Abstract
Background: In 2020, WHO reported that stroke is the second leading cause of
death worldwide. Stroke recovery is the complex process due to its heterogeneous
nature. Identification of reliable recovery biomarkers assist to advance the practice,
rehabilitation and recovery after stroke.
Objective: This systematic review aimed to assess the biomarkers and predictors of
recovery in ischemic stroke.
Methods: Case-control studies reporting the biomarkers and predictors of recovery
in ischemic stroke patients were identified through PubMed, Wiley online library,
Rehab data and PEDro databases. Studies in English language published after the year
2000 were included. Studies on animal models and patients with other psychiatric or
neurological condition (other than stroke) were excluded. The Newcastle-Ottawa Scale
(NOS) was used to assess the quality of studies. Relative levels of recovery biomarkers
in post-ischemic stroke patients were compared with the levels found in control
subjects. Random effect model was used to calculate the mean effect size of studies. This
systematic review was registered with PROSPERO (CRD42020209833).
Results: The results of this systematic review are based on 29 studies that covered
2528 participants (case:926 patients, control:1602 participants). A total of 22 markers
were reported in this review. The core biomarkers differentiated ischemic stroke’s
recovery from controls with good performance: Motor evoked potential [standardized
mean difference -2.14, P=0.03], laterality index [standardized mean difference -1.32,
P=0.03] and stimulus intensity [standardized mean difference 1.63, P=0.18].
Conclusion: The motor evoked potential, laterality index and stimulus intensity
were found as relevant biomarkers that predict motor recovery in ischemic stroke
patients.
Keywords: Laterality Index; Motor Evoked Potential; Recovery; Biomarker; Ischemic Stroke
Abbreviations: NIHSS: National Institutes of Health Stroke Scale; EHI: Edinburg Handedness; UE-MAS: Upper Extremity- Motor Assessment Scale; ARAT: Action Research Arm Test; BIT: Behavioral Inattention Test; CMSA: Chedoke Mc Master Score; MoCA: Montreal Cognitive Assessment; FM: Fugyl Meyer; WMFT: Wolf Motor Function Test; MAS: Modified Ashworth Scale; MRS: Modified Rankin Scale; AMAT: Arm Motor Ability Test; MMSE: Mini–Mental State Examination; DASS: Depression and Anxiety Stress Scale; MRI: Magnetic Resonance Imaging; fMRI: functional MRI; FA: Fractional Anisotropy; MR-CHIROD: Magnetic Resonance -Compatible Hand-Induced, Robotic Device; WAISR: Wechsler Adult Intelligence Scale-Revised; WCST: Wisconsin Card Sorting Test
Introduction
According to the World Health Organization, 15 million
people worldwide suffer a stroke each year. Of these, nearly five
million die and another five million are left permanently disabled
[1]. Millions of stroke survivors are left with very limited motor
functions or complete paralysis and depend on assistance [2]. Poststroke
survivors majorly suffer from motor, sensory, cognition and
language impairments. These impairments occur due to the loss
of functions of brain regions surrounding the area of injury due
to lesions which further lead to death of neurons of those regions
[3]. Majority of cases of stroke are ischemic stroke [4]. Stroke
recovery is the complex process due to its heterogeneous nature,
where the making choices of treatment and prediction of outcomes
and treatment responses are difficult [5].Prediction of outcomes
in stroke can be useful to determine which intervention (such as
behavioural and therapeutic interventions) will be more effective
in stroke patients during the rehabilitation.
However, the methods for accurate prediction of longterm
outcomes would allow clinical trials of restorative and
rehabilitation interventions to be stratified based on the potential
for neurobiological recovery in a way that is not possible if trials
are performed in the absence of valid biomarkers [5,6]. A Stroke
Recovery Biomarker (SRB) can be defined as an indicator of disease
state that can be used as a measure of underlying molecular or
cellular processes that may be difficult to measure directly in
humans, and could be used to understand the outcome, or predict
the recovery or treatment response [5]. Thus, when dealing with
a condition as heterogeneous as stroke, validated biomarkers
of recovery could help plan treatments and support efficient
allocation of resource while maximizing outcome for the patients
[7]. Identification of recovery biomarkers shall assist to advance the
practice, rehabilitation and recovery after stroke. This systematic
review has assessed the role of biomarkers and predictors of
recovery in ischemic stroke.
