ABSTRACT
Background: Fibromyalgia syndrome (FMS) is an extra-articular rheumatologic
disease with an estimated prevalence of 2–4%, whose etiology and pathogenesis are
still unknown. Oxidative stress is an imbalance between products of oxidation and
antioxidant defenses and plays a critical role in the etiopathogenesis of FMS. Lidocaine is
a local anesthetic with rapid onset of action, safety profile, low cost and wide availability
that can reduce nerve signal conduction and consequently attenuate pain. Intravenous
repetitive injections have been used to manage chronic pain.
Purpose: This study aimed to investigate the effects of repetitive intravenous
lidocaine injections associated with pregabalin on oxidative and antioxidant parameters
in women with FMS.
Methods: Oxidative stress was determined by measuring the levels of GSH,
lipoperoxidation, catalase, and superoxide dismutase in the blood of 48 female patients
with FMS who were treated with lidocaine or saline. The FIQ and VAS valued clinical
parameters of FMS as instruments of valuation of pain.
Results: Regarding the Visual Analog Scale (VAS) and Fibromyalgia Impact
Questionnaire (FIQ) scores, patients treated with lidocaine/pregabalin (GPL) revealed
improvements in the parameter analyzed compared to the initial and final treatment
time. Moreover, lidocaine administration significantly increased GSH levels (p<0.001)
and non-enzymatic capacity (FRAP, p<0.01) in blood and plasma and decreased
lipoperoxidation in blood cells without altering sulfhydryl groups comparing the first
and second times of treatment.
Conclusion: Lidocaine plus pregabalin significantly stimulated antioxidant defenses
and reduced oxidative stress.
Keywords: Lidocaine; Glutathione; Oxidative stress; Fibromyalgia; Pain
Introduction
Fibromyalgia syndrome (FMS) is a clinically well-defined
disease of extra-articular rheumatologic origin with an estimated
2-4% prevalence. It is the most found chronic musculoskeletal
pain condition, and its etiology is still unknown [1]. Its symptoms
also include non-recoverable sleep patterns, cognitive dysfunction,
headache, morning stiffness, fatigue, depression, and anxiety [2].
Oxidative stress is an imbalance between oxidation products and
antioxidant defenses and has recently been associated with several events in the pathogenesis of FMS [3,4]. Patients with FMS have
decreased levels of glutathione (GSH) and increased lipoperoxidation
in blood and plasma, which seems to be associated with a worsening
of patients’ clinical condition [5]. The pathology of fibromyalgia
(FM) causes a negative regulation of catalase activity in patients’
erythrocytes and leukocytes [6,7]. Research has also described the
effects of antioxidant redox systems, levels of protein carbonylation
and lipoperoxidation on the pathogenesis of FMS, and increased
oxidative stress is strongly associated with the FMS severity [5].
Thus, managing the oxidative profile can be a promising approach
to optimize the FMS treatment, although there is a little-explored
gap in this profile. There is still no specific pharmacological therapy
to date to relieve such syndrome, and the currently available drugs
are used to manage symptoms. Although there are drugs that can
treat FMS symptoms, such as neuromodulators, antidepressants,
and muscle relaxants, some side effects and their low efficacy
have been reported to limit therapeutic adherence. Lidocaine is an
aminamide-type local anesthetic with fast onset, safety profile, low
cost, and wide availability. It inhibits neuron-dependent voltagegated
sodium channels, reducing the nervous signal conduction
and consequently blocking pain. Intravenous lidocaine showed
positive results in treating acute and chronic neuropathic pain
syndromes, such as trigeminal neuralgia and peripheral nerve
damage. However, the role of intravenous lidocaine injection in FMS
needs to be clarified.
Pregabalin was the first drug approved by the Food and Drug
Administration (FDA) to treat FMS. Its structure is similar to the
gamma-aminobutyric acid (GABA) neurotransmitter, without
pharmacological action in this way, but in voltage-dependent
calcium channels [8]. Few studies have reported using lidocaine
with pregabalin to manage FM, and we have not found reports of
studies valuating the oxidation parameters of FM patients with
this treatment. Therefore, the hypothesis that there could be an
effect on oxidative stress allowed us to assess its correlation with
oxidative symptoms and parameters.
