ABSTRACT
Background: Matrix metalloproteinase enzymes (MMPs) play a destructive role in
atherosclerosis resulting in occurrence of cardiac ischemia. The aim of this study was
to investigate the effect of concurrent training during cardiac rehabilitation on plasma
levels of MMP-9 and tissue inhibitor of metalloproteinase-1 (TIMP-1) in patients with
myocardial ischemia.
Methods: Sixteen cardiac ischemic patients who were in the process of clinical
cardiac rehabilitation were randomly assigned to experimental (n = 8) or control (n =
8) groups. The patients in experimental group performed concurrent exercise for eight
weeks (one hour / three session per week) with the intensity of 40-60% of one repetition
maximum and 60-80% maximum heart rate. Pre- and post-intervention blood samples
were taken to assess plasma MMP-9 and TIMP-1 concentrations using ELISA method.
Findings: Independent T-test showed the patients in experimental group had
a significant decrease (t = 2.431; p = 0.029) in MMP-9 plasma concentration and a
significant increase (t = 3.202; p = 0.006) in plasma levels of TIMP-1 compared to the
control group. A significant within-group reduction in MMP-9 levels (t = 0.695; P =
0.008) and a significant within-group increase in TIMP-1 levels (t = 3.964; P = 0.005)
were observed in the experimental group, while the pre- to post-value changes in MMP-
9 and TIMP-1 levels in the control group were not statistically significant.
Conclusion: It seems that the favorable changes in MMP-9 and TIMP-1 following
eight weeks of physical exercise prevent the progression of atherosclerosis and
recurrence of cardiac ischemia during the process of cardiac rehabilitation.
Keywords: Matrix Metallopeptidases, Tissue Inhibitor of Metalloproteinase, Exercise Training, Cardiac Rehabilitation, Cardiac Ischemia
Abbreviations: MMPs: Matrix Metalloproteinase Enzymes; TIMP-1: Tissue Inhibitor of Metalloproteinase-1; 1RM: One Repetition Maximum; EG: Experimental Group; CG: Control Group; MHR: Maximum Heart Rate; RPE: Borg Rating of Perceived Exertion; ELISA: Enzyme-Linked Immunosorbent Assay; MCP-1: Monocyte Chemoattractant Protein-1; Ox-LDL: Oxidized Low-Density Lipoprotein
Introduction
Cardiac ischemia is considered as one of the deadliest heart
diseases. According to the reports from American Heart Association,
one third of cardiac patients suffer from ischemic heart ailment
[1]. Several genomic and environmental factors including but
not limited to family history, gender, age, smoking, hypertension,
diabetes, hyperlipidemia, and lack of physical activity can potentially
contribute to atherosclerosis and subsequently ischemic heart
disease [2,3]. Atherosclerosis, a chronic inflammatory disease, is
characterized as the accumulation of fat and other substances on
the inner surface of artery walls [4]. In the atherosclerotic situation,
due to the deficiency of blood flow in the coronary arteries, an
acute blockage occurs which is defined as a heart attack [5]. With
the progression of atherosclerosis, the smooth muscle cells move
toward the intima in response to growth factors released by active
macrophages and endothelial cells. Active macrophages weaken the
extracellular matrix and support the platelet fibrosis by producing
matrix metalloproteinases (MMPs) suggesting a prominent role
in the process of atherosclerosis and rupturing the artery walls
[6]. Matrix metalloproteinases (MMPs), which are a large group
of protease enzymes, are responsible for the breaking down of
extracellular matrix.
To date, 26 members of MMP family have been identified, and
among them, gelatinase-B (or MMP-9) has a higher activity than
other members [7] with a well-documented potential in developing
cardiovascular disease in human populations [8]. Based on the
evidence, myocardium expression of MMP-9 is increased in the
patients with coronary artery disease [9,10]. However, due to the
damaging role of MMPs, their activity is strongly regulating by four
tissue inhibitors (TIMPs) namely TIMP-1, TIMP-2, TIMP-3, and
TIMP-4 which can control the detrimental activities of MPPs in
vertebrates [11]. Among the TIMP family, it has been reported that
TIMP-1 is a potent inhibitor of many MMPs including MMP-9 [12]
and play a key role in the structure and function of myocardium
ae well as the control of extracellular matrix proteolysis in
cardiovascular system [12]. Recurrence of cardiac ischemia
and its unfavorable consequences are a major concern among
cardiac ischemic patients as well as cardiovascular specialists [8].
