) was performed. Dilated with balloon and stent
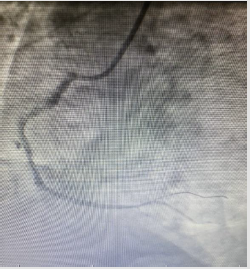
placed. After coronary angiography, a good outcome was detected
with the TIMI III flow (Figures 3 & 4). The patient did not have
chest pain after percutaneous coronary intervention. On the fifth
day of hospitalization, the patient had minimal hypokinesia of the
right inferior wall and was discharged with a cured left ventricular
ejection fraction of 60-65%.
Figure 1: ST elevation in lead II, III and AVF (blue arrow) and ST depression in I and AVL (red arrow).
Discussion
Although vaccines dedicated to COVID-19 disease have been
approved as safe, both the disease and the vaccine are relatively
new [4-6]. This requires that even very rare events be widely shared
and discussed with the medical community. We report a case of AMI
occurring within two hours of the second dose of the COVID-19
vaccine. The angiographic features of the case suggested acute
thrombotic events as the underlying mechanism. Although it is not
possible to establish a causal relationship between vaccination and
cardiac events, it is important to remember that the mechanisms
of thrombotic events in COVID-19 are still not fully understood [7].
Vaccines are considered the most effective drugs in public health.
Side effects may also occur due to some reactions after vaccination.
It is often difficult to say whether the reactions are caused by the
vaccine itself or by other factors causing AMI. Excipients used
in vaccines; it is used to improve stability, increase solubility
and improve absorption. However, these substances contribute
significantly to the development of IgE-mediated anaphylactic
reactions during vaccination [8].
Kounis syndrome is an allergic reaction to various substances,
including excipients, resulting in acute coronary syndrome [9].
This could be a possible explanation for the AMI clinic post-
COVID-19 vaccine. It may also occur through prothrombotic
immune thrombocytopenia, which is similar to heparin-induced
thrombocytopenia, leads to vaccine-induced thrombotic events
[10]. Although there is limited data on the risk of thrombotic events
with vaccines currently in use in the United States, there have been
recent reports of thrombotic events after COVID-19 vaccines in
Europe. No acute myocardial infarction was reported in the original
study of vaccines used in Covid-19. They also did not present a
significant difference in thrombotic events between the vaccinated
and placebo groups. However, cases have started to be presented
after these vaccines, rarely. Boivin et al. reported a 96-year-old
female case who had AMI one hour after the Moderna COVID-19
vaccine, and one case of AMI in a 63-year-old healthy man two
days after the AstraZeneca COVID-19 vaccine [11]. Maadarani
AstraZeneca presented a case of AMI after the COVID-19 vaccine
[12]. Tajstra on the other hand, reported an 86-year-old male
case of AMI after Pfizer/BioNTech Covid-19 vaccine [13]. Finally,
Jonathan presented two cases of AMI, one male, and one female,
after the Moderna vaccine [3]. Our case can be seen as the second
case in the literature after Pfizer/BioNTech Covid-19 vaccine.
The Vaccine Adverse Event Reporting System (VAERS) [14], the
National Vaccine Safety passive monitoring system, reported that
side effects occurred after receiving the second dose of 76% of
mRNA vaccines made so far. According to the Centers for Disease
Control and Prevention (CDC) case definitions, The median interval
from vaccination to symptom onset was 2 days (range = 0-40 days). Some of the symptoms were seen within 7 days of vaccination in
92% of patients. Acute clinical courses were generally mild [15].
About 78% of Suspected Adverse Events (AEs) were reported in
individuals younger than 60 years of age. In clinical trials of mRNA
vaccines, it has been noted that people younger than 60 years tend
to experience more reactogenic AEs than those aged 60 and over. In
general, younger individuals have more active immune responses
and may experience more AEs to vaccines. This is part of the
body’s natural response to build immunity to COVID-19 infection.
Approximately 66% of AEs have been reported in women.
Of the 7.5 million doses of Pfizer-BioNTech and Moderna mRNA
vaccine administered in total, 0.12% were reported as suspicious
and 0.005% as severe AEs. The most commonly reported serious
side effects are anaphylaxis and other serious allergic reactions.
