Abstract
COVID-19 is a disease caused by Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2). The introduction of vaccines against COVID-19 caused great impacts in decreasing the severity of disease and in reducing further infection. However, COVID-19 vaccination related adverse events have caused a great concern in the public. We recently had an unusual case who presented with multi-organ involvement after Pfizer-BioNTech COVID-19 vaccination. The history was reported and the literatures were reviewed.
Keywords: Adverse Events; COVID-19 Vaccine; Gastroenteritis; Myocarditis; Pericarditis; Pneumonitis
Abbreviations: SARS-CoV-2: Severe Acute Respiratory Syndrome Coronavirus-2; FDA: Food and Drug Administration; BNT: BioNTech COVID-19; DOE: Dyspnea on Exertion; PCT: Procalcitonin; CT: Computed Tomography; CV: Cardiovascular; CDC: Centers for Disease Control; MIS-C: Multisystem Inflammatory Syndrome in Children; ATAGI: Australian Technical Advisory Group on Immunization; CSANZ: Cardiac Society of Australia and New Zealand; CK: Creatine Kinase; GOT: Glutamic Oxaloacetic Transaminase; GPT: Glutamic Pyruvic Transaminase; ALP: Alkaline Phosphatase; Bil. (T), Bil.(D): Total Bilirubin Or Direct Bilirubin; CK: Creatine Phosphokinase; CK-MB: Creatine Phosphokinase-MB Isoenzyme; Pro-BNP: NT-Pro-BNP or N-Terminal Pro-Brain Natriuretic Peptide; CRP: C-Reactive Protein
Introduction
The two mRNA vaccines [Pfizer/BioNTech (BNT162b2) and Moderna (mRNA-1273)] demonstrated excellent safety and clinical efficacy profiles in clinical trials in adults. The United States Food and Drug Administration (FDA) has granted emergency authorization to the Pfizer/BioNTech (BNT162b2) and Moderna (mRNA-1273) COVID-19 vaccines on December 11, 2020 and December 18, 2020, respectively [1,2]. The commonly reported side effects of the vaccines included pain, redness and swelling at the administration site, malaise, headache, myalgias, chills, nausea, and fever. Myocarditis and/or pericarditis have been reported as rare side effects after mRNA COVID-19 vaccines (including Pfizer/ BioNTech and Moderna) in adults, particularly young adults [3-5]. According to the best of our knowledge, the COVID-19 vaccination that involved multi-organs simultaneously (including acute gastroenteritis, hepatitis, pneumonitis, myocarditis and pericarditis) has not been reported previously. We hereby reported an interesting case with the above adverse events after Pfizer- BioNTech COVID-19 vaccination and review the relevant literatures.
Case Report
The patient was a 20 year-old healthy male with no previous
medical history. He had received the first injection of Pfizer-
BioNTech COVID-19 (BNT) vaccine on October 14, 2021. High fever
(39 oC) developed in the evening after BNT vaccination. He received
antipyretic drug which alleviated the fever temporarily. He then visited a local clinic and received antipyretic/analgesic medicines
(Panadol 1tab PO TID) but the fever occurred intermittently. Due to
persistent fever, cough, impaired appetite, and dyspnea on exertion
(DOE), so he was brought to our emergency department for further
evaluation and treatment on October 18, 2021. In the emergency
department, a real-time reverse transcriptase–polymerase chain
reaction (RT-PCR) test for COVID-19 was performed with a negative
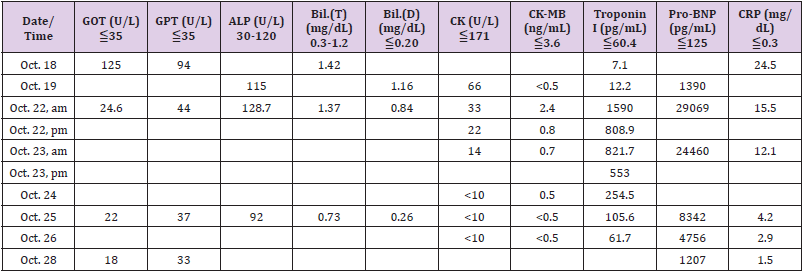
result. Other laboratory data showed increased C-reactive protein
(CRP, 24.5mg/dL), hyperbilirubinemia [Bil(D)=1.16 mg/dL;
Bil(T)=1.42 (mg/dL)], and abnormal liver function test [GOT=125
U/L, GPT(ALT)=94U/L]. Patient was then admitted to the
gastrointestinal (G-I) ward with preliminary impression of acute
hepatitis on October 18th. At the G-I ward, infection was suspected
due to high CRP and leukocytosis with left shift (WBC=12.1 10 ^3/
uL; Neutrophil=81.9 %). The Flumarin (Oxacephem) was used
for board-spectrum coverage to treat the infection. In addition,
patient had abnormal pro-BNP level (Pro-BNP=1390.0pg/mL)
and echocardiogram was studied, which revealed normal chamber
size and left ventricular function (left ventricular ejection fraction,
LVEF=67%). No regional wall motion abnormality was found. On
October 20th, patient again developed high fever (39.3oC), right
shoulder pain, epigastric pain, impaired appetite, and abdominal
distention. Passage of loose stool and sometimes watery stool for
more than 3 times daily was also noted.