Nowadays, with developing clinical centers, medical strategies,
and patients’ knowledge in diagnosing early signs of heart disease
the rate of mortality has been clearly decreased; however, some of
these patients with the acute symptoms have been hospitalized for
the second and third time and some of them still may have died
[13].
In addition to pharmacological treatments and implementing
medical interventions on coronary arteries, cardiac rehabilitation
program has been well-documented in controlling the risk factors,
improving life quality, and reducing the mortality rate [14]. The
cardiac rehabilitation program is designed to limit the physiological
and psychological consequences of cardiovascular disease, reduce
the risk of sudden death or stroke, control cardiac symptoms, and
decline the atherosclerosis [15]. Physical exercise is frequently
used as a non-pharmacological and supplemental method in the
process of medical treatment for a variety of reasons [16]. As an
incentive method, physical exercise can be used during cardiac
rehabilitation programs due to its low cost and attractiveness
[17]. The type of physical exercise used in cardiac rehabilitation
programs is generally aerobic in forms of walking, running, or
cycling [18]. However, it has been recently reported that a welldesigned
resistance training program accompanied by aerobic
exercise can be considered as a helpful strategy in rehabilitation
programs. In this regard, the American Heart Association has also
recommended that resistance training be performed twice a week
in cardiac rehabilitation programs [18].
According to the recent scientific reports, the implementation
of physical exercise in rehabilitation programs can lead to effective
outcomes on some clinical indicators involved in the occurrence
of atherosclerosis [19]. In a study examining the effect of cardiac
rehabilitation on the atherosclerosis biomarkers in patients with
cardiac ischemia, the authors reported that performing physical
exercise during cardiac rehabilitation programs will prevent reoccurrence
of cardiac ischemia [20]. However, due to the increasing
prevalence of ischemic heart disease [1] and considering the
importance role of MMPs particularly MMP-9 in the formation
and stabilization of atherosclerotic plaque, few studies have yet
examined the effects of a well-planed physical exercise during
cardiac rehabilitation on the levels of metalloproteinases and
their tissue inhibitors in patients with cardiac ischemia [21,22].
Therefore, the need for a comprehensive investigation in this
promising area is clearly evident and the present study is designed
to examine this imperative issue.
Materials and Methods
The present study is an experimental controlled clinical trial.
Sixteen cardiovascular patients with atherosclerotic symptoms who
were referred to a cardiac hospital in Mashhad, Iran were selected
and randomly assigned to an experimental (n = 8) or control (n = 8)
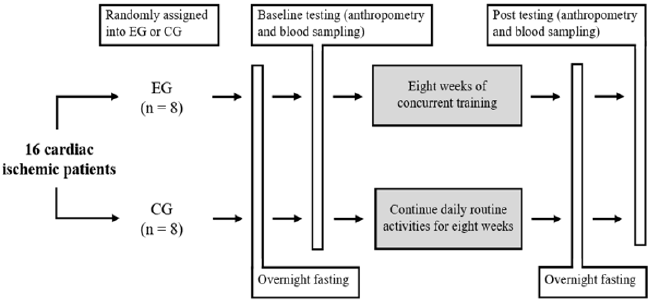
group. Scheme of the study design is shown in Figure 1. Before the
commencement of the study, patients in both groups were monitored
by a cardiologist and were reported to have symptoms of coronary
heart disease. For each patient, a file including demographic
information, history, clinical cardiac reports, and anthropometric
data was provided. At the beginning of the rehabilitation program,
an initial assessment of cardiac function was conducted using
echocardiographic devices. During the study, participants in both
groups followed the prescribed medications and diets (with a
particular amount of calorie and macronutrients per kilogram of
body weight for all participants) which were provided by a dietitian and a cardiologist. The patients in experimental group completed
an eight-week moderate concurrent exercise program with 40-60%
of their one maximum repetition.