Material and Methods
This systematic review was designed as per the guidelines of Preferred Reporting Items for Systematic reviews and Meta- Analysis (PRISMA) [8-10] and registered with PROSPERO (CRD42020209833) on 18th October 2020 [11].
Eligibility Criteria
Case-control studies reporting the biomarkers and predictors
of recovery in ischemic stroke patients in English language
published after the year 2000 were included. Studies on animal
models, including diagnostic markers, prognostic markers and
patients with other psychiatric or neurological conditions (other
than stroke) were excluded.
The PICO (Population, Intervention, Comparator, and Outcome)
format for this systematic review is:
• P: Patients diagnosed with ischemic stroke and no other
psychiatric or neurological condition (other than stroke).
• I: Biomarkers or predictors of recovery in ischemic stroke.
• C: Relative levels of recovery biomarkers in post ischemic
stroke patients were compared with the levels of recovery
biomarkers found in control subjects.
• O: The most important outcome is the recovery of patients
with ischemic stroke. The extent of recovery in ischemic
stroke patients is mainly in terms of motor recovery, cognition
recovery, sensory and language recovery. Recovery outcomes
were measured using the standardized mean difference in
level of biomarkers for individual studies.
Information Source
A systematic literature search was conducted using PubMed, Wiley online library, Rehab Data and PEDro Database. First, a comprehensive search was performed individually for all components of PICO. Later individual searches were combined with the help of Boolean operators (AND, OR, NOT). Search was performed with a combination of MeSH (Medical Subject heading terms) and keywords terms. The MeSH terms searched were “stroke”, “Rehabilitation”, “Biomarker” and “Brain Infarction”. Search was performed in September 2020 and was also updated before the submission. Search strategy was developed as per the Cochrane checklist of developing search strategy. [12] Structured strategy for PubMed is given in the Appendix A1.
Study Selection
Studies obtained through initial search from different databases were combined after removing duplicates using the Endnote “find duplicates” filter. A single reader read the title and abstract of all the obtained references. Screening of search records was performed against the predefined inclusion criteria and irrelevant studies were excluded. The reasons for excluding the studies during screening were also documented. Once preliminary articles were identified as potentially eligible, the papers were fully reviewed by two members to determine inclusion; and, disagreements in between were resolved by consensus. In case of multiple publications of the same study, the most recent publication was considered. Bibliographies and citation sections of retrieved articles had been reviewed for additional pertinent studies.
Data Extraction
A pre-specified data collection template was used to extract data
from the articles obtained after screening and all disagreements were resolved by discussion among team members. The following
information was extracted from the potentially eligible full-text
studies;
• Publication details: Author’s name, year, journal’s name,
country.
• Population related details: Total enrolled participants, gender,
age, days post stroke, stage of stroke, number of participants in
case-control groups.
• Intervention related details: Name of biomarker or predictor,
type of biomarker or predictor, method to recognize biomarker
or predictor, levels of biomarker or predictor in case-control
groups.
• Outcome related details: Type of recovery, recovery in casecontrol
groups, tools for assessing recovery, National Institute
of Health Stroke Scale (NIHSS) score, Modified Ranklin Scale
(MRS) score, correlation values in between biomarker/
predictor and recovery.
Quality Assessment
The Newcastle-Ottawa Scale (NOS) was used to assess the quality of studies independently by two authors. The NOS consists of three domains: selection, comparability, and outcome/exposure. It assigns a maximum of four stars for the selection, two stars for comparability and three stars for the exposure category. Thus, nine stars altogether indicate the high quality, seven to eight stars indicate medium quality and six or less stars indicate low quality. Any conflicts were resolved by consensus and individual score of each study was recorded which represents the quality of study.