The gastroenteritis was highly suspected. The Kascoal
(Simethicone, 40mg) and Smecta (dioctahedral smectite, 3g) three
times daily were prescribed. Meanwhile, the antibiotic was changed
to ciprofloxacin for possible atypical infection. The passage of loose
stool was persisted and Smecta was used until October 25th when his
discomfort was under controlled. Abdominal ultrasound revealed
no obvious focal lesions. However, hypotension (BP=81/52mmHg)
was noted on October 20th. After 500 cc Lactated ringer infusion,
the blood pressure increase to 101/66mmHg. Unfortunately,
hypotension (81/49mmHg) recurred in the evening of October
21st even after 1000 cc of the normal saline infusion. Chest X-ray
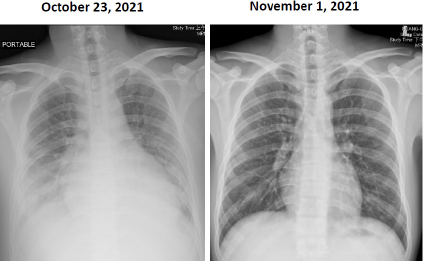
showed cardiomegaly with bilateral pleural effusion and increased
infiltration in both lower lungs, which suggesting the possibility
of pneumonitis (Figure 1). Due to DOE, cardiomegaly with pleural
effusion and elevated troponin I level (Troponin I=1590.0pg/
mL), the acute myocarditis related to Pfizer-BNT vaccination
was highly suspected. Thus, patient was transferred to ICU for
further evaluation and treatment on October 22nd. In the ICU, due
to suspected myocarditis, echocardiography was studied again
on October 22nd, which revealed normal chamber size, small
amount pericardial effusion, preserved LV systolic performance
( LVEF=62 %), and mild tricuspid regurgitation and mitral
regurgitation. Since the pericardial effusion was not observed on
the initial echocardiography performed 4 days ago, the new ECHO
result indicated the newly developed pericarditis. Despite the
use of antipyretic medication (Panadol 500mg TID), the patient
had persistent fever (Figure 2), although the fever was trending
down gradually. Since the procalcitonin (PCT) level was not high
(PCT=1.15ng/mL), the possibility of significant bacterial infection
was excluded. Due to abdominal distention, dyspnea on exertion,
and chest pain, we performed abdomen and chest computed
tomography (CT) scans on October 22nd. These studies revealed
1) Air space infiltration in both lungs, consistent with pneumonia,
2) Subcutaneous edema of body wall of abdomen, suggesting of
hypoalbuminemia,
3) Ascites and bilateral pleural effusions.
Figure 1:
Left: A chest film showing cardiomegaly with bilateral pleural effusion and increased infiltration in both lower lungs, which
consistent with bilateral pneumonitis.
Right: Chest film examined after discharge, which demonstrates considerable improvement compared to that of the right-side
chest film.