According to the American Heart Association guidelines, the
patient’s exercise intensity was monitored to be between 60 and
80% of their maximum heart rate [23] using the individualized
electrocardiograms connecting to each participant during exercise
sessions. Moreover, Borg Rating of Perceived Exertion (RPE) scale
was also used to a further control of the exercise intensity, and the
patients in experimental group were asked to keep their activity
intensity between level 11 (relatively light) and level 13 (somewhat
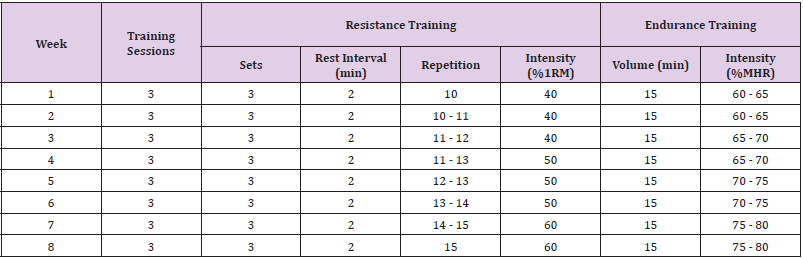
difficult) [24]. The concurrent training protocol is shown in Table
1. Before the commencement of training sessions, participants in
experimental group were informed and introduced to devices and
exercises in one session and then practiced three sessions per
week for eight weeks. Each training session lasted about one hour
performing mixed aerobic and anaerobic exercises using treadmills,
bikes, ergometers, physio balls, and light weights. At the starting of
each raining sessions 5 minutes of warm-up, 5 minutes of walking
on treadmill, 5 minutes of bike pedaling, and 5 minutes of upperbody
ergometer pedaling were done with three minutes of inactive
rest between each aerobic exercise.
After 5 minutes of rest, the individuals gradually performed
the following workouts with 10 repetitions in three sets in the
initial training sessions and to 15 repetitions in the advanced
sessions. The workouts consisted of squat with a physio ball,
shoulder flexion, shoulder abduction, elbow flexion, hip flexion,
hip abduction, ankle plantar flexion, and ankle dorsi flexion. The
workouts were initially performed using the patient’s own body
weight or limb weight, and gradually improved using therabands
and light weights [25]. A careful supervision was applied during
the training sessions, and the patients were constantly questioned
about the amount of pressure based on the Borg scale. If the value
of Borg scale was reported below 11 or above 13, the participant
was asked to increase or decrease the effort, respectively.
Laboratory Methods: Fasting 10cc blood samples were
obtained 48 hours before the first training session and 48 hours
after the last training session from the participants’ antecubital
vein in both groups. Blood samples were collected in lavendertop
tubes containing anti-coagulant EDTA, and then were carried
to laboratory for plasma separation by 3000rpm centrifuging
for 10 minutes. Subsequently, plasma samples were frozen and
stored at -80°C for further analysis. Plasma MMP-9 and TIMP-1
concentrations were assessed by Enzyme-Linked Immunosorbent
Assay (ELISA) method using Awareness Stat Fax 2100 device
according to the instructions of the ELISA kits (R&D Systems,
Minneapolis, MN, USA).
Results
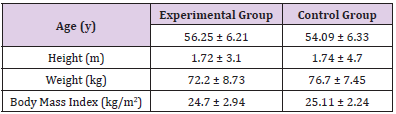
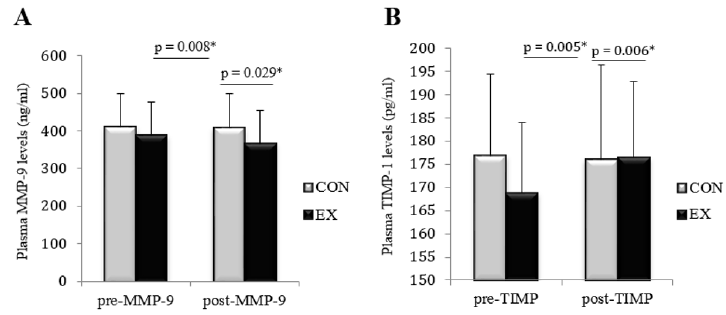
Data distribution was normal according to the results from Shapiro Wilk test. The baseline characteristics of the participants are shown in Table 2. The independent T-test showed a significant decrease in plasma MMP-9 concentration (t = 2.431; p = 0.029) and a significant increase in TIMP-1 plasma levels (t = 3.202; p = 0.006) following the exercise intervention in the exercise group compared to the control group. According to the correlated T-test, a significant within-group decrease in MMP-9 levels (t = 0.695; p = 0.008) and a significant increase in plasma TIMP-1 concentrations (t = 3.964; p = 0.005) were observed in the experimental group, while within-group changes in MMP-9 and TIMP-1 levels were not significant in the control group. (t = 0.21; p = 0.838 and t = 0.46; p = 0.66, respectively) (Figure 2).