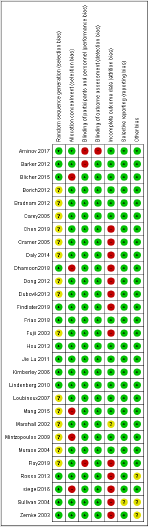
Risk of Bias in Individual Study
The Cochrane Collaboration’s tool for assessing the risk of bias was used for the risk-bias-assessment at the study level. The assessment was done for the domains namely random sequence generation and allocation concealment for selection bias; incomplete outcome data (attrition bias); selective reporting of outcome (reporting bias); and other biases including publication bias [12].
Data Synthesis and Analysis
The extraction of data from eligible studies was done in Excel spreadsheet, Microsoft Office 2010 (Washington, USA). A meta-analysis of studies was performed through standardized mean difference of recovery biomarker levels. The mean levels of recovery biomarkers or predictors obtained by selected studies were compared in between case and control groups. A value of P<0.05 was interpreted as statistically significant. The I² index was used to assess heterogeneity between studies [13,14]. Random effect and fixed effect models were used to calculate the mean effect size of studies with significant heterogeneity (I² >75%) and without significant heterogeneity (I² <75%), respectively. For systematic review and risk of bias assessment, Review Manager 5.3, Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014 was used [15].
Results
Study Selection
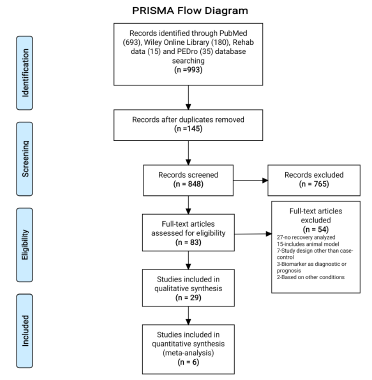
A total of 993 potentially relevant studies were identified using online databases (PubMed, Wiley online library, Rehab Data and PEDro Database) through a systematic search strategy. The 145 duplicates were found and removed after combining the studies obtained from different databases. After screening the titles of 848 studies, 765 studies were excluded, as they were found irrelevant. Further abstract screening of the remaining 83 studies found 32 studies ineligibles as study design was other than case-control, animal models, diagnostic or prognostic markers, based on other conditions and no recovery was assessed. Thus, a total of 51 studies were identified for full text screening. Identified full text studies were retrieved and screened for the eligibility. After detailed evaluation, 29 studies were found to be eligible for final analysis and measuring at least one or more of the considered outcomes in this systematic review [7,16-43]. The PRISMA flow diagram for selection of studies has been represented in (Figure 1).
Study Characteristics
The results of this systematic review are based on 29 studies
involving 2528 participants. Out of 2528 participants, 926 were in
the case group (patients diagnosed with ischemic stroke) and 1602
were in the control group (healthy volunteers). The average age and
range of pooled patients were 59.49±5.98 years and 21-93 years,
respectively. The results of 12 studies were based on acute stage
stroke patients, nine studies on chronic stage, and three studies on
sub-acute, two studies on combination of acute and chronic stage
and in remaining three studies stage was not specified. Thus, the
majority of participants (567/926) in the case group were found
with acute stage of Ischemic stroke while, 237/926 participants
were with chronic stage. The average months after the occurrence
of stroke, when recovery was assessed in participants were found
to be 19.36±27.55 months.
The majority of studies (20/29) reported motor recovery
while few studies reported cognition recovery (3/29), sensory
recovery (2/29), combination of motor and cognition recovery
(3/29) and combination of motor and sensory recovery (1/29).
