ABSTRACT
Cinnamic acid is a white crystalline substance, chemically known as 2(E)- 3-phenylprop-2-enoic acid or benzopropenoic acid. It is a mono-carboxylic acid naturally occurring in plants with low toxicity and serves as a flavoring agent in the food, pharmaceutical and perfumery industries. The acetylation of the acid led to acetyl derivative which has been identified to be cinnamyl acetate using the IR spectral technique. Both the cinnamic acid and acetyl derivative demonstrated remarkably significant antioxidant activity of IC50 at 0.18 and 0.16μg/mL respectively which compare favorably with 0.12μg/mL elicited by Vitamin C (a standard antioxidant drug). The obtained results indicate that acetylation slightly enhances the antioxidant activity of cinnamic acid.
Keywords: Cinnamic Acid; Acetylation; Cinnamyl Acetate; Derivative; Antioxidant Activity
Abbreviations: ROS: Reactive Oxidative Species; DPPH: 2, 2-diphenyl-1-picrylhydrazyl hydrate
Introduction
In the search for novel pharmacologically active compounds, research efforts are being focused on some lead chemicals/ compounds. One of such is cinnamic acid and its derivatives which are chemical compounds with high potentials for development into drug templates. Some of these compounds especially the ones containing phenolic hydroxyl group are well-known for their several health benefits due to inherent strong free radical scavenging properties. Hence, these compounds are being studied as potential antioxidants due to the multi-functional activities they exhibit. Furthermore, previous studies have shown that this class of compounds posses anti-microbial, anti-inflammatory, anti-cancer, anti-oxidative and cardiovascular protective properties. A derivative namely, p-coumaric acid (4-hydroxyl-trans-cinnamic acid) has been found to exhibit antioxidant activity involving direct scavenging of ROS (Reactive Oxidative Species) by minimizing the oxidation of low-density lipoprotein [1-3]. In addition, ethyl cinnamate and cinnamyl alcohol have been found to posses antioxidant activity using the rapid bench-top DPPH (2, 2-diphenyl-1-picrylhydrazyl hydrate) test [4]. In this present study, the acid was acetylated and the resultant derivative screened for antioxidant activity (IC50) using the DPPH test. Comparison of the activities given by the acid and the derivative was done with a view to determining if any improvements would be observed.
Experimental
Reagents/chemicals
DPPH (2, 2-diphenyl-1-picryl hydrazyl hydrate) and cinnamic acid were purchased from Sigma Aldrich Chemicals, Germany while acetic acid, acetic anhydride, di-ethyl ether, methanol and sulphuric acid were obtained as AnaLAR Grade Chemicals from British Drug House Chemicals Limited, Poole, England.
Acetylation of Cinnamic Acid
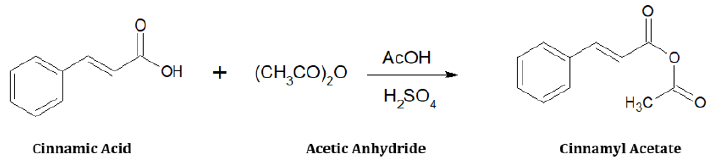
0.4g of cinnamic acid was dissolved in a beaker containing 10mL of acetic anhydride and 10mL of acetic acid. The solution was heated for 20 minutes and allowed to cool. 5mL of concentrated H2SO4 was added as catalyst. Further heating was done for few minutes and it was covered with an aluminum foil and kept in the refrigerator at -4 0C. After two weeks, crystals were formed in the beaker. 5mL of warm di-ethyl ether was added and the mixture gently heated again for a few minutes. The crystals dissolved on warming, but formed back after some hours. They were then filtered and allowed to dry [5,6]. The crystals were then weighed (Figure 1).
Determination of Melting Point
Cinnamic acid and the synthesized derivative (0.05g) were separately filled to a quarter of the length of a micro-capillary tube and the melting point determined [7] using an Electro-thermal Melting Point apparatus (Electro-thermal Engineering Limited, England).