Methodology
Ethical Aspects
The Ethics Committee of the Federal University of Sergipe (UFS) approved the clinical study opinion No. 2.637.928 (Certificate of Presentation for Ethical Consideration (CAAE) protocol No. 85503418.2.0000.5546). All subjects who volunteered for the trial were women included in the study only after signing the free and informed consent form. The Universal Trial Number (UTN) is U1111-1257-3477. ReBEC trial: (req:10410) Benefits of innovative treatment for women with fibromyalgia: a procedure to follow.
Experimental Groups and Schematic Design of Experiments
Forty-eight female patients who met the 2016 American
College of Rheumatology (ACR) FMS classification criteria were
enrolled. The inclusion/exclusion criteria adopted in the study
excluded patients with other comorbidities such as epilepsy,
recent trauma (≤ 3 months), psychiatric and rheumatic disorders,
moderate or severe neuromuscular disorders, hypothyroidism
or hyperthyroidism, infectious arthroplasty, other chronic pain
syndromes, hypersensitivity to drugs, and neoplasms disorders.
Twenty-four patients formed two groups randomly.
Pregabalin Group (GP): Women taking 150 mg pregabalin
daily and submitted to a hospital procedure performed at
the surgical center for an intravenous administration of 0.9%
physiological solution for three consecutive weeks.
Pregabalin/lidocaine Group (GPL): Women taking 150 mg
pregabalin daily and submitted to a hospital procedure performed
at the surgical center for intravenous administration of lidocaine (3
mg.kg) for three consecutive weeks.
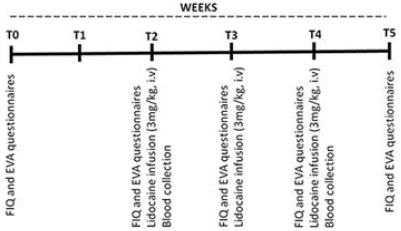
FM patients valuated for five weeks. The experimental
procedure schematic design shown in Figure 1. The lidocaine
infusion was applied at the third (T2), fourth (T3), and fifth (T4)
weeks. The FIQ and VAS questionnaires were performed at the first
(T0), second (T1), third (T2), fourth (T3), and fifth (T4) weeks.
All patients took pregabalin (150 mg.kg) for treatment as the
therapeutic protocol of the HU-UFS outpatient clinic. Thus, since
our research ethics committee did not authorize an experimental
group of fibromyalgia patients treated with lidocaine alone, they
used the previous pregabalin because it is a pain clinic, which is a
limitation of this study Table 1 and Table 2.
Table 1: Demographic features of patients.
Note: BMI, body mass index; GPL, pregabalin/lidocaine group; GP, pregabalin group; R, Rank-Biserial Correlation. Values are expressed in means ± SD. Mann-Whitney Test.
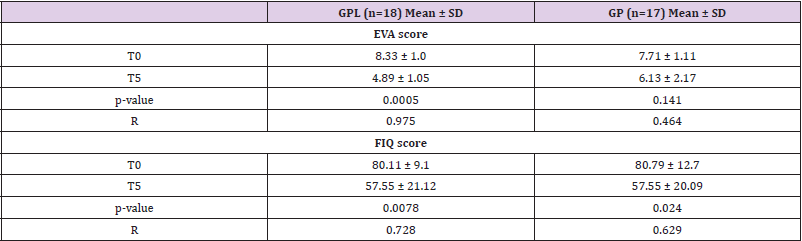
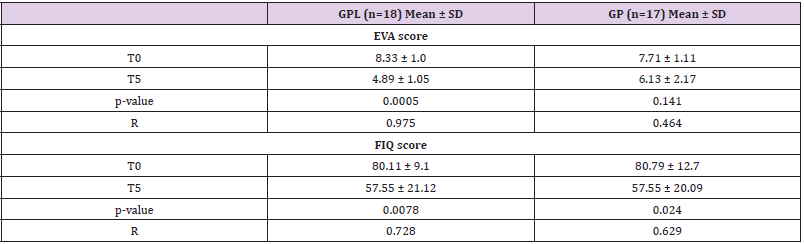
Table 2: Intensity of resting pain (EVA score) and Impact of Fibromyalgia (FIQ score) determined in initial time (T0) and final time (T5).