Table 2: Baseline characteristics of the participants in experimental (n = 8) and control (n = 8) groups (mean ± SD).
Figure 2: The average changes in plasma levels of MMP-9 (part A) and TIMP-1 (part B) between per- and post-test values in experimental (EX) and control (CON) groups.
Discussion
Present study showed that after eight weeks of moderate
physical exercise during the cardiac rehabilitation period, MMP-
9 levels decreased and TIMP-1 levels increased, and significant
within-group changes in MMP-9 and TIMP-1 levels were observed
in the experimental group. To date, the investigations assessing
the effects of exercise on MMPs and TIMPs levels have often
been conducted in healthy, obese, or diabetic populations as well
as animal models. Seydanlou and Farzanegi in 2014 reported a
significant 20% decrease in MMP-2 levels and a significant 26% increase in TIMP-1 levels of overweight individuals following eight
weeks of Pilates training [26]. Consistent with our results, Kadoglou,
et al. in 2013 found a significant increase in TIMP-1 concentrations
following six weeks of treadmill running in animal models [27],
similar to the results of Koskkinen’s study but in humans [28].
Although the effectiveness of physical exercise on MMP-9 and
TIMP-1 levels are well documented in the literature, limited studies
have reported no change in the aforementioned variables following
different exercise interventions. Previously, Mackey, et al. reported
that a 10-kilometer road and water running has no effect on MMP-2
and MMP-9 levels in young men [29].
Moreover, according to the results of a study by Hoier, et al.,
eight weeks of cycling training with the intensity of 60% maximum
oxygen consumption did not affect the TIMP-1 levels in healthy
men [30]. However, these inconsistent findings might be justified
by different training methods, varied participants’ characteristics,
and different sampling methods (muscle biopsy vs blood sample)
used in the aforesaid investigations. Metalloproteinase matrix
enzymes are believed to alter the formation of the cardiovascular
matrix during natural biological processes [31]. However, in
pathophysiological processes of various diseases, the expression
and activity of these type of proteolytic enzymes are increased
due to the increased secretion of proinflammatory cytokines
leading to breakdown of several collagens and gelatinases, as well
as detrimental effects on microanatomical tissue structures. As a
result, exacerbation of inflammatory state and emerging of various
heart and vascular diseases might occur with advancing time
[32,33]. In a study examining the role of matrix metalloproteinase
1, 2, 3, and 9 in acute myocardial infarction, the authors reported
that the plasma concentrations of various MMPs in patients with
myocardial infarction were significantly increased [8].
They concluded MMP-9 may play a prominent physiological
and pathological role during the stage of myocardial infarction to
heart failure, more especially considering the fact that improper
vascular redisposition of MMP-9 may promote atherosclerosis via
weakening of atherosclerotic plaques [34]. It seems the potential
mechanisms for reducing exercise-induced MMP-9 levels during
cardiac rehabilitation is the changes in the levels of monocyte
chemoattractant protein-1 (MCP-1) [35]. MCP-1 attracts monocytes
to inflammatory sites located in the vascular subendothelial space.
These monocytes are able to differentiate into macrophages and
be converted to foam cells via absorbing oxidized low-density
lipoproteins (Ox-LDL), suggesting an important role for MCP-1 in
the pathogenesis of atherosclerosis [36]. The inhibition of MCP-
1 by relevant inhibitors prevents plaque inflammation and stops
the rupture of disposed plaques [37]. On a better note, MCP-1 in
myocytes and smooth vascular cells stimulates the expression of
MMP enzymes and consequently induces inflammatory cytokines
and MMP enzymes in cardiac myocytes [38].