Determination of Optical Rotation and Refractive Index
Each sample (0.05g) was dissolved in 10mL of methanol. The tube of the Polarimeter (ADP-220, Bellingham Stanley, England) was filled with distilled water and the machine subsequently zeroed. The tube was refilled with 5mL of sample and the optical rotation and was measured at the wavelength (λ) of sodium D line (589.3nm) at 20.5 0C. Similarly, the refractive index of sample was obtained on a refractometer (WAY-15, Abbe, England) at the wavelength (λ) of sodium D line (589.3nm) at 20.5 0C [8,9].
Antioxidant Activity
Spectrophotometric Determination of Antioxidant Activity
using DPPH Reagent: Substances which are capable of donating
electrons or hydrogen atoms can convert the purple-colored DPPH
radical (2, 2-diphenyl-1-picrylhydrazyl hydrate) to its yellowcolored
non-radical form; 1, 1-diphenyl-2-picryl hydrazine [10,11].
This reaction can be monitored by spectrophotometry.
Preparation of Calibration Curve for DPPH Reagent:
DPPH (4mg) was weighed and dissolved in methanol (100mL)
to produce the stock solution (0.004 % w/v). Serial dilutions of
the stock solution were then carried out to obtain the following
concentrations; 0.0004, 0.0008, 0.0012, 0.0016, 0.0020, 0.0024,
0.0028, 0.0032 and 0.0036 % w/v. The absorbance of each of the
sample was taken at λm 512nm using the Ultra-Violet
Spectrophotometer (Jenway 6405, USA). This machine was
zeroed after an absorbance had been taken with a solution of
methanol without DPPH which served as the blank.
Determination of the Antioxidant Activity of Cinnamic
Acid, Dderivative and Vitamin C: 2mg each of sample was
dissolved in 50mL of methanol. Serial dilutions were carried out
to obtain the following concentrations; 0.0004mg mL-1, 0.0008 mg
mL-1, 0.0012mg mL-1, 0.0016mg mL-1 and 0.0020mg mL-1 using
methanol. 5mL of each concentration was incubated with 5mL
of 0.004 % w/v methanolic DPPH solution for optimal analytical
accuracy. After an incubation period of 30 minutes in the dark at
room temperature (25 ± 2 0C), observation was made for a change
in the color of the mixture from purple to yellow. The absorbance
of each of the samples was then taken at λm 512nm. The Radical
Scavenging Activity (RSA %) or Percentage Inhibition (PI %) of free
radical DPPH was thus calculated:
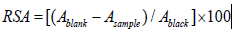
Ablank is the absorbance of the control reaction (DPPH solution
without the test sample and Asample is the absorbance of DPPH
incubated with the sample. Cinnamic acid /derivative / Vitamin C
concentration providing 50 % inhibition (IC50) was calculated from
a graph of inhibition percentage against the concentration of the
cinnamic acid/ derivative /vitamin C [12,13]. Vitamin C was used
as a standard antioxidant drug.
Infra-Red Spectroscopy of Samples: Each sample (0.2g)
was analyzed for IR characteristics using the FTIR 84005
Spectrophotometer (Shimadzu, Japan).
Results
Structural Elucidation
Cinnamic Acid: C9H8O2; mol. wt. (148g/mol); white crystalline
solid; m.pt. (134-136 0C); [n]D20 (1.516); [α]D20 (00); FTIR (cm-1):
1576 (Ar-C=C), 1627 (acyclic -C=C), 1682 (C=O) and 2923 (-OH)
(Tables 1 & 2).
Cinnamyl Acetate : C11H10O3; mol. wt. (190g/mol); yellow
crystals; m.pt. (169-171 0C); [n]D20 (1.552); [α]D20(00); FTIR (cm-
1): 771 (alkyl substitution ), 1071 (C-O-C, ether linkage), 1577 (Ar-
C=C), 1629 (acyclic -C=C) and 1683 (C=O).
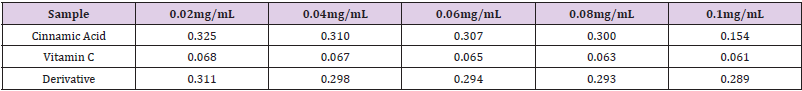
Table 1: Absorbance of samples incubated with DPPH at different concentrations at λmax 512nm (Blank absorbance of 0.004% methanolic DPPH reagent: 0.803).