Note: BMI, body mass index; GPL, pregabalin/lidocaine group; GP, pregabalin group; R, Rank-Biserial Correlation; T0, initial time; T5, final time. Values are expressed in means ± SD. Mann-Whitney Test
Fibromyalgia Impact Questionnaire (FIQ score)
The FMS functional status was valuated by the FM impact questionnaire (FIQ). The questions aimed to highlight well-being and the loss of daily work, difficulty to work, fatigue, pain, and well-being in the morning after waking up, stiffness, anxiety, and depression. High scores showed functional limitations [9].
Intensity of Resting Pain (VAS score)
A numerical scale of 11 points measured the intensity of the pain according to how intense patients report the pain to be, in which 0 is considered painless and 10 is the worst pain. The test is an analogic visual scale completed by the patients themselves [10]. The FMS functional status was valuated by the FM impact questionnaire (FIQ). The questions aimed to highlight well-being and the loss of daily work, difficulty in working, fatigue, pain, and well-being in the morning after waking up, stiffness, anxiety, and depression. High scores showed functional limitations [9]. The visual analog scale (VAS) is a subjective measure validated for acute and chronic pain. Patients reported their scores by making a handwritten mark on a 10 cm line representing a continuum between “no pain” and “worst pain”.
Biochemical Measures
Blood Collection and Preparation of Blood Samples
During the second and third trials, corresponding to the second and third weeks, venous blood samples of all individuals were collected in test tubes with no anticoagulant or EDTA. Fresh whole blood samples were separated and used for determined reduced glutathione (GSH) and lipoperoxidation. A portion of EDTA-blood was centrifuged for 10 minutes at 3000 xg to separate plasma and erythrocytes that were then washed three times with PBS and kept frozen at -80°C until analysis for activities of RBC antioxidant enzymes, reduced sulfhydryl groups, and ferric reducing ability of plasma (FRAP).
Ferric-Reducing Ability (FRAP assay)
FRAP level in plasma was measured according to the method by Benzie and Strain, as described previously [11]. The plasma samples were mixed with a reagent mixture containing acetate buffer (pH 3.6), 5 mM tripyridyltriazine in 40 mM HCl, and 20 mM ferric chloride. Absorbance was assessed at a 593 nm wavelength. FRAP values were expressed as μg/mg protein.
Reduced Sulfhydryl Groups and GSH Determination
To measure the levels of reduced thiol (-SH) groups in protein and nonprotein fractions from plasma, an 80 𝜇g sample aliquot reacted with a 10 mM 5,5- dithionitrobis 2-nitrobenzoic acid. After 60 minutes of incubation at room temperature, the absorbance was read in a spectrophotometer set at 412 nm [12].
Lipid Peroxidation Level Determinations
Lipoperoxidation levels in the total blood samples were measured with the thiobarbituric acid reaction according to the method by (Draper & Hadley 1990 [13].
Catalase Activity
Catalase activity was assayed in blood cell lysates by measuring the hydrogen peroxide (H2O2) absorbance decrease ratio in a spectrophotometer at 240 nm [14].
Superoxide Dismutase Activity
Superoxide dismutase (SOD) activity was determined in blood cell lysates from the inhibition of superoxide anion-dependent adrenaline autoxidation in a spectrophotometer at 480 nm as previously described [15].