Although MCP-1 levels were not measured in the present study,
several studies have reported a reduction in MCP-1 following
exercise suggesting a beneficial effect of physical exercise on heart
patients. Thus, MCP-1 reduction is likely links to decreased plasma
MMP-9 levels following regular exercise [39]. The activity of MMP
inhibitors and the ratio of MMPs to TIMPs are as important as the
secretion of tissue MMPs. Under normal conditions, a physiological
balance between MMPs and TIMPs is stablished, and the
extracellular matrix breakdown and synthesis is well-adjusted. Any
disease or mechanical stress that results in decreased immune cells,
increased inflammation, secretion of proinflammatory cytokines,
and the activity of MMPs, consequently triggers the immediate
inhibitory response particularly activation of TIMPs [40]. Hence,
the inhibition of MMP proteolytic activity has been suggested as a
therapeutic approach in various heart diseases [41-43].
Conclusion
In the present study, an inverse relationship between the bioactivity of MMP-9 and TIMP-1 was observed following eight weeks of exercise intervention in ischemic cardiac patients. Considering the favorable effects of physical exercise on cardiac rehabilitation, it can be suggested that patients with cardiac ischemia can accelerate their recovery process and reduce the risk of stroke reoccurrence by doing moderate intensity of physical exercise at least three time per week.
Acknowledgement
The authors wish to thank the participants in this research project.
Funding
This research did not receive any specific grant or funding from agencies in the public, commercial, or not-for-profit sectors.
Conflict of Interest
No conflicts of interest are declared by the authors.
References
- Chughtai H, Ratner D, Pozo M, Crouchman JA, Niedz B, et al. (2011) Prehospital Delay and its Impact on Time to Treatment in ST-elevation Myocardial Infarction. Am J Emerg Med 29(4): 396-400.
- Kim JH, Kim DJ, Jang HC, Choi SH (2011) Epidemiology of Micro- and Macrovascular Complications of Type 2 Diabetes in Korea. Diabetes Metab J 35(6): 571-577.
- Lusis AJ (2000) Atherosclerosis. Nature 407(6801): 233-241.
- Malekpour Dehkordi Z, Javadi E, Doosti M, Paknejad M, Nourbakhsh M, et al. (2011) The Effect of Alcoholic Garlic (Allium sativum) Extract on ABCA1 Expression in Human THP-1 Macrophages. Tehran Univ Med J 69(3): 146-152.
- Fariba F, Esna Ashari F, Moradi M (2015) Comparison of Risk Factors Frequency in Patients with ST-elevation MI & Patients with non ST-elevation MI in Hamadan City. Scientific Journal of Hamadan University of Medical Sciences 22(1): 11-15.
- Weiss TW, Furenes EB, Trøseid M, Solheim S, Hjerkinn EM, et al. (2010) Prediction of Cardiovascular Events by Matrix Metalloproteinase (MMP)-9 in Elderly Men. Thromb Haemost 103(3): 679-681.
- Kjaer M (2004) Role of Extracellular Matrix in Adaptation of Tendon and Skeletal Muscle to Mechanical Loading. Physiol Rev 84(2): 649-698.
- Tabatabaei Panah A, Akbarzadeh R, Khodaii Z, Ghaderian S (2016) Role of Matrix Metalloproteinase 1, 2, 3 and 9 in Acute Myocardial Infarction. Scientific-Research Journal of Shahed University 23(122): 29-39.
- Cleutjens JP, Kandala JC, Guarda E, Guntaka RV, Weber KT (1995) Regulation of Collagen Degradation in the Rat Myocardium after Infarction. J Mol Cell Cardiol 27(6): 1281-1292.
- Romanic AM, Burns Kurtis CL, Gout B, Berrebi Bertrand I, Ohlstein EH (2001) Matrix Metalloproteinase Expression in Cardiac Myocytes Following Myocardial Infarction in the Rabbit, Life Sciences 68(7): 799-814.
- Lee Johnson J (2014) Matrix Metalloproteinases and Their Inhibitors in Cardiovascular Pathologies: Current Knowledge and Clinical Potential. Metalloproteinases in Medicine 1: 21-36.