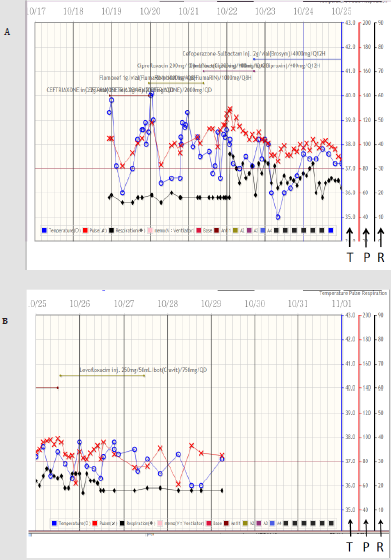
Figure 2:
Upper panel showing the curves of temperature, pulse rate, and respiratory rate, respectively from the 23:30 of October 18th
(10/18) to the 0:00 of October 25th (10/25). The blue circle, red cross, and black diamond represent the body temperature, pulse
rate, and respiratory rate, respectively. The scales over the right side of the figures indicating those of the temperature (T), pulse
rate (P), and respiratory rate (R), respectively. Please note, the high fever (>39 oC) exists during the initial 3 admission days.
Lower panel showing the curves of the temperature, pulse rate, and respiratory rate, respectively from 0:00 of October 25th to
0:00 of November 1st.
Due to hypoalbuminemia (Alb=2.72g/dL), dyspnea on exertion,
cough, ascites, and bilateral pleural effusion, Albumin 100 ml and
Lasix 20mg IV drip daily were used continuously for 6 days. Patient
received vitamin D3 supplement treatment due to insufficient
serum vitamin D level (Vitamin D, 25-OH-D=20.4ng/mL). The
dosing schedule was 70,000 IU orally per day for 4 days, followed
with 4000 IU daily until discharge. Lab data revealed that the
Troponin I decreased from 1590.0pg/mL to 61.7pg/mL; Pro-BNP
dropped from 29069pg/mL down to 1207pg/mL; CRP dropped
from 24.5mg/dL to 1.5mg/dL after 7 days treatment.
His body temperature decreased gradually (ranged from 37.5 oC
to 37.8 oC) during October 24th to 27th. Follow up echocardiography
disclosed decrease in the amount of pericardial effusion on October
25th. On October 26th, the follow-up Lab data revealed considerable
improvement (Table 1). Chest x-ray film also showed improvement
of bilateral pleural effusion. After our aggressive treatments, his GI
discomfort, chest pain, and shortness of breath improved gradually.
Patient was transferred from the ICU to cardiovascular (CV) ward
on October 26th and discharged on October 28th. After discharge, he
has visited our CV clinic on November 1st. The follow-up chest x-ray
film showed considerable improvement compared to previous chest
x-ray film (Figure 1). There was no more cough or DOE. He received
Kascoal (Simethicone) 40mg/tablet and Alginic acid (Algitab)
200mg/tab for symptomatic treatment of the GI discomfort.
Discussion
In the WHO’s website, it seems that the reported side effects
of COVID-19 vaccines have mostly been mild to moderate and
have lasted no longer than a few days [6]. However, it was not
the case for this patient. This patient developed persistent fever,
cough, impaired appetite, and dyspnea on exertion after the
first dose of Pfizer-BioNTech COVID-19 vaccine injection. The
clinical and laboratory data subsequently showed the presence
of acute gastroenteritis, hepatitis, pneumonitis, myocarditis and
pericarditis. A recent article [7] has reported that the local and
systemic reactions to mRNA-based COVID-19 vaccines occurred 0
to 7 days after vaccination, based on the Centers for Disease Control
and Prevention (CDC) vaccine safe (v-safe) surveillance system.