Note: DPPH = (2, 2-diphenyl-1-picrylhydrazylhydrate)
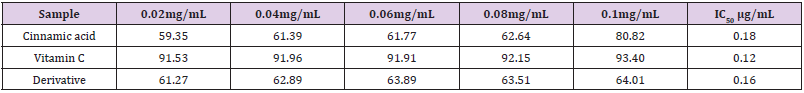
Table 2: Radical scavenging activity (RSA)/ percentage inhibition (PI) of samples at different concentrations and the computed IC50 values.
Note: IC50 = Concentration at which 50% of DPPH is scavenged or inhibited RSA% (PI) = Radical Scavenging Activity (Percentage Inhibition).
Discussion
The cinnamic acid used in this study had been put through some
monographic determinations in a previous research [4] where its
identity, purity, integrity, assay and suitability were established. Its
IR spectrum shows diagnostic stretching’s at 1576, 1627, 1682 and
2923cm-1 which indicate the characteristic Ar-C=C, acyclic C=C, α, β
unsaturated C=O and –OH groups respectively. The –OH peak of the
acid absorbed particularly lower the expected (>3000) at 2923 cm-1
because of inherent intra-molecular hydrogen bonds. Cinnamyl
acetate was synthesized as yellow crystals with a balsamic flavor.
The compound was found to be soluble in methanol, petroleum
ether, acetone, chloroform and ethanol. However, it was insoluble
in water conferring a lipophilic character on it. Its refractive index
was found to be 1.552 compared with the parent compound at
1.516 while its melting point was 169-171 0C. IR peaks of the acetyl
product at 771, 1071, 1577, 1629 and 1683 cm-1 are characteristic of
alkyl substitution (found in -COCH3 substitution in the derivative),
ether linkage -C-O-C, aromatic Ar-C=C (which was observed to
absorb slightly higher than that seen in the cinnamic acid), acyclic
-C=C and C=O stretching’s (both slightly higher than that seen in the
acid) respectively.
The ether linkage is particularly elucidative indicating that
the hydrogen atom in the -OH had been substituted with an acetyl
group. Both the acid and its acetyl derivative showed an optical
rotation of 0o indicating that the compounds are optically inactive.
Consequently, neither compounds can rotate plane of polarized
light in any directions. Hence, the compounds cannot demonstrate
either dextro-rotation or laevo-rotaion [8,9]. The derivative has also
been found to be of immense benefit to the pharmaceutical, winery
and perfumery industries. It is pertinent to mention that open or
acyclic carboxylic acids such as cinnamic acid are much difficult
to acetylate compared with the ringed or aromatic counterparts.
Consequently, the acetylation procedure had to be left for 14 days
in a refrigerator for the reaction to go into completion.
Antioxidant Activity
A calibration curve was prepared for DPPH (2, 2-Diphenyl-1- picryl hydrazyl hydrate) reagent with the aim of confirming its purity and suitability for use in the antioxidant determinations. The Beer- Lambert’s Law is the basis of all absorption spectrophotometry [9]. The reduction of the DPPH radical was determined by taking its absorption at a wavelength of λm 512nm. It was observed that the absorbance of DPPH decreased as the concentration of added free radical scavenger (cinnamic acid/derivative/vitamin C) increased which suggested that the DPPH reagent was being reduced. The tables show radical scavenging activity (RSA %) or percentage inhibition (PI %) and the computed IC50 values of cinnamic acid / derivative / vitamin C. The RSA % is an indicator of the antioxidant activity of cinnamic acid/ derivative/ vitamin C. Interestingly, both the cinnamic acid and derivative demonstrated significant antioxidant activity (IC50) of 0.18 and 0.16μg mL-1 respectively. The values compare remarkably with the antioxidant activity given by vitamin C (standard antioxidant drug) at 0.12μg mL-1. However, it should stated be that the derivative was slightly more active than the parent compound. Hence, it can be inferred that acetylation enhances the antioxidant activity of the acid. This was not surprising because the solubility profile of the synthesized derivative showed that it was soluble in organic solvents whereas it was insoluble in water implying that it has some lyphophillic characteristics. This feature most probably could account for its slightly better activity than the acid which can enable it to get it the active sites (allosteric sites) faster than the acid to effect anti-oxidation Aside from the DPPH assay, other methods for determining the antioxidant activity of compounds include the hydrogen peroxide, nitric oxide, conjugated diene, superoxide, phosphomolybdenum, peroxynitrile and xanthine oxidase assay methods amongst many others [14,15].