Total 27 different measures were used to capture the outcome
(recovery) in the participants enrolled in the included studies. The
most common used measures of recovery were: General measures-
National Institute of Health Stroke Scale (NIHSS), Modified Ranklin
Scale (MRS); Motor recovery measures- Fugl Meyer Assessment
(FMA), Wolf Motor Function Test (WMFT), Action Research Arm
Test (ARAT), Arm Motor Ability Test (AMAT), Motor Assessment
Scale, Finger Tapping (FT), Ashworth Scale, Chedoke-McMaster
Stroke Assessment (CMSA), Barthel Index (BI), Grip strength,
and Edinburg Handedness Inventory (EHI quotient); Cognition
recovery measures-Mini–Mental State Examination (MMSE) and
Montreal Cognitive Assessment (MCA). The publication, population,
intervention and outcome characteristics of all selected studies are
presented in the Appendix Table 1.
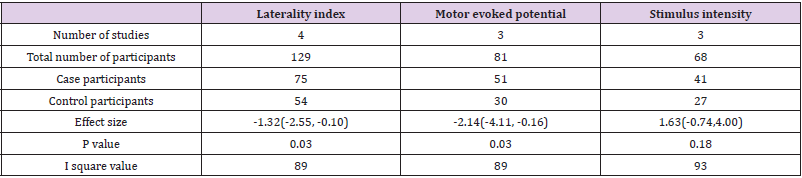
Table 1: Overall effect estimate of laterality index, motor evoked potential and stimulus intensity.
Biomarkers and Predictors
A total of 22 markers were reported in this systematic review. Further, these markers were categorized in sections: Motor Evoked Potential (MEP), Laterality, Cortico-Spinal Tract (CST), brain oscillatory activity, brain connectivity and activation, location, size and volume, cerebral blood flow and others. The details of the markers and their association with the recovery in Ischemic stroke patients are presented in the Appendix Table 2. Predictors like MEP (onset, amplitude, area and selectivity), laterality (laterality changes and index), brain oscillatory activity (event related desynchronization), brain connectivity and activation (resting state functional connectivity, ipsilesional cortico-cerebellar functional connectivity, brain activation and stimulus intensity), CST (damage, weighted CST lesion load) were found positively correlated with the recovery in ischemic stroke. On another hand, predictors like lesion size, infarct volume, lesion topography and white matter integrity were found negatively correlated with the recovery.
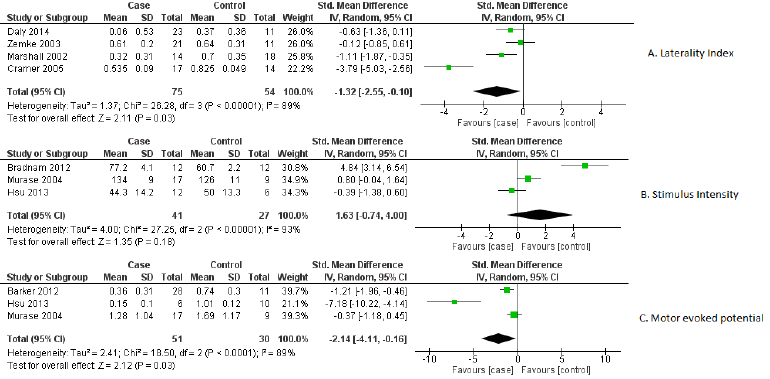
Out of 22 markers, three relevant markers were analysed by performing meta-analysis. The performance of these markers was measured in terms of their capability of differentiating the recovery of ischemic stroke from healthy participants. The standardized mean difference value of markers was used as an outcome measure for differentiation between case-control groups. As a significant heterogeneity was found, therefore the random-effects model was chosen over the fixed effects model. These relevant biomarkers differentiated ischemic stroke’s recovery from controls with good performance: MEP [standardized mean difference -2.14, 95% CI (-4.11, -0.16), P =0.03], laterality index [standardized mean difference -1.32, 95% CI (-2.55, -0.10.), P =0.03] and stimulus intensity [standardized mean difference 1.63, 95% CI (-0.74, 4.00), P =0.18]. The individual study effect sizes were reported in the forest plots based on outcomes. In view of the study wise reporting of markers, sample size was the highest for laterality index (129 participants), whereas the sample size for MEP and stimulus intensity were 81 and 68 participants respectively. The overall effect estimates of laterality, MEP and stimulus intensity were represented in (Table 1). The visual examination through the forest plot reflects that laterality index and MEP markers favours the case group. While the marker stimulus intensity favours the control group. The forest plot of these above mentioned three markers were represented in (Figure 2).
