Abstract
Background: Yunnan is a province of China most seriously affected by plague outbreaks. From 2005 to 2016, Plague in human was sporadically reported in Yunnan. Understanding the composition of small mammals and parasitic fleas in a plague natural focus might inform plague prevention and control measures.
Objective: This study aimed to investigate the distribution and composition of small mammals and parasitic fleas in the Yulong plague focus by comparing with those in the Jianchuan plague focus in Yunnan Province.
Methods: This study was conducted during December 2015 to October 2016, two wild plague foci included Yulong and Jianchuan were selected as study region, small mammals were captured by Dead traps in the spring, summer, autumn and winter and the parasitic fleas were collected by combing. The relevant indicator of small mammals and parasitic fleas were calculated, with compared by the Chi-square test and Fisher’s exact test.
Results: In the study, 884 small mammals representing 18 species were captured in the Yulong plague focus, at a density of 17.98%. From these mammals, 484 parasitic fleas representing 9 species were collected, with the flea prevalence of 24.77%. Further, 1069 small mammals representing 17 species were captured in the Jianchuan plague focus, at a density of 21.29%. From these mammals, 618 parasitic fleas representing 9 species were collected, with the flea prevalence of 27.60%. The small mammal density in the Jianchuan plague focus was significantly higher than that in the Yulong plague focus (Chi-squared test, P<0.01), but there was no significant difference in flea prevalence (Chi-squared test, P=0.159). The seasonal composition of small mammals and parasitic fleas in the two plague foci was significantly different (small mammals, Chi-squared test, P<0.01, parasitic fleas, Chi-squared test, P<0.05).
Conclusion: Apodemus chevrieri, Apodemus draco, and Eothenomys miletus were the dominant hosts in the two plague foci. In Yulong focus, Neopsylla specialis specialis and Ctenophthalmus (Sinoctenophthalmus) quadratus were the common dominant vectors. The compositions of small mammals and parasitic fleas in the two plague natural foci are similar, while some unique characteristics were existed in the Yulong plague focus. A. draco thrives better and E. proditor was only a secondary host or popular host. N. specialis specialis was the most abundant in summer of the Yulong plague focus, but C. (S.) quadratus was the most abundant in spring of the Jianchuan plague focus.
Keywords: Small Mammal; Parasitic Flea; Plague Natural Focus; Yunnan Province
Abbreviations: Y. pestis: Yersinia Pestis; A. chevrieri: Apodemus Chevrieri; E. miletus: Eothenomys Miletus; A. draco: Apodemus Draco, E. proditor: Eothenomys Proditor; A latronum: Apodemus Latronum; Ma. Himalayana: Marmota (Ma.) Himalayana; N. specialis specialis: Neopsylla Specialis Specialis; C. (S.) quadrates: Ctenophthalmus (Sinoctenophthalmus) Quadrates; Macrostylophora (M.) euteles: M. Euteles, C. (S.) Yunnanus: Ctenophthalmus (Sinoctenophthalmus) Yunnanus; F. spadix spadix: Frontopsylla (F.) Spadix Spadix
Introduction
Plague is caused by Yersinia pestis which a gram-negative bacterium, usually transmitted by fleas bitten and small mammals as the main reservoir. Plague is one of the most serious infectious diseases in human history. To date, three plague pandemics-the Justinian Pandemic, which started in AD 541, the Black Death, which started in AD 1347, and the Third Modern Pandemic, which started in AD 1772-have caused hundreds of millions of deaths worldwide [1,2]. Plague as a natural epidemic disease, is independent of human and exists in natural foci [3]. The disease continues to be a threat to human health, with plausible outbreaks in Madagascar, sporadically spreading in Tanzania, Vietnam, China, and the United States et al in last decades [4]. According to the WHO(World Health Organization), 2417 cases of plague, including 1854 cases of pneumonic plague and 355 cases of bubonic plague, and 209 deaths were reported in Madagascar between August 1 to November 26, 2017 [5]. This outbreak highlights the notion that plague are exists and continues to pose a threat to human health.
China is one of the most complex plague areas in the world, consisting of twelve types of plague natural foci. In the 21st century, the plague natural foci area in northwest China (such as Inner Mongolia, Gansu) are still active, while other foci area (like Yunnan, Gunangxi, Guizhou, etc.) are relatively stable, only animal plague and sporadic human cases reported [6]. The most serious plague outbreak in China was occurred in the Yunnan province. Currently, the plague epidemic affects approximately 10 million people in Yunnan. Plague is therefore an important public health problem in this province [7]. There are wild rodent plague foci and commensal rodent plague foci existed in Yunnan. The former includes those in Yulong and Jianchuan, with plague outbreaks reported several times since the first confirmation of plague foci, among the two