Statistical Analysis
The results were expressed as a mean ± S.E.M or mean ± S.D. Samples were assessed for normal distribution according to the Shapiro-Wilk test. Differences between the two groups were analyzed using the Mann-Whitney U test for independent samples or the Wilcoxon test for dependent samples. The rank-biserial correlation was used as a side effect. Differences were considered significant if p<0.05. The statistical analyses were made using the GraphPad Prism® 5.0 software (GraphPad Prism Software Inc., San Diego, CA, USA).
Sample Size Calculation
Although a 1:1 proportion for randomization was proposed, the final proportion was 3:2 due to a lack of follow-up. Thus, for a significant difference at the 5% level with 80% power, with a very large effect size (Cohen’s D = 1.2), 25 patients with a 3:2 ratio are necessary, considering a correction of asymptotic relative efficiency for non-parametric tests.
Results
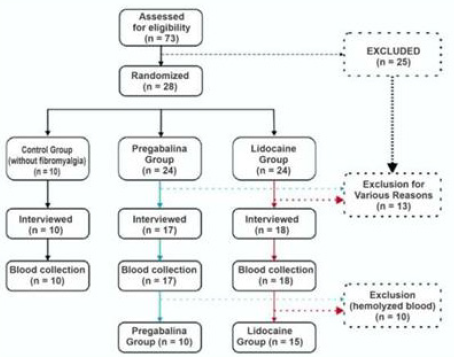
According to the study flowchart, the steps occurred as shown
in Figure 1.
Study flowchart
The description of the patients is reported in Figure 2.
No difference was found in demographic values for patients
with FM treated with pregabalin (GP) or treated with pregabalin/
lidocaine (GPL) (Table 1).
Table 3 Regarding the VAS scores, patients treated with
lidocaine/pregabalin (GPL) showed improvements in the parameter
analyzed compared initial (T0) and final time (T5) of treatment. No
difference was found in patients from the group pregabalin (GP).
Besides, a considerable improvement also was seen in the FIQ score
compared initial (T0) and final (T5) time of vasluation in GPL and
GP groups. No difference was reported between the GPL and GP
groups in VAS, neither the FIQ score in the final time (T5) (Table 2).
Table 4 The effect of repetitive intravenous lidocaine 3 mg.kg.h,
diluted in 250 ml of 0.9% saline through an infusion pump,
administration associated with pregabalin (150 mg.kg) on the
redox status are described in Figures 2 and 3. The blood samples
were collected before first lidocaine administration (at the third
week (T2)) and after the third lidocaine infusion (at the fifth
week (T4)), and the analyses was performed at T2 and T4 in the
same patient from the GPL (lidocaine/pregabalin) and GP groups
(pregabalin) (Figure 1).
Table 3: Demographic features of patients.
Note: BMI: Body mass index; GPL: Pregabalin/lidocaine group; GP: Pregabalin group; R: Rank-biserial correlation. Values are expressed in means ± SD. Mann-Whitney U Test.
Table 4.
Note: BMI: Body mass index; GPL: Pregabalin/lidocaine group; GP: Pregabalin group; R: Rank-biserial correlation; T0: Initial time; T5: Final time. Values are expressed in means ± SD. Mann-Whitney U Test.
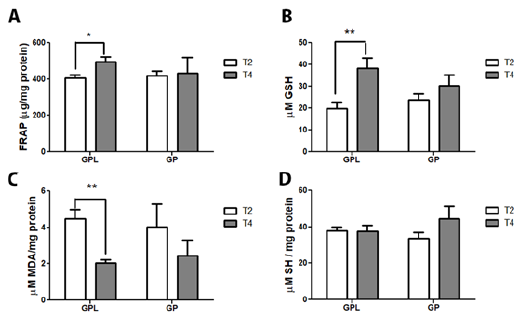
Figure 2 represents the oxidative damage and non-enzymatic
antioxidant profile. The results showed that lidocaine/pregabalin
administration attenuates the redox damage caused by FM. We
found that lidocaine/pregabalin increased GSH levels (p<0.01)
and non-enzymatic capacity (FRAP) (p<0.05) in blood and plasma
of the GPL group (Figure 2A & 2B). Moreover, lidocaine decreases
lipoperoxidation (p<0.001) in blood cells of the GPL group (Figure
2C) without changing the sulfhydryl groups (Figure 2 D). No
changes were found in the GP group (pregabalin) between T2 and
T3 in oxidative damage and non-enzymatic antioxidant status.