Figure 2: Forest plot of biomarkers
A) Laterality index,
B) Stimulus intensity and
C) Motor evoked potential.
Quality of Studies
Three parameters of quality: selection, comparability, and outcome/exposure were measured through NOS scale. One study was found to have high quality (nine point’s altogether), 23 studies had medium quality (seven-eight points) and five studies were poor quality reports (≤ six points).
Risk of Bias
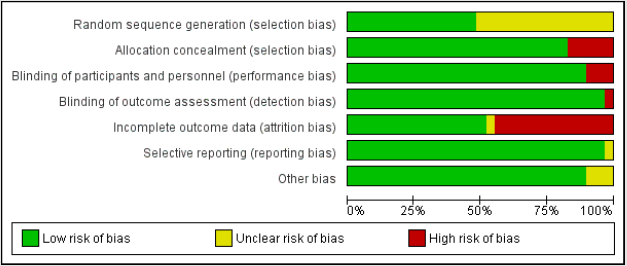
Mainly selection and attrition bias were observed within the selected studies. In few studies reporting and performance biases were also detected as the outcome was found to be reported only for selected groups. The risk of bias was considered adequate for the outcomes. The risk of bias for individual study and their summary were represented in (Figures 3 & 4) respectively.
Discussion
This review has systematically evaluated the biomarkers
and predictors of recovery in ischemic stroke as well as their
association with the recovery. A number of studies have assessed
the association of various markers with the recovery after stroke.
Among these, studies with case-control design are growing in
number to determine the best predictor which can differentiate
recovery in stroke patients from severely affected ones as well
as healthy participants. To the best of knowledge, this is the first
systematic review based on a case-control design to evaluate the
markers which can predict and differentiate the recovery after
the stroke. Through the comprehensive literature search, a total
of 22 rapidly growing markers were found to be associated with
the recovery after stroke. These markers were further categorized
into eight broad categories in this review. The results of this review
indicate that out of 22 markers, the laterality index and MEP were
the only biomarkers that show relevant association with recovery.
Hayward, et al. [44] reported MEP as the relevant biomarker to
predict motor recovery which is consistent with our results. In
addition, the results of this study demonstrated that the laterality
index is also strongly associated with the outcome. Single- or
repetitive-pulse stimulation of the brain causes the spinal cord
and peripheral muscles to produce neuro electric signals known as
MEPs [45]. MEPs or their absence serve as indications of the cortico
motor pathway’s functional integrity and excitability, facilitating
the assessment of associated motor impairment at the time of
testing [46]. In this review, different variables of MEP such as MEP
onset, MEP amplitude and MEP area were also found to be strongly
associated with the recovery. The presence of MEP indicated better
recovery and the mean values of MEP amplitude were found to be
higher in healthy subjects than ischemic stroke participants.
Aside from MEP, the laterality index was revealed to be a
relevant biomarker in this study. The laterality index is a method
of determining hemisphere dominance in a range of activities,
including language, cognitive skills, and changes in laterality in
clinical populations, as in post-stroke condition [47]. In post-stroke
fMRI investigations, it has been used to identify neuro plastic
alterations in stroke survivors [37]. Further, it was found to be
associated with greater activation in the hemisphere contralateral
to the working limb (lesioned hemisphere) versus the hemisphere
ipsilateral to the working limb (non-lesioned hemisphere) [24].