Conclusion
This present study shows that the cinnamic acid and its cinnamyl acetate elicited significant antioxidant activity. However, the activity demonstrated by the derivative was slightly better. Hence, acetylation enhances antioxidant activity of the acid.
Acknowledgement
The authors are grateful for the kind gesture of the Department of Pharmaceutical and Medicinal Chemistry, University of Uyo for the use of its Jenway 6405UV/VS Spectrophotometer in the antioxidant assays. Also, we acknowledge the contribution of the University of Ibadan, Nigeria for the use of their facilities in obtaining the IR spectra.
References
- Lei L, Hudgins WR, Shack S, Yin M Q, Samid D (1995) Cinnamic acid: A natural product with potential use in cancer intervention. Inter J of Cancer 62(3): 345-350.
- Lobo U, Patil A, Phatak A, Chandra N (2010) Free radicals, antioxidants and functional foods: impact on human health. Pharmacol Rev 4(8): 118-126.
- Sova M (2012) Antioxidant and antimicrobial activities of cinnamic acid and derivatives. Mini Rev in Mel Chem 12(8): 749-767.
- Oladimeji HO, Aniekeme EE, Arthur JS, Taiwo EA (2019) Evaluation of antioxidant activity of cinnamic acid and some of its derivatives. Eur Chem Bull 8(7): 224-226.
- Olugbade TA, Adesanya SA (2000) Prieurianoside, a protolimonoid glucoside from the leaves of Trichilia prieuriana, Phytochemistry 54(8): 867-870.
- Chris IA (2010) Laboratory organic chemistry: Techniques, qualitative analysis, organic preparations and spectroscopy (17th)., In: Chris IA (Edt.)., Maybinson Book Publishers, p. 55-56.
- (1993) British pharmacopeia. The pharmaceutical press, London, appendix VA, A109, pp. 754.
- Olaniyi AA (2000) Principles of quality assurance and pharmaceutical analysis. Mosuro publishers, pp. 151-158.
- Olaniyi A, Ogungbamila FO (1991) Experimental pharmaceutical chemistry. In: Olaniyi A, Ogungbamila FO (Eds.)., Shaneson CI limited, Nigeria, pp. 49-52.
- Nagalapur SK, Paramjyothi S (2010) In-vitro antioxidant activity of Launaea pinnatifida Cass leaves. The Bioscan 5(1): 105-108.
- Guangron g H, Jiaxin J, Dehui D (2008) Anti-oxidative and antibacterial activity of the methanol extract of Artemisia anomala S. Moore. Afri J Biotech 7(9): 1335-1338.
- Lebeau J, Furman G, Bernier JL, Durietz P, Teissier E, et al. (2000) Antioxidant properties of di-tert-butylhydroxylated flavonoids. Free-Radical Biology and Med 29(9): 900-912.
- Leitao GG, Leitao SG, Vilegas WZ (2002) Quick preparative separation of natural naphthoquinones with antioxidant activity by high speed counter current chromatography Z. Naturforsch 57(11-12): 1051-1055.
- Bunta GW, Ingold RU (1984) β-carotene: An unusual type of lipid antioxidant Sci 224: 569-573.
- Dapkevicius A, Vanskulons R, Van Beek TA, Linssen JP (1998) Antioxidant activity of extracts obtained by different isolation procedures from some aromatic herbs grown in Lithuania. J Sci Food Agric 77(1): 140-146.

 Research Article
Research Article