ABSTRACT
Background: Mud therapy is a simple and effective treatment of several human disorders. The term “fango,” which originated in Italy, means a special kind of mud and its application is named as fangotherapy. A topper with fango molecule structure of natural mineral in its textile fibers was tested in study with cultivated organ-specific cells for its beneficial health effects on the lines of fango.
Materials and Methods: In the present study, current cell biological test methods
were used to investigate whether the PHYSIO NIGHT DELUXE topper with fango
molecule structure is able
i. to compensate an excess of endogenously generated radicals and
ii. to stimulate cell regeneration.
Human promyelocytes (cell line HL-60) were differentiated to functional neutrophils
in the presence of pieces of the topper material and superoxide anion generation was
measured in the course of an induced oxidative burst. Connective tissue fibroblasts
(cell line L-929) were used to examine cell regeneration by closing a cell-free space due
to migration and proliferation in the presence of the topper material. Untreated cell
cultures of both cell lines from the same basic cultures served as corresponding controls.
Results: Exposure of HL-60 cells to the topper for six days during their differentiation process to functional neutrophils resulted in a marked and statistically significant reduction of endogenously generated superoxide anion radicals by the cells. Regeneration of connective tissue fibroblasts showed a significant stimulation of the colonization of the cell-free space due to the exposure to the topper when compared to the untreated control.
Conclusion: Both effects can work synergistically and might have a beneficial effect on inflammatory reactions or cell regenerative processes after excessive stress or physical burden or even overload. Thus, the PHYSIO NIGHT DELUXE topper with fango molecule structure might be able to maintain and improve individual health and wellbeing.
Keywords: Fango; Mineral mud; Oxidative stress; Inflammation; Cell regeneration; Wound healing; Connective tissue fibroblasts; Functional neutrophils; HL-60; L-929; Cell culture
Introduction
Basically, mud therapy is a simple and effective treatment of several disorders of neurological, rheumatologically, cardiovascular, gynecological and inflammatory origin [1,2]. The term “fango,” which originated in Italy, means a special kind of mud deposited from the thermal springs of sulfur bearing sulfuro or sulfated water [3] and its application is named as fangotherapy. In general, fango holds heat and is useful as a thermal application for chronic health conditions [4]. It also stimulates circulation and lymph flow, supports detoxification and helps the body to relax. Some types of fango have anti-inflammatory and pain relieving properties that make them useful for soft tissue injury [5]. The topper with fango molecule structure as tested in this study was primarily designed that the textile fibers of the topper correspond to the molecular structure of natural mineral mud and can, therefore, reflect infrared waves from the body. Thus, the topper might possess the beneficial health effects of fango. According to the manufacturer, many users report amazing improvements in vitality, health and well-being.In the present study, current cell biological test methods were used to investigate whether the topper might be able to inactivate an excess of endogenously generated radicals and thus prevent oxidative stress. Local oxidative stress in the tissue plays an important role in inflammatory reactions [6-9], in the course of wound healing [10,11] or after physical exercise or even overload [12-14]. Therefore, it was also examined whether the topper has a direct beneficial effect on the cell regeneration/wound healing process.
Material and Methods
Topper with Fango Molecule Structure
The company Physio Night GmbH from D-85134 Stammham, Germany, states that the textile fibers of the PHYSIO NIGHT DELUXE topper with fango molecule structure correspond to the molecular structure of natural mineral mud and can therefore reflect infrared waves from the body. In addition, the topper is complemented all around with a mix of stone pine and new wool. According to the company, a renowned textile manufacturer from Italy has succeeded with the help of nanotechnology in integrating the molecular structure responsible for the beneficial effects of fango into the cotton textile of the topper.
Basic Cell Culture
Human promyelocytes (cell line HL-60; ACC-3; ECACC 98070106; Leibniz-Institute; DSMZ German Collection for Microorganisms and Cell Cultures, Braunschweig, Germany) were cultivated for at least 10 passages upon receipt from the cell culture collection and were taken for the experiments over a period of several weeks to ensure independent experiments. Cells were routinely cultivated in RPMI 1640 medium supplemented with 10 % growth supplement and 0.5 % gentamycin. The non-adherent cells were routinely cultivated as suspended mass cultures in special culture flasks with a ventilated lid (25 cm2 growth area; TPP, Switzerland) and were routinely subculture twice a week with fresh culture medium. Cultures were incubated at 37 °C in an atmosphere of 5 % CO2 and 95 % air at almost 100 % humidity. By addition of 1.5 % dimethyl sulfoxide to the culture medium, cells were differentiated over a period of 6 days into functional neutrophils, which are capable of generating superoxide anion radicals in the course of an oxidative or respiratory burst after addition of phorbol- 12-myristate-13-acetate (Sigma-Aldrich, Deisenhofen, Germany) [15-19]. Connective tissue fibroblasts (cell line L-929; ACC-2; Leibniz Institute DSMZ - German Collection of Microorganisms and Cell Cultures, Braunschweig, Germany) were cultivated for at least 20 passages upon receipt from the cell culture collection and were taken for the experiments over a period of several weeks to ensure independent experiments. Cells were routinely grown in RPMI 1640 with 10 % growth supplement and 0.5 % gentamycin and incubated in an incubator at 37 °C in a humidified atmosphere of 5 % CO2 and 95 % air. All cell culture reagents were purchased from Pan-Biotech, Aiden Bach, Germany.