In this review, the laterality index was mainly extracted from
functional MRI (fMRI) in most of the included studies and revealed
that stroke is associated with a less lateralized pattern of activation
when compared to healthy participants. It further indicated, more
the shift towards the normal state of brain function is associated
with a better recovery stage after stroke. In contrast to MEP and
the laterality index, the pooled values of brain stimulus intensity
were identified as statistically insignificant biomarker in this
review. Increased stimulus intensity has been linked to a reduction
in response time which represents rapid sensory and perceptual
processing manifested in the presence of more intense physical
stimuli. Further, in stroke survivors, the role of stimulus intensity
was also detected in preparing voluntary movements using various
techniques such as transcranial direct current stimulation (tDCS)
and transcranial magnetic stimulation (TMS) [48]. However, the
stimulus intensity for tDCS cannot be individualized based on
motor thresholds as for repetitive TMS and may produce variable
effects between individuals [39].
In this review the pattern of mean values in the case of brain
stimulus intensity was found to be higher in ischemic stroke
participants than healthy participants. The mean values of markers
of the CST (CST integrity), brain oscillatory activity and connectivity
were slightly higher in healthy subjects. The markers like lesion
size, location, and infarct volume and lesion topography were
not found to have any significant association with the recovery. Authors also want to address the limitations of presented study.
Our literature search was constrained to articles only in the English
language, which creates the possibility to have somewhat biased
results. The outcome measurement in all the included studies was
made with a variety of tools and different endpoints. Due to the
limited individual patient data, pooling was not possible for all the
biomarkers. Thus, it further suggests future studies to investigate
recovery biomarkers mainly for cognition and sensory recoveries
after stroke.
Conclusion
This review concludes that laterality index, MEP and stimulus intensity are the most relevant biomarkers to predict motor recovery in ischemic stroke patients. The biomarkers of cognition and sensory recoveries, association of markers with individual recovery as well as combination of recoveries (like sensory & motor, cognition & motor and sensory, cognition & motor) need to be investigated more in future studies to predict the recovery with greater precision.
Acknowledgement
We thank National Institute of Plharmaceutical Education and Research (NIPER), Mohali, to make available library, computer laboratory facility and online accessibility of articles and other resources. We also present our gratitude towards Dr Mona Pathak for her guidance.
References
- (2019) World Health Organization. Who.int.
- Feigin VL, Norrving B, George MG, Foltz JL, Roth GA, et al. (2016) Prevention of stroke: a strategic global imperative. Nat Rev Neurol 12(9): 501.
- Feigin VL, Abajobir AA, Abate KH, Abd-Allah F, Abdulle AM, et al. (2017) Global, regional, and national burden of neurological disorders during 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet Neurol 16(11): 877-897.
- Mozaffarian D, Benjamin EJ, Go AS, Arnett DK, Blaha MJ, et al. (2016) Heart disease and stroke statistics—2016 update: a report from the American Heart Association. Circulation 133(4): e38-e360.
- Boyd LA, Hayward KS, Ward NS, Stinear CM, Rosso C, et al. (2017) Biomarkers of stroke recovery: consensus-based core recommendations from the stroke recovery and rehabilitation roundtable. Int J Stroke 12(5): 480-493.
- Kwakkel G, Lannin NA, Borschmann K, English C, Ali M, et al. (2017) Standardized measurement of sensorimotor recovery in stroke trials: consensus-based core recommendations from the stroke recovery and rehabilitation roundtable. Neurorehabil Neural Repair 31(9): 784-792.
- Ray AM, Figueiredo TD, López‐Larraz E, Birbaumer N, Ramos‐Murguialday A (2020) Brain oscillatory activity as a biomarker of motor recovery in chronic stroke. Hum Brain Mapp 41(5): 1296-1308.
- Moher D, Liberati A, Tetzlaff J, Altman DG, Prisma Group (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 6(7): e1000097.
- Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, et al. (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol 62(10): e1-e34.
- Beller EM, Glasziou PP, Altman DG, Hopewell S, Bastian H, et al. (2013) PRISMA for abstracts: reporting systematic reviews in journal and conference abstracts. PLoS Med 10(4): e1001419.
- Tiwari P, Rani P (2020) Recovery biomarkers of Ischemic Stroke: A Systematic Review and Meta-analysis. PROSPERO.
- Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, et al. (2019) Cochrane handbook for systematic reviews of interventions. John Wiley & Sons.
