ABSTRACT
Annotation: Up to 20-25% of women with PCOS are resistant to clomiphene citrate.
Aim: The study is to optimize the baseline ovarian response in clomiphene-resistant
women to ovulation induction with minimal drug costs.
Materials and Methods of Research: The study included 40 clomiphene-resistant
women with PCOS. For women in group I, we used clomiphene citrate 100mg+
recombinant follicle-stimulating hormone p FSH 37.5 IU / day. Group II received only
p FSH 37.5 using a low dose escalating protocol. The use of this protocol enables
monofollicular growth and a decrease in the risk of multiple pregnancies and, in turn, is
the prevention of ovarian hyper stimulation.
Results: The study showed that in group I compared with group II, the frequency of
ovulation was significantly higher (60% versus 35%).
Conclusion: The combined administration of CC + rFSH in clomiphene-resistant
women with PCOS compared to the use of rFSH alone, gives higher ovulation rates and
lower financial costs.
Keywords: Clomiphene Citrate (CC); Recombinant Follicle-Stimulating Hormone (rFSH); Ovulation Stimulation
Introduction
It has been proven that the induction of ovulation is the main method of treatment in infertile women with PCOS [1-3]. According to the WHO, from 10 to 15% of married couples suffer from infertility. In the conditions of Central Asia, where large families are common and this is traditionally encouraged, childlessness is considered a great misfortune and often leads to family disintegration [4,5]. Up to 20-25% of women with PCOS are resistant to clomiphene citrate [6,7].
Materials and Research Methods
Our randomized trials were carried out in the central polyclinic
of Urgench from 2018 to 2020. It included 40 clomiphene-resistant
women with PCOS. In group I (n = 20) women, we used clomiphene
citrate 100mg + recombinant follicle-stimulating hormone p FSH
37.5 IU / day. Group II (n = 20) received only p FSH 37.5 using a low
dose escalating protocol. Informed written consent was obtained
from all patients. Women were considered clomiphene resistant if
ovulation did not occur when taking CC at a dose of 150 mg / day.
PCOS was diagnosed based on the Rotterdam criteria, in which at
least 2 of the following three criteria were met:
1) Oligo menorrhea (a cycle lasting 35 or more days) and / or
amenorrhea (absence of menstruation for 6 or more months);
2) Hyperandrogenism (defined as a Ferriman-Gallvi index of
more than 8) which is clinically manifested by acne / hirsutism and / or biochemical - the determination of testosterone in the
blood serum of more than 0.7ng / mg;
3) Sonographic manifestations of polycystic ovary: if the ovary
contains 12 or more follicles with a diameter of 2 to 9 mm and
/ or the volume of the ovaries is more than 10 ml.
Inclusion criteria are, clomiphene citrate resistant women
with PCOS aged 20 to 38 years, BMI, no previous ovulation
induction, partners with normal sperm counts according to
WHO standards, opening of the fallopian tubes (confirmed
by hysterosalpingography in the previous 6 months), without
presence operations on the genitals. The exclusion criterion is the
presence of any factors of infertility, except for CV-resistant women
with PCOS. The study also included the measurement of blood
pressure, abdominal circumference, hormonal study of the serum
of patients such as basal FSH, LH / FSH ratio, free testosterone (T),
insulin, progesterone, AMG on the 3rd day of the menstrual cycle.
HDL High Density Lipoproteins, serum estradiol was determined
on the day of ovulation trigger administration. Insulin resistance
(HOMA-IR) was determined as follows: HOMA-IR = fasting insulin
(IU / ml) x fasting glucose (mol / l) / 22.5. Ultrasound of the ovaries
with a transvaginal sensor on the 2nd - 3rd day of the menstrual
cycle to assess the number of antral follicles with a diameter of 2
to 9 mm (in an amount of 12 or more is considered polycystic) and
an assessment of the volume of the ovary, which is determined by
measuring three perpendicularly directed ovarian diameters and
applying the formula: D1xD2xD3x0.5236.