Also asthma exacerbation, difficulty breathing, fast heart rate,
increase or decrease in blood pressure, chest discomfort and pain,
pericarditis or myocarditis, syncope, numbness of limb, weakness
or pain, vision changes, increase in liver enzymes, thyroid gland
dysfunction, muscle injury, joint pain, seizures, tinnitus, infections,
and blood clots. Most individuals who develop severe AE are
reported to have recovered. A higher incidence of heart attacks
and strokes was not observed in locally vaccinated persons. It’s
also important to note that heart attacks and strokes can occur
naturally, regardless of whether people are vaccinated or not. It is
argued that the benefits of COVID-19 vaccines continue to outweigh
the known risks in the pandemic [2].
Conclusion
As the COVID-19 vaccine campaign moves into a new phase worldwide and vaccines become available to the general public, there will be more data on safety and potential side effects. Further data on nationwide myocardial infarction or other thrombotic events before and after vaccination initiation should be carefully considered and interpreted before commenting on any possible causal relationship.
Authorship
The conception and design of the work; the acquisition, analysis, and interpretation of data for the work; drafting the work and revising it critically for important intellectual content; and final approval of the manuscript was done by Abuzer Coşkun, Assoc. Prof., Ahmet Barıs Ozkan, MD, and Burak Dermirci, MD.
Acknowledgement
None declared.
Funding
None declared.
Availability of Data and Materials
All data is available on request without restriction.
Conflict of Interest
None declared.
Ethical Approval
Informed consent and permission were obtained from the patient.
References
- (2019) Information Document: Against COVID-19 (Corona Virus Disease 2019) -with mRNA vaccines– (BioNTech/Pfizer's Comirnaty® and Moderna's COVID-19 Vaccine Moderna®): For vaccination
- (2021) HSA’s Safety Update 4; COVID-19 Vaccines. Health Sciences Authority (HSA), Singapore.
- Jonathan GS, Piotr SS, Deepak LB (2021) Acute Myocardial Infarction within 24 Hours after COVID-19 Vaccination. Am J Cardiol 156: 129-131.
- Polack FP, Thomas SJ, Kitchin N, Judith Absalon, Alejandra Gurtman, et al. (2020) Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N Engl J Med 383: 2603-26
- Jackson LA, Anderson EJ, Rouphael NG, PC Roberts, Mamodikoe Makhene et al. (2020) An mRNA vaccine against SARS-CoV-2 preliminary report. N Engl J Med 383: 1920-19
- (2020) Medicines & Healthcare Products Regulatory Agency (MHRA). Information for Healthcare Professionals on COVID-19 Vaccine AstraZeneca.
- McFadyen JD, Stevens H, Peter K (2020) The emerging threat of (micro)thrombosis in COVID-19 and its therapeutic implications. Circ Res 127(4): 571-5
- Chatterjee S, Ojha UK, Vardhan B (2021) Myocardial infarction after COVID-19 vaccination-casual or causal? Diabet Metab Syndr Clin Res Rev 15(3): 1055-1056.
- Stone CA, Rukasin CR, Beachkofsky TM, Phillips EJ (2019) Immune-mediated adverse reactions to vaccines. Br J Clin Pharmacol 85(12): 2694-2706.
- Maadarani O, Bitar Z, Shoeb S, Alsaddah J (2020) From Wellens to Kounis syndrome: an unlucky patient. Eur J Case Rep Inter Med 7(9).
- Boivin Z, Martin J (2021) Untimely Myocardial Infarction or COVID-19 Vaccine Side Effect. Cureus 13: e13651.
- Maadarani O, Bitar Z, Elzoueiry M, Nader M, Abdelfatah M, et al. (2021) Myocardial infarction post COVID-19 vaccine -coincidence. Kounis syndrome or other explanation- time will tell 12(8): 1-3.
- Tajstra M, Jaroszewicz J, Gasior M (2021) Acute Coronary Tree Thrombosis After Vaccination for COVID-19. JACC Cardiovasc Interv 14(9): e103-e104.
- Shimabukuro TT, Nguyen M, Martin D, DeStefano F (2015) Safety monitoring in the Vaccine Adverse Event Reporting System (VAERS). Vaccine 33(66): 4398-4405.
- Julia WG, Wallace M, Stephen CH, Langley G, John RS, et al. (2021) Use of mRNA COVID-19 Vaccine after Reports of Myocarditis among Vaccine Recipients: Update from the Advisory Committee on Immunization Practices-United States, June. Morbidity and Mortality Weekly Report (MMWR) 70(27): 977-982.

 Case Report
Case Report