- Higgins JP, Thompson SG (2002) Quantifying heterogeneity in a meta‐analysis. Stat Med 21(11): 1539-1558.
- Sterne JA, Egger M (2001) Funnel plots for detecting bias in meta-analysis: guidelines on choice of axis. J Clin Epidemiol 54(10): 1046-1055.
- Review Manager (2014) (RevMan) [Computer program]. Version 5.3. Copenhagen: The Nordic Cochrane Centre, the Cochrane Collaboration.
- Aminov A, Rogers JM, Johnstone SJ, Middleton S, Wilson PH, et al. (2017) Acute single channel EEG predictors of cognitive function after stroke. PLoS One 12(10): e0185841.
- Barker RN, Brauer SG, Barry BK, Gill TJ, Carson RG, et al. (2012) Training-induced modifications of corticospinal reactivity in severely affected stroke survivors. Exp Brain Re 221(2): 211-221.
- Blicher JU, Near J, Næss-Schmidt E, Stagg CJ, Johansen-Berg H, et al. (2015) GABA levels are decreased after stroke and GABA changes during rehabilitation correlate with motor improvement. Neurorehabil Neural Repair 29(3): 278-286.
- Borich MR, Mang C, Boyd LA (2012) both projection and commissural pathways are disrupted in individuals with chronic stroke: investigating microstructural white matter correlates of motor recovery. BMC Neurosci 13(1): 1-11.
- Bradnam LV, Stinear CM, Barber PA, Byblow WD (2012) Contralesional hemisphere control of the proximal paretic upper limb following stroke. Cereb Cortex 22(11): 2662-2671.
- Carey LM, Abbott DF, Egan GF, Bernhardt J, Donnan GA (2005) Motor impairment and recovery in the upper limb after stroke: behavioral and neuroanatomical correlates. Stroke 36(3): 625-629.
- Chen L, Luo T, Wang K, Zhang Y, Shi D, et al. (2019) Effects of thalamic infarction on the structural and functional connectivity of the ipsilesional primary somatosensory cortex. Eur Radiol 29(9): 4904-4913.
- Cramer SC, Crafton KR (2006) Somatotopy and movement representation sites following cortical stroke. Exp Brain Res 168(1-2): 25-32.
- Daly JJ, Hrovat K, Holcomb J, Pundik S (2014) Brain control of functional reach in healthy adults and stroke survivors. Restor Neurol Neurosci 32(5): 559-573.
- Dhamoon MS, Cheung YK, Moon Y, DeRosa J, Sacco R, et al. (2018) cerebral white matter disease and functional decline in older adults from the Northern Manhattan Study: A longitudinal cohort study. PLoS Med 15(3): e1002529.
- Dong W, Yan B, Johnson BP, Millist L, Davis S, et al. (2013) Ischaemic stroke: the ocular motor system as a sensitive marker for motor and cognitive recovery. J Neurol Neurosurg Psychiatry 84(3): 337-341.
- Dubovik S, Ptak R, Aboulafia T, Magnin C, Gillabert N, et al. (2013) EEG alpha band synchrony predicts cognitive and motor performance in patients with ischemic stroke. Behav Neurol 26(3): 187-189.
- Findlater SE, Hawe RL, Mazerolle EL, Al Sultan AS, Cassidy JM, et al. (2019) Comparing CST lesion metrics as biomarkers for recovery of motor and proprioceptive impairments after stroke. Neurorehabil Neural Repair 33(10): 848-861.
- Frías I, Starrs F, Gisiger T, Minuk J, Thiel A, et al. (2018) Interhemispheric connectivity of primary sensory cortex is associated with motor impairment after stroke. Sci Rep 8(1): 1-10.
- Fujii Y, Nakada T (2003) cortical reorganization in patients with subcortical hemiparesis: neural mechanisms of functional recovery and prognostic implication. J Neurosurg 98(1): 64-73.
- Hsu YF, Huang YZ, Lin YY, Tang CW, Liao KK, et al. (2013) Intermittent theta burst stimulation over ipsilesional primary motor cortex of subacute ischemic stroke patients: a pilot study. Brain Stimul 6(2): 166-174.