Data from millions of v-safe participants showed that the injection
site pain and swelling were common for the local reactions after
either of the mRNA-based vaccines (Pfizer and Moderna). As for
the systemic reactions, more than 3% of the reported systemic
symptoms included fatigue, headache, myalgia, chills, fever, joint
pain, nausea, diarrhea, and abdominal pain. Although these
reactions could occurred in participants after the first dose, they
were more common after the second dose with either Pfizer-
BioNTech or Moderna vaccine. These reactions were thought to be
caused by the host’s immune response. That article only described
the local and systemic reactions within 7 days. In our report, the
patient had high fever (>39 oC), measured by ear temperature, that
persisted for 7 days (from October 14th to 20th) after receiving the
first dose of vaccine injection. The body temperature >38 oC was
noted for 3 days (from October 21st to 23rd) and the temperature
>37.5 oC was noted for 4 more days (from October 24th to 27th).
Thus, the period of high fever days (7 days) and the total fever days
(14 days) were unusual for this case.
Another point needed to be mentioned was that it usually takes
a few weeks for the body to build immunity against SARS-CoV-2
virus after vaccination. Thus, it’s possible that this individual could
be infected with SARS-CoV-2 virus just before or after vaccination
because the body did not have enough time to provide protection.
Since this patient’s RT-PCR test for COVID-19 was negative in the
ED, the possibility of SARS-CoV-2 infection was unlikely. This case
had evidence of myocarditis and pericarditis after Pfizer-BioNTech
COVID-19 vaccination. It was consistent with previous report that there was absence of myocarditis and rare cases of pericarditis after
receiving the non-mRNA COVID-19 vaccines (such as AstraZeneca
vaccine or Johnson & Johnson vaccine). However, cases of
myocarditis and pericarditis in young men after mRNA vaccination
were rising [8]. Several explanations have been hypothesized
regarding why the myocarditis and pericarditis only occurred after
the mRNA-based vaccines. First, a very strong antibody response
was suspected to exist in a small proportion of the young adults
that could produce a response similar to multisystem inflammatory
syndrome in children (MIS-C) associated with the SARS-CoV-2
infection [9]. Second, the mRNA vaccines could induce a non-specific
innate inflammatory response or a molecular mimicry mechanism
between the viral spike protein and an unknown cardiac protein
[10]. Molecular mimicry mechanism was caused by a significant
similarity between certain pathogenic elements contained in the
vaccine and the specific human proteins.
This similarity may lead to immune cross-reactivity, in which
the reaction of the immune system towards the pathogenic
antigens may harm human proteins that were similar in structures,
essentially causing autoimmune diseases. Third, the mRNA, a
potent immunogen, in the vaccine itself could trigger an adjuvant
effect [11] by cytokine activation of the pre-existing autoreactive
immune cells, which subsequently could induce autoimmune
myocarditis. In August 2021, the Australian Technical Advisory
Group on Immunization (ATAGI) and the Cardiac Society of Australia
and New Zealand (CSANZ) have proposed a guidance in evaluating
myocarditis and pericarditis after mRNA COVID-19 vaccination.
This guidance suggested that findings consistent with myocarditis
may include elevated troponin, ST or T-wave abnormalities
observed on the ECG, premature atrial or ventricular complexes,
and abnormal echocardiogram or cardiac MRI [12]. However, this
guideline only suggested evaluating troponin but not the other
cardiac enzymes such as creatine kinase (CK) or CK-MB isoenzyme.
In our patient, it is interesting that the troponin I increased to a
high level (1590.0pg/mL) on October 22nd and persisted for 4 more
days (until October 25th). However, the other cardiac enzyme never
elevated significantly during the whole course of hospitalization
(Table 1). This specific phenomenon is worth to be mentioned.
Previous reports have found that vitamin D had immense
benefits in the immune dysfunction observed in COVID-19 patients
with cytokine storms [13-15]. It was also well known that vitamin D
modulated the immune system by down-regulating the production
of self-damaging pro-inflammatory cytokines and promoted
the expression of anti-inflammatory cytokines by the immune
cells [16-19]. Since the patient in this report had relatively low
serum vitamin D level, he therefore received high dose vitamin
D supplement treatment. The pneumonitis, myocarditis and
pericarditis resolved smoothly after our treatment. Since vitamin D
has immune-modulatory effects, we speculated that vitamin D may
contribute to some extent to the recovery of this patient.