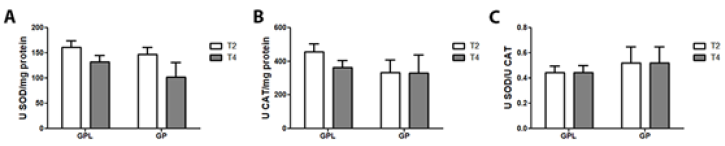
Figure 3 describes the effect of lidocaine/pregabalin
administration on enzymatic antioxidant defense catalase and
superoxide dismutase. Lidocaine/pregabalin could not alter
enzymatic antioxidant activities in blood cells in both the GPL and
GP groups (Figure 3).
Figure 4 Mild headache, dizziness, and drowsiness have been
reported.
BMI: Body mass index; GPL: Pregabalin/lidocaine group; GP: Pregabalin group; R: Rank-biserial correlation. Values are expressed in means ± SD. Mann-Whitney U Test.
Figure 3: Effect of repetitive intravenous injection of lidocaine (3 mg.kg IV) on parameters of oxidative imbalance in blood cells and plasma of FM patients. Ferric-reducing ability (FRAP) (A); GSH levels (B); Thiobarbituric acid reactive substance (TBARS) (C); Total reduced thiol content (SH) (D). Data are reported as mean ± SEM of 15 patients (GPL group) and ten patients (GP group). Statistically significant differences from T2 (before lidocaine injection) and T4 (after third lidocaine injection) as determined by the Wilcoxon test. GPL: Pregabalin/lidocaine group; GP: Pregabalin group.
Figure 4: Effect of repetitive intravenous injection of lidocaine (3 mg/kg IV) on enzymatic antioxidant defenses in FM patients’
blood cells. Superoxide dismutase activity (A); Catalase activity (B); SOD and CAT ratio (C). Data are reported as mean ± SEM
of 15 patients (GPL group) and ten patients (GP group).
Statistically from T2 (before lidocaine injection) and T4 (after third lidocaine injection) as determined by the Wilcoxon test.
GPL: Pregabalin/lidocaine group; GP: Pregabalin group.
Discussion
This study investigated the beneficial effects of repetitive
intravenous injection of lidocaine associated with pregabalin on
FM and the role of oxidative stress in the lidocaine/pregabalin
pharmacological action. There were significantly improved VAS
and FIQ scores in the GPL group (lidocaine/pregabalin). Moreover,
to the best of our knowledge, our findings also showed for the
first time that lidocaine/pregabalin attenuated the oxidative
stress caused by FM, especially by regulating the non-enzymatic
antioxidants defense. Lidocaine is a classical local anesthetic
and antiarrhythmic drug that changes neuron depolarization by
blocking the fast voltage-gated sodium (Na+) channels. It is also
used as an analgesic for several painful conditions [16]. Nowadays,
several works have described the effect of lidocaine in attenuating
fibromyalgia symptoms. Intravenous lidocaine administration
inhibits the pain caused by a deep ischemic but not the superficial
cutaneous pain modalities or tactile sensation [17]. Also, repeated
lidocaine injections into myofascial points attenuated clinical FM
pain and several tender points [8,17], and a single administration of
lidocaine into the trapezius muscle reduced secondary hyperalgesia
in FM patients [18]. Our results showed that intravenous injections of lidocaine associated with pregabalin (orally) reduced the pain,
which is supported by the VAS and FIQ scores. Chronic pain is one
of FMS’s main symptoms and modulating that parameter could
lead to a better quality of life. Moreover, our findings also showed
improved functional and physical capacity and mental health status
assessed by the FIQ score in the pregabalin/lidocaine group.