Results of the Study
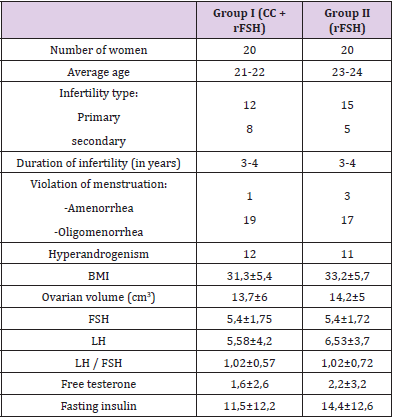
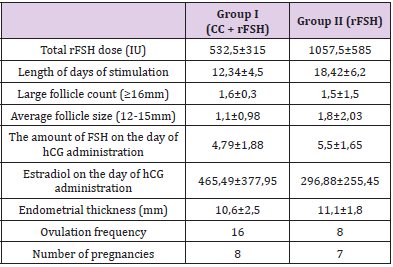
(Table 1) shows the results of clinical and laboratory studies of both groups, which reflects the average age of a woman, type of infertility, BMI, abdominal circumference, ovarian volume, type of menstruation disorder, hormonal and biochemical studies (Table 1). As our study showed, group I (CC + rFSH) received a lower dose of rFSH (532.5 ± 315) and the duration of stimulation days (12.34 ± 4.5) was less than in group II (18.42 ± 6.2 days of stimulation). The number of growth of the middle and dominant follicle, the thickness of the endometrium, the number of ovulations and the frequency of pregnancy are shown in (Table 2). The study showed that the dose of gonadotropin preparations for obtaining ovulation can be reduced by the simultaneous administration of CC + rFSH.
Conclusion
The combined administration of CC + rFSH in clomipheneresistant women with PCOS compared to the use of rFSH alone, gives higher ovulation rates and lower financial costs. The use of this protocol enables monofollicular growth and a decrease in the risk of multiple pregnancies and, in turn, is the prevention of ovarian hyper stimulation.
References
- Dobrokhotova YuE, Matrizayeva GD, Kurbaniyazova MZ (2020) Various methods of ovarian stimulation as a solution to the problem of endocrine infertility. Евразийский вестник педиатрии 2(5): 43-49.
- Kurbaniyazova MZ, Matrizayeva GD. Ovarian hyper stimulation syndrome as a complication of ovulation induction. Central Asian Journal of Pediatrics 2(2): 123-128.
- Kurbaniyazova Madina Zafarjanovna, Bekbauliyeva Gulistan Niyetbayevna (2021) OPTIMIZATION OF OVULATION INDUCTION METHODS IN INFERTIL WOMEN WITH PCOS. Euro-Asia Conferences 1(1): 158-162.
- Bart CJM Fauser, BC Tarlatzis, RW Rebar (2012) Amsterdam ESHRE / ASRM - Sponsored 3rd PCOS Consensus Working Group Consensus on Women's Health Aspects in Polycystic Ovary Syndrome (PCOS). Buzz Reprod 27: 14-24.
- Курбаниязова М, Сапарбаева НР (2018) Оптимизация индукции овуляции у инфертильных женщин с эндокринным бесплодием. Проблема биологии и медицины 4(104): 68-70.
- Balen A (2007) Ovulation induction strategies in the treatment of anovulatory polycystic ovary syndrome. In: Azziz R (Edt.)., Polycystic ovary syndrome: Current understanding of pathogenesis and clinical care. Springer Science, Business Media LLC, pp. 99-112.
- Balasz J, Fabregues F, Creus M, Kasamityana R, Puerto B, et al. (2000) Recombinant human follicle-stimulating hormone for ovulation induction in polycystic ovary syndrome: A prospective randomized trial of two initial doses with chronic dose escalation protocol. J Assist Reprod Genet 17(10): 561-566.

 Short Communication
Short Communication