- Kimberley TJ, Khandekar G, Skraba LL, Spencer JA, Van Gorp EA, et al. (2006) Neural substrates for motor imagery in severe hemiparesis. Neurorehabil Neural Repair 20(2): 268-277.
- Lindenberg R, Renga V, Zhu LL, Nair D, Schlaug GM (2010) Bihemispheric brain stimulation facilitates motor recovery in chronic stroke patients. Neurology 75(24): 2176-2184.
- Loubinoux I, Dechaumont-Palacin S, Castel-Lacanal E, De Boissezon X (2007) Prognostic value of FMRI in recovery of hand function in subcortical stroke patients. Cereb Cortex 17(12): 2980-2987.
- Lu J, Liu H, Zhang M, Wang D, Cao Y, et al. (2011) Focal pontine lesions provide evidence that intrinsic functional connectivity reflects polysynaptic anatomical pathways. J Neurosci 31(42): 15065-15071.
- Mang CS, Borich MR, Brodie SM, Brown KE, Snow NJ, et al. (2015) Diffusion imaging and transcranial magnetic stimulation assessment of transcallosal pathways in chronic stroke. Clin Neurophysiol 126(10): 1959-1971.
- Marshall RS, Perera GM, Lazar RM, Krakauer JW, Constantine RC, et al. (2000) Evolution of cortical activation during recovery from corticospinal tract infarction. Stroke 31(3): 656-661.
- Mintzopoulos D, Astrakas LG, Khanicheh A, Konstas AA, Singhal A, et al. (2009) Connectivity alterations assessed by combining fMRI and MR-compatible hand robots in chronic stroke. NeuroImage 47: T90-T97.
- Murase N, Duque J, Mazzocchio R, Cohen LG (2004) Influence of interhemispheric interactions on motor function in chronic stroke. Ann Neurol 55(3): 400-409.
- O’Sullivan M, Morris RG, Huckstep B, Jones DK, Williams SC, et al. (2004) Diffusion tensor MRI correlates with executive dysfunction in patients with ischaemic leukoaraiosis. J Neurol Neurosurg Psychiatry 75(3): 441-447.
- Rosso C, Valabregue R, Attal Y, Vargas P, Gaudron M, et al. (2013) Contribution of corticospinal tract and functional connectivity in hand motor impairment after stroke. PloS One 8(9): e73164.
- Siegel JS, Ramsey LE, Snyder AZ, Metcalf NV, Chacko RV, et al. (2016) Disruptions of network connectivity predict impairment in multiple behavioral domains after stroke. Proc Natl Acad 113(30): E4367-E4376.
- Zemke AC, Heagerty PJ, Lee C, Cramer SC (2003) Motor cortex organization after stroke is related to side of stroke and level of recovery. Stroke 34(5): e23-e26.
- Hayward KS, Schmidt J, Lohse KR, Peters S, Bernhardt J, et al. (2017) Are we armed with the right data? Pooled individual data review of biomarkers in people with severe upper limb impairment after stroke. NeuroImage Clin 13: 310-319.
- Talelli P, Greenwood RJ, Rothwell JC (2006) Arm function after stroke: neurophysiological correlates and recovery mechanisms assessed by transcranial magnetic stimulation. Clin Neurophysiol 117(8): 1641-1659.
- Bembenek JP, Kurczych K, Karliński M, Członkowska A (2012) The prognostic value of motor-evoked potentials in motor recovery and functional outcome after stroke-a systematic review of the literature. Funct Neurol 27(2): 79-84.
- Ito KL, Liew SL (2016) Calculating the laterality index using FSL for stroke neuroimaging data. Giga Science 5(1): s13742-016.
- Coombes SA, Janelle CM, Cauraugh JH (2009) chronic stroke and aging: The impact of acoustic stimulus intensity on fractionated reaction time. Neurosci Lett 452(2): 151-155.

 Review Article
Review Article