- Arpino V, Brock M, Gill SE (2015) The Role of TIMPs in Regulation of Extracellular Matrix Proteolysis. Matrix Biol 44-46: 247-254.
- Farshidi H (2004) Causes of Readmission in Ischemic Heart Disease Patients. Scientific information database 8(2): 67-71.
- Antam ME, Selwyn AP, Loscalszo (2012) Ischemic heart disease. In: Lango DL, (Edi.). Harrisons principles of medicine. (18th). New York: McGrow Hill, Health Profession Division, pp. 1998-2015.
- Dalir Z, VahdatFeizabadi E, Mazlom S, RajaeeKhorasani A (2013) The Effect of Short-term Cardiac Rehabilitation Program on Anxiety and Depression in Patients After Coronary Artery Bypass Surgery. Evidence Based Care Journal 3(8): 33-42.
- Piepoli MF, Davos C, Francis DP, Coats AJ (2004) Exercise Training Meta-Analysis of Trials in Patients with Chronic Heart Failure (ExTraMATCH). BMJ 328(7433): 189.
- Dakei SZ, Hemmat Far A, Azizbeigi K (2014) Effect of Resistance and Endurance Training Protocols on Functional Capacity and Quality of Life in Male Patients after Myocardial Infarction. Cardiovascular Nursing Journal 3(1): 26-33.
- Volaklis K, Spassis A, Tokmakidis S, Komotini (2007) Land Versus Water Exercise in Patients with Coronary Artery Disease: Effects on Body Composition, Blood Lipids, and Physical Fitness. American Heart Journal 154(3): 560-566.
- Afzalaghaiee E, Hadian M, Attarbashi Moghadam B, Tavakol K, Zandparsa A, et al. (2010) The Effects of Cardiac Rehabilitation on Changes in The Lipid Profile of Iranian Male and Female with Coronary Artery Disease Refered to Emam Khomeini Hospital Complex. MRJ 4(1-2): 45-49.
- Hajighasemi A, Ravasi A, Kordi M, Rashidlamir A, Ghorghi A (2017) Investigation of the Effect of Cardiac Rehabilitation Program on Peripheral Blood Mononuclear Cells ABCA1 Gene Expression in Myocardial Infractions Patient. Journal of Knowledge & Health 11(4): 23-29.
- Shokrallahnia Roshan A, Sadeghi H, Shirani SH, Nejatian M (2013) Effects of Strength Training and Cardiac Rehabilitation Programs on the Biomechanical Parameters of Blood Flow Velocity and Blood Flow Rate and its Relation with Arterial Stiffness Index in Brachial and Femoral Arteries with Coronary Artery Bypass Grafting Patients (CABG). Journal of Rehabilitation 14(2): 38-45.
- Hoang A, Tefft C, Duffy SJ, Formosa M, Henstridge DC, et al. (2008) ABCA1 Expression in Humans is Associated with Physical Activity and Alcohol Consumption. Atherosclerosis 197(1): 197-203.
- Mostafavi S, Saeidi M, Heidari H (2012) Effects of a Comprehensive Cardiac Rehabilitation Program on the Quality of Life of Patients with Cardio-Vascular Diseases. Journal of Research in Rehabilitation Sciences 7(4): 516-524.
- Borg GA (1962) Physical performance and perceived exertion: Gleerup Lund.
- Pollock ML, Franklin BA, Balady GJ, Chaitman BL, Fleg JL, et al. (2000) Resistance Exercise in Individuals with and without Cardiovascular Disease Benefits, Rationale, Safety, and Prescription an Advisory from the Committee on Exercise, Rehabilitation, and Prevention, Council on Clinical Cardiology, American Heart Association. Circulation 101(7): 828-833.
- Seidanloo F, Farzanegi P (2015) Changes in Matrix Metallo-proteinases 2, 9 and Tissue Inhibitor of Matrix Metalloproteinase 1 to Synchronized Exercise Training and Celery, as an Herbal Supplement, in Overweight Women. Modaress Journal of Medical Sciences: Pathobiology 18(1): 107-118.
- Kadoglou NP, Moustardas P, Kapelouzou A, Katsimpoulas M, Giagini A, et al. (2013) The Anti-Inflammatory Effects of Exercise Training Promote Atherosclerotic Plaque Stabilization in Apolipoprotein E Knockout Mice with Diabetic Atherosclerosis. Eur J Histochem 57(1): e3.