Thus, administering lidocaine/pregabalin modulates morning
tiredness, stiffness, anxiety, pain, and depression in FM patients,
thus decreasing FMS symptoms [19]. However, no differences were
found between the GPL and GP groups regarding the FIQ scores.
Such finding agrees with a study by Oliveira [20], which showed
that clinical pain was attenuated after lidocaine treatment. The
effect was similar to that of saline administration, which indicated
that the additional factor to overall analgesia could be inferred in
the lidocaine effect. Additionally, the pain parameters improved
in the group treated with lidocaine/pregabalin, reinforcing the
hypothesis that combining those drugs could bring associated
benefits. Studies have associated oxidative stress parameters and
FMS symptoms, showing a strong relationship between redox
status and FIQ scores [21]. Thus, we analyzed the effect of repetitive
intravenous lidocaine administration on the redox status of FM
patients. Lidocaine/pregabalin could improve the total antioxidant
capacity, mainly by upregulating the glutathione (GSH) levels
compared to the GPL and GP groups. The tripeptide GSH contains
the thiol group that protects the organism from oxidative stress by
modulating the enzyme glutathione peroxidase [22]. It could be a
biomarker of improved redox balance in FM patients [23].
Those reduced thiol group levels deteriorate in FM patients
and the thiol-disulfate rate increases in favor of disulfide amounts.
Increased GSH levels seen in GPL patients could contribute to better
physical and psychological response after the lidocaine treatment
measured by the FIQ scores. Moreover, increased antioxidant redox
systems, such as GSH, protect lipids and membranes from oxidative
damage [5,24]. FM includes increased plasma lipoperoxides levels
in blood [25,26], resulting in altered membrane fluidity and proteinlipid
bilayer, altered membrane potentials and eventual integrity
leading to the release of cell organelle contents in extracellular
fluid. Besides, lipid peroxidation plays a key role in the central
nervous system mechanism of depression, anxiety, cognitive
dysfunctions, and pain. All of those symptoms are described in FM
patients, so lipid oxidation protection is an important FM therapy
target [5,24,27].
Together, our data showed in the experiment that lidocaine/
pregabalin therapy stimulate antioxidant defenses in the blood,
reducing oxidative stress and consequently lipid bilayer damage
and cell membrane disruption. Attenuating oxidative injury could
mitigate FM symptoms. These results could propose intravenous
lidocaine injection as a safe treatment that may significantly
improve FMS patient’s quality of life.
Acknowledgments
This study was supported by the Brazilian Agencies National Council for Scientific and Technological Development (CNPq), Coordination for the Improvement of Higher Education Personnel (CAPES), and Foundation to support Research and Technological Innovation of the State of sergipe (FAPITEC-SE).
References
- Cohen H (2017) Controversies and challenges in fibromyalgia: a review and a proposal. Royal National Orthopaedic Hospital, Stanmore, Brockley Hill, Stanmore, Middlesex, HA7 4LP, UK. Ther Adv Musculoskelet Dis 9(5): 115-127.
- Goldenberg DL (1997) Fibromyalgia, chronic fatigue syndrome, and myofascial pain syndrome. National Library Of Medicine. Hospital Newton-Wellesley, Massachusetts. Curr Opin Rheumatol 9(2): 135-143.
- Hudson JI, Goldenberg DL, Pope HGJR, Keck PE JR, schlesinger L (1992) Comorbidity of fibromyalgia with medical and psychiatric disorders. Biological Psychiatry Laboratory, McLean Hospital, Belmont, Massachusetts 02178. Am J Med 92(4): 363-367.
- Wolfe F, Anderson J, Harkness D, Bennett RM, Caro XJ, et al. (1997) Health status and disease severity in fibromyalgia: results of a six-center longitudinal study. Arthritis Research Center, and University of Kansas School of Medicine, Wichita 67214, USA. Arthritis Rheum 40(9): 1571-1579.