Anti-Inflammatory Effect (Functional Neutrophils)
For the entire duration of the differentiation process, cells were grown in culture flasks, which were placed on pieces of the topper (about 5 × 5 cm in size) and were shielded in several layers of aluminum foil to avoid any unwanted interactions between the exposed cells and the unexposed control cultures... Untreated control cells from the same basic culture were cultivated under identical conditions, but without the topper, and also shielded with multilayers of aluminum foil. Finally, the cells of each experimental series were prepared by two centrifugation steps (6 min at 190 × g) and repeated washings in phosphate buffered saline with calcium and magnesium. Aliquots of the suspended cells in phosphate buffered saline with calcium and magnesium containing 10 mm glucose were taken for the tests and induced to undergo an oxidative burst by the addition of phorbol-12-myristate-13-acetate to the reaction mixture. The reactive superoxide anion radicals in the reaction mixture caused the cleavage of the tetrazolium dye WST-1 (Roche Diagnostics, Mannheim, Germany), which was also present in the reaction mixture. The amount of superoxide anion radicals present in the reaction mixture was directly related to the cleavage and color change of the dye. The color change was recorded at various selected time points up to 40 min as a differential measurement ΔOD = 450 – 690 nm by an Elisa reader (Biotech Elks 808 with software Gen 5 version 3.00) and calculated with Microsoft Excel 2016. Three independent experiments (n = 3) with duplicate wells for each experiment were conducted.
Cell Regeneration/Wound Healing (Connective Tissue Fibroblasts)
Cells were seeded at a density of 100,000 cells/ml into the four cell culture compartments of silicone Culture-Insert 4 Well (ibidi, Gräfelfing, Germany). The four compartments are separated by a 500 μm thick silicone wall. Due to their especially designed surface, the inserts stick firmly to the cell culture dish and completely prevent any cell attachment and growth under their silicone walls. When the silicone frame is removed, a cell-free space (artificial wound) with sharp edges is left. The cell-free space is then closed by cell migration and proliferation. 48 h after seeding, the cells have become confluent and the silicone frames were removed. After placing the pieces of the topper (about 5 × 5 cm in size) below the cell cultures and shielding them in several layers of aluminum foil, the cells were allowed to migrate and proliferate into the cell-free space for 21 hours. Untreated control cells from the same basic culture were treated and cultivated at identical conditions, but without the topper, and also shielded with multi-layer aluminum foil. Finally, cells were fixed with methanol and stained with a Giemsa’s azur eosin methylene blue solution (Merck, Darmstadt, Germany), and air-dried. The width of the remaining wound space was measured at a minimum of six different areas. Three independent experiments were conducted.
Statistical Analysis
Statistical analysis was done by using the non-parametric, twotailed Wilcoxon-Mann-Whitney test.
Results
Anti-Inflammatory Effect
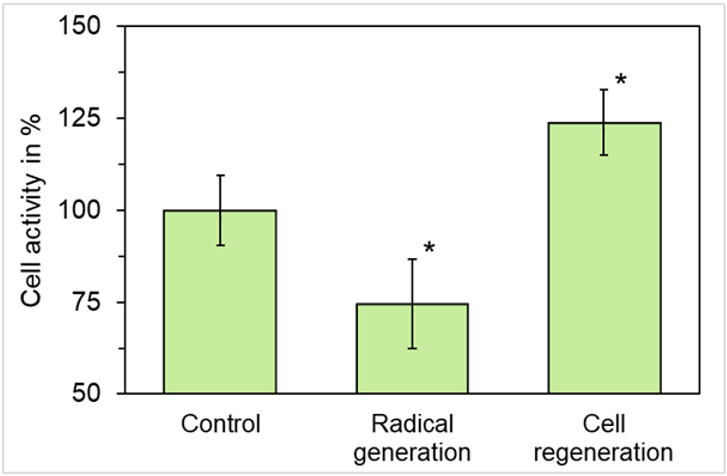
Exposure of HL-60 cells to the topper for six days during the differentiation process resulted in a reduced generation of endogenous radicals after stimulation (Figure 1). The radical generation in the exposed cell cultures was reduced by 22.6 ± 12.2% (mean value ± standard deviation, n = 3). This reduction was statistically significant (p ≤ 0.05).