In conclusion, this report described an interesting case that
had unusual presentations and with multi-organ involvements
post Pfizer-BioNTech COVID-19 vaccination. After our aggressive
treatment including vitamin D supplementation, he recovered
completely without persistent symptoms.
Authors’ Contribution
All authors have substantial contributions on the data collection, interpretation, and writing the manuscript.
Declaration of Conflict of Interest
The authors declare that there was no conflict of interest.
Financial Support
The authors declare that they have not received any financial support.
References
- Polack FP, Thomas SJ, Kitchin N, Judith Absalon, Alejandra Gurtman, et al. (2020) Safety and efficacy of the BNT162b2 mRNA covid-19 vaccine. N Engl J Med 383(27): 2603-2615.
- Baden LR, El Sahly HM, Essink B, Karen Kotloff, Sharon Frey, et al. (2021) Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N Engl J Med 384(5): 403-416.
- Kim HW, Jenista ER, Wendell DC, Clerio F Azevedo, Michael J Campbell, et al. (2021) Patients with acute myocarditis following mRNA COVID-19 vaccination. JAMA Cardiol 6(10): 1196-1201.
- Abu Mouch S, Roguin A, Hellou E, Amorina Ishai, Uri Shoshan, et al. (2021) Myocarditis following COVID-19 mRNA vaccination. Vaccine 39(29): 3790-3793.
- Larson KF, Ammirati E, Adler ED, Leslie T Cooper, Kimberly N Hong, et al. (2021) Myocarditis after BNT162b2 and mRNA-1273 Vaccination. Circulation 144(5): 506-508.
- (2021) Side effects after COVID-19 Vaccine.
- Chapin Bardales J, Gee J, Myers T (2021) Reactogenicity following receipt of mRNA-based COVID-19 vaccines. JAMA 325(21): 2201-2202.
- Das BB, Moskowitz WB, Taylor MB, Palmer A (2021) Myocarditis and pericarditis following mRNA COVID-19 vaccination: what do we know so far? 8(7): 607.
- Grimaud M, Starck J, Levy M, Clémence Marais, Judith Chareyre, et al. (2020) Acute myocarditis and multisystem inflammatory emerging disease following SARS-CoV-2 infection in critically ill children. Ann Intensive Care 10: 69.
- Segal Y, Shoenfeld Y (2018) Vaccine-induced autoimmunity: The role of molecular mimicry an immune cross-reaction. Cell Mol Immunol 15(6): 586-594.
- Root Berstein R, Fairweather D (2015) Unresolved issues in theories of autoimmune disease varying myocarditis as a framework. J Theor Biol 375: 101-123.
- (2021) Guidance on myocarditis and pericarditis after mRNA COVID-19 vaccines.
- Jain A, Chaurasia R, Sengar NS, Singh M, Mahor S, et al. (2020) Analysis of vitamin D level among asymptomatic and critically ill COVID-19 patients and its correlation with inflammatory markers. Sci Rep 10(1): 20191.
- Huang C, Wang Y, Li X, Lili Ren, Jianping Zhao, et al. (2020) Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 395(10223): 497-506.
- Ye Q, Wang B, Mao J (2020) The pathogenesis and treatment of the “Cytokine Storm” in COVID-19. J Infect 80(6): 607-613.
- Chambers ES, Hawrylowicz CM (2011) The impact of vitamin D on regulatory T cells. Curr Allergy Asthma Rep 11(1): 29-36.
- Adams JS, Hewison M (2008) Unexpected actions of vitamin D: New perspectives on the regulation of innate and adaptive immunity. Nat Clin Pract Endocrinol Metab 4(2): 80-90.
- Lemire JM, Adams JS, Kermani Arab V, Bakke AC, Sakai R, et al. (1985) 1,25-Dihydroxyvitamin D3 suppresses human T helper/ inducer lymphocyte activity in vitro. J Immunol 134(5): 3032-3035.
- Gombart AF, Pierre A, Maggini S (2020) A Review of micronutrients and the immune system-working in harmony to reduce the risk of infection. Nutrients 12(1): 236.

 Case Report
Case Report