- Ghizal F, Das SK, Mahdi AA (2017) Some oxidative and antioxidative parameters and their relationship with clinical symptoms in women with fibromyalgia syndrome. Department of Biochemistry, King George's Medical University, Lucknow, India. Department of Rheumatology, King George's Medical University, Lucknow, India.nt J Rheum Dis 20(1): 39-45.
- Celik F, Gocmez C, Bozkurt M, Kaplan I, Kamasak K, et al. (2013) Neuroprotective effects of carvacrol and pomegranate against methotrexate-induced toxicity in rats. Department of Anesthesiology, Department of Neurosurgery, Department of Physical Medicine and Rehabilitation, Department of Biochemistry, Department of Neurology, Dicle University, Diyarbakir, Turkey. Eur Rev Med Pharmacol Sci 17(22): 2988-2993.
- Rúas J, Capdevila A (2017) Political communication today: challenges and threats. Communication & Society. Universitat Rovira i Virgili, Espanha 30 (3).
- Affaitati G, Costantini R, Fabrizio A, Lapenna D, Tafuri E, et al. (2011) Effects of treatment of peripheral pain generators in fibromyalgia patients. Pathophysiology of Pain Laboratory, Ce.S.I., G. D'Annunzio Foundation, Department of Medicine and Science of Aging, Chieti University, via Carlo de Tocco n. 3, Chieti, Italy. Eur J Pain 15(1): 61-69.
- Bennett RM, Russell J, Cappelleri JC, Bushmakin AG, Zlatvas G, et al. (2010) Identification of symptom and functional domains that fibromyalgia patients would like to see improved: a cluster analysis. Oregon Health & Science University, Portland, Oregon, USA. BMC Musculoskelet Disord.
- Williamson A, Hoggart B (2005) Pain: a review of three commonly used pain rating scales. J Clin Nurs. Research Nurse (Pain), Birmingham Heartland's and Solihull NHS Trust, Birmingham, UK.
- Benzie IF, Strain JJ (1996) The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: the FRAP assay. Department of Health Sciences, Hong Kong Polytechnic University, Kowloon, Hung Hom, Hong Kong. Human Nutrition Research Group, University of Ulster, Coleraine, Northern Ireland, United Kingdom 239(1): 70-76.
- Ellman GL (1959) Tissue sulfhydryl groups. Archives of Biochemistry and Biophysics. From the Biochemical Research Laboratory, The Dow Chemical Company, Midland, Michigan USA 82: 70-77.
- Draper HH, Hadley M (1990) Malondialdehyde determination as index of lipid peroxidation. Meth Enzymol. Methods Enzymol. Academic Press 186: 421-31.
- Aebi H (1984) Catalase in vitro. Methods Enzymol. Methods in Enzymology. Academic Press 105: 121-126.
- Misra HP, Fridovich I (1972) The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. F.rom the Department of Biochemistry, Duke University Medical Center, Durham, Xorth Carolina 27710 J Biol Chem 247(10): 3170-3175.
- Masic D, Liang E, Long C, Sterk EJ, Barbas B, et al. (2018) Intravenous Lidocaine for Acute Pain: A Systematic Review. Loyola University Medical Center, Maywood, Illinois. Pharmacotherapy 38(12): 1250-1259.
- Frölich MA, Mckeown JL, Worrell MJ, Ness TJ (2010) Intravenous lidocaine reduces ischemic pain in healthy volunteers. Department of Anesthesiology, University of Alabama at Birmingham, Birmingham, AL, USA 35(3): 249-254
- Staud R, Weyl EE, Bartley E, Price DD, Robinson ME (2014) Analgesic and anti-hyperalgesic effects of muscle injections with lidocaine or saline in patients with fibromyalgia syndrome. Department of Medicine, University of Florida, Gainesville, USA. Eur J Pain pp. 803-812.