Cell Regeneration/Wound Healing
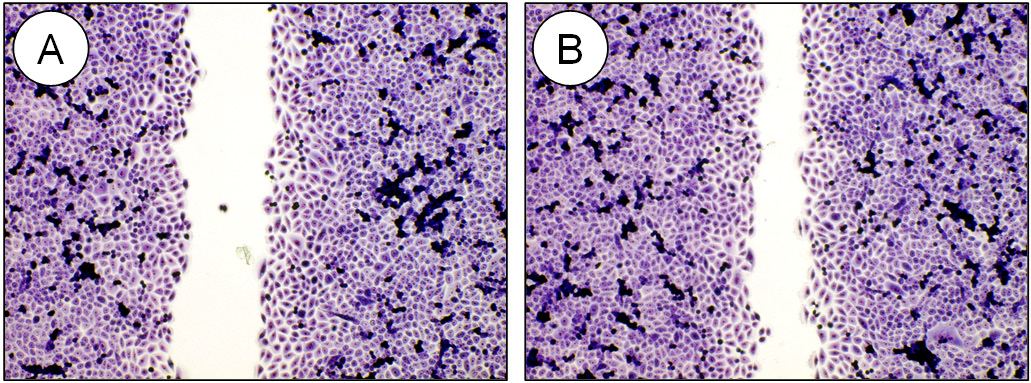
The microscopic examination of the fixed and stained samples of cell regeneration showed at first sight a clear stimulation of the colonization of the cell-free space by exposure to the topper when compared to the untreated control (Fig. 2). The quantification documented a statistically significant improvement by the topper by 23.9 ± 8.8 % (mean value ± standard deviation; Fig. 1) in comparison to the untreated control (p ≤ 0.05).
Figure 1: Effect of the PHYSIO NIGHT DELUXE topper with fango molecule structure on the generation of superoxide anion radicals of functional neutrophils and the regeneration of connective tissue cells. Corresponding controls are set as “100 %. Note that radical generation is decreased, and cell regeneration is increased. Data represent mean values ± standard deviation of 3 independent experiments. *p ≤ 0.05 vs. control; non-parametric, two-tailed Wilcoxon-Mann-Whitney test.
Figure 2: Representative micrographs of fixed and stained cell cultures demonstrating regeneration and wound closure within 21 hours for the untreated control (A) in comparison to the culture which was treated with the PHYSIO NIGHT DELUXE topper (B). Note the significantly reduced cell-free space after treatment in (B). Olympus IX-50 inverted microscope equipped with an Olympus Planachromate 10x and an Olympus E-10 digital camera with 4-megapixel resolution at bright field illumination.
Discussion
Cell cultures are frequently used for a better understanding of the mechanisms that underlie cell activity in vivo. This includes differentiation, migration, proliferation, and metabolism. For over a century, two-dimensional cell cultures as used in this present study, have been used as in vitro models and are well accepted in preclinical research. However, one might argue that cell cultures do not represent the human body with its complex metabolic pathways and facets, but it should be considered that cell cultures can shed light on selected aspects of living matter and are a useful tool to reduce or even avoid animal experiments. Neutrophils are the most abundant type of granulocytes which make up about 60 % of all white blood cells in humans and are normally found in the bloodstream. They form an essential part of the innate immune system [20] and, therefore, play a key role in the front-line defense against invading microbial pathogens. However, neutrophils are also one of the first responders of inflammation and migrate from the blood into the inflamed tissue and generate highly reactive superoxide anion radicals in the course of an oxidative or respiratory burst [21]. The test results of this study indicate that the topper can have a direct effect on inflammatory processes in vivo. The generation of unwanted reactive oxygen radicals is reduced, so that a local oxidative stress in the tissue is also reduced. This can have a positive effect on the healing process of the inflamed tissue after occurrence of local oxidative stress. Synergistically to the first finding is the beneficial effect of the topper on cell regeneration. In this present test system, especially the granulation phase is simulated which is characterized by migration and proliferation of the cells in order to close the cell-free space (= wounded area) in vivo. However, a complicated wound healing process is also related to local oxidative stress [22-24] which is also reduced by the topper. Taken together, both effects can work synergistically and have a beneficial effect on inflammatory reactions or cell regenerative processes after excessive stress or physical burden or even overload. Thus, the PHYSIO NIGHT DELUXE topper might be able to maintain and improve individual health and well-being.