- Koskinen SO, Heinemeier KM, Olesen JL, Langberg H, Kjaer M (2004) Physical Exercise Can Influence Local Levels of Matrix Metalloproteinases and their Inhibitors in Tendon -Related Connective Tissue. J Appl Physiol 96(3): 861-864.
- Mackey AL, Donnelly AE, Swanton A, Murray F, Turpeenniemi Hujanen T (2006) The Effects of Impact and Non-Impact Exercise on Circulating Markers of Collagen Remodelling in Humans. J Sports Sci 24(8): 843-848.
- Hoier B, Nordsborg N, Andersen S, Jensen L, Nybo L, et al. (2012) Pro- and Anti-Angiogenic Factors in Human Skeletal Muscle in Response to Acute Exercise and Training. J Physiol 590(3): 595-606.
- Kim HE, Dalal SS, Young E, Legato MJ, Weisfeldt ML, et al. (2000) Disruption of the Myocardial Extracellular Matrix Leads to Cardiac Dysfunction. The Journal of Clinical Investigation 106(7): 857-866.
- Murphy G, Cockett MI, Stephens PE, Smith BJ, Docherty AJ (1987) Stromelysin is an Activator of Procollagenase. A Study with Natural and Recombinant Enzymes. Biochemistry Journal 248(1): 265-268.
- Lee SW, Song KE, Shin DS, Ahn SM, Ha ES, et al. (2005) Alterations in peripheral Blood Levels of TIMP-1, MMP-2, and MMP-9 in Patients with Type-2 Diabetes. Diabetes Res Clin Pract 69(2): 175-179.
- Galis ZS, Khatri JJ (2002) Matrix Metalloproteinases in Vascular Remodeling and Atherogenesis: the Good, the Bad, and the Ugly. Circ Res 90(3): 251-262.
- Farzanegi P (2014) Impact of the Synchronization of Portulaca Oleracea and Aerobic Training on Levels of MMP2 and MMP9 and TIMP1 in Diabetic Women Type II. Res Mol Med 2(2): 34-39.
- Lin J, Kakkar V, Lu X (2014) Impact of MCP-1 in Atherosclerosis. Curr Pharm Des 20(28): 4580-4588.
- Niu J, Kolattukudy PE (2009) Role of MCP-1 in Cardiovascular Disease: Molecular Mechanisms and Clinical Implications. Clin Sci 117(3): 95-109.
- Azfer A, Niu J, Rogers LM, Adamski FM, Kolattukudy PE (2006) Activation of Endoplasmic Reticulum Stress Response During the Development of Ischemic Heart Disease. Am J Physiol Heart Circ Physiol 291(3): H1411-1420.
- Kazemi A (2017) Effect of High Intensity Interval Training on Visceral and Subcutaneous Levels of MCP-1 and Plasma Insulin and Glucose in Male Rats. Razi Journal of Medical Sciences 23(152): 29-37.
- Dastani M, Rashidlamir A, Alizadeh A, Seyedolhosseini M, Ebrahimi AA (2014) Effects of 8 Weeks of Aerobic Exercise on Matrix Metalloproteinase-9 and Tissue Inhibitor Levels in Type II Diabetic Women. Zahedan Journal of research in medical Sciences 16(6): 12-15.
- Woessner JF (1991) Matrix Metalloproteinases and their Inhibitors in Connective Tissue Remodeling. FASEB Journal 5(8): 2145-2154.
- Spinale FG, Coker ML, Krombach SR, Mukherjee R, Hallak H, et al. (1999) Matrix Metalloproteinase Inhibition during the Development of Congestive Heart Failure: Effects on Left Ventricular Dimensions and Function. Circulation Research 85(4): 364-376.
- Haq M, Shafii A, Zervos EE, Rosemurgy AS (2000) Addition of Matrix Metalloproteinase Inhibition to Conventional Cytotoxic Therapy Reduces Tumor Implantation and Prolongs Survival in a Murine Model of Human Pancreatic Cancer. Cancer Research 60(12): 3207-3211.

 Research Article
Research Article