- Simone Azevedo de Zanette, Rafael Vercelino,Gabriela Laste, Joanna Ripoll Rozisky, André Schwertner, et al. (2014) Melatonin analgesia is associated with improvement of the descending endogenous pain-modulating system in fibromyalgia: a phase II, randomized, double-dummy, controlled trial. Pain and Palliative Care Service at the Hospital de Clínicas de Porto Alegre (HCPA), Universidade Federal do Rio Grande do Sul (UFRGS), Porto Alegre, Brazil. Pharmacology Department, Instituto de Ciências Básicas da Saúde, UFRGS, Porto Alegre, Brazil. Post Graduate Program in Medical Sciences, School of Medicine, Universidade Federal do Rio Grande do Sul (UFRGS), Porto Alegre, Brazil BMC Pharmacol Toxicol. 15: 40.
- Oliveira MA, Guimarães AG, Araújo AAS, Quintans-Júnior LJ, Quintans JSS (2017) New drugs or alternative therapy to blurring the symptoms of fibromyalgia-a patent review. Multiuser Center for Health (C Multi-Saúde), Federal University of Sergipe, Sergipe, Brazil. Departament of Health Education, Federal University of Sergipe, Sergipe, Brazil. Expert Opin Ther Pat 27(10): 1147-1157.
- Sarifakioğlu B, Güzelant AY, Güzel EC, Güzel S, Kiziler AR (2014) Effects of 12-week combined exercise therapy on oxidative stress in female fibromyalgia patients. Department of Physical Medicine and Rehabilitation, School of Medicine, Namik Kemal University, 100, Yıl Mah, Barbaros Cad. Merkez, Tekirdağ, Rheumatol Int 34: 1361-1367.
- Dickinson DA, Forman HJ (2002) Cellular glutathione and thiols metabolism. Biochem Pharmacol. Department of Environmental Health Sciences, Center for Free Radical Biology, School of Public Health, University of Alabama at Birmingham, 1530 3rd Avenue S, RPHB-317, Birmingham, AL 35294-0022, USA 64(5-6): 1019-1026.
- La Rubia M, Rus A, Molina F, Del Moral ML (2013) Is fibromyalgia-related oxidative stress implicated in the decline of physical and mental health status? Clin Exp Rheumatol. Department of Experimental Biology, University of Jaén, Campus Las Lagunillas, Jaen, Spain 31(6 Suppl 79): S121-7.
- Iqbal R, Mughal MS, Arshad N, Arshad M (2011) Pathophysiology and antioxidant status of patients with fibromyalgia. Rheumatol Int. Zoology Department, G. C. University, Lahore, 54000, Pakistan 31(2): 149-152.
- Akkuş S, Naziroğlu M, Eriş S, Yalman K, Yilmaz N, et al. (2009) Levels of lipid peroxidation, nitric oxide, and antioxidant vitamins in plasma of patients with fibromyalgia. Medical Faculty, Department of Physical Medicine and Rehabilitation, Suleyman Demirel University, Isparta, Turkey. Cell Biochem Funct 27(4): 181-185.
- Altindag O, Celik H (2006) Total antioxidant capacity and the severity of the pain in patients with fibromyalgia. Department of Physical Medicine and Rehabilitation, Harran University, Sanliurfa, Turkey. Redox Rep 11(3): 131-135.
- Oliveira MA, Guimarães AG, Araújo AAS, Quintans-Júnior LJ, Quintans JSS (2017) New drugs or alternative therapy to blurring the symptoms of fibromyalgia-a patent review. Multiuser Center for Health (C Multi-Saúde), Federal University of Sergipe, Sergipe, Brazil. Departament of Health Education, Federal University of Sergipe, Sergipe, Brazil. Expert Opin Ther Pat 27(10): 1147-1157.
- Mccleane G (2007) Intravenous lidocaine: an outdated or underutilized treatment for pain? Rampark Pain Centre, Lurgan, Northern Ireland, United Kingdom. J Palliat Med 10(3): 798-805.

 Research Article
Research Article