References
- Patel N, Raiyani D, Kushwah N, Parmar P, Hapani H, et al. (2015) An introduction to mud therapy: A review. Int J Pharm Therapeutics 6(4): 227-231.
- Nieder R, Benbi DK, Reichl FX (2018) Soil Components and Human Health. Springer Science + Business Media BV Dordrecht the Netherlands.
- 3 Ziemska J, Szynal T, Mazańska M, Solecka J (2019) Natural medicinal resources and their therapeutic applications. Rocz Panstw Zakl Hig 70(4): 407-413.
- 4 Galzigna L, Ceschi-Berrini C, Moschin E, Tolomio C (1998) Thermal mudpack as an anti-inflammatory treatment. Biomed Pharmacother 52(9): 408-409.
- 5 Chadzopulu A, Adraniotis J, Theodosopoulou E (2011) The therapeutic effects of mud. Progr Health Sci 1(2): 132-136.
- Nathan C (2002) Points of control in inflammation. Nature 420(6917): 846-852.
- Nathan C, Ding A (2010) Nonresolving inflammation. Cell 140(6): 871-882.
- Pawelec G, Goldeck D, Derhovanessian E (2014) Inflammation, ageing and chronic disease. Curr Opinion Immunol 29: 23-28.
- Kotas ME, Medzhitov R (2015) Homeostasis, inflammation, and disease susceptibility. Cell 160(5): 816-827.
- Clark RAF (1996) Wound repair: overview and general considerations. In: The Molecular and Cellular Biology of Wound Repair, Clark RAF (2nd). Plenum Press, New York, NY, p. 3-50.
- Keller auf dem U, Kümin A, Braun S, Werner S (2006) Reactive oxygen species and their detoxification in healing skin wounds. J Investigat Dermatol Symp Proc 11(1): 106-111.
- Apor P, Rádi A (2006) Physical exercise, oxidative stress and damage. Orvosi Hetilap 147(22): 1025-1031.
- Vollaard NB, Cooper CE, Shearman JP (2006) Exercise-induced oxidative stress in overload training and tapering. Med Sci Sports Exercise 38(7): 1335-1341.
- Larsen MK, Matchkov VV (2016) Hypertension and physical exercise: The role of oxidative stress. Medicina 52(1): 19-27.
- Babior BM (1999) NADPH oxidase: An update. Blood 93(5): 1464-1476.
- Tan AS, Berridge MV (2000) Superoxide produced by activated neutrophils efficiently reduces the tetrazolium salt WST-1 to produce a soluble formazan: a simple colorimetric assay for measuring respiratory burst activation and for screening anti-inflammatory agents. J Immunol Meth 238: 59-68.
- Teufelhofer O, Weiss RM, Parzefall W, Schulte-Hermann R, Micksche M, et al. (2003) Promyelocytic HL60 cells express NADPH oxidase and are excellent targets in a rapid spectrophotometric microplate assay for extracellular superoxide. Toxicol Sci 76(2): 376-383.
- Droge W (2002) Free radicals in the physiological control of cell function. Physiol Rev 82(1): 47-95.
- Dartsch PC (2006) TIIOS - A sensitive and cell-based test assay for the screening of biologically active substances for their antioxidant potential. Innov Food Technol 32: 72-75.
- Witko-Sarsat V, Rieu P, Descamps-Latscha B, Lesavre P, Halbwachs-Mecarelli L (2000) Neutrophils: molecules, functions and pathophysiological aspects. Lab Invest 80: 617-653.
- Ward PA (1999) the acute inflammatory response and its regulation. Arch Surg 134(6): 666-669.
- Cano Sanchez M, Lancel S, Boulanger, E, Neviere R (2018) Targeting oxidative stress and mitochondrial dysfunction in the treatment of impaired wound healing: A systematic review. Antioxidants 7(8): 98.
- Schafer M, Werner S (2008) Oxidative stress in normal and impaired wound repair. Pharmacol Res 58(2): 165-171.
- Dunnill C, Patton T, Brennan J, Barrett J, Dryden M, et al. (2017) Reactive oxygen species (ROS) and wound healing: The functional role of ROS and emerging ROS-modulating technologies for augmentation of the healing process. Int Wound J 14(1): 89-96.

 Research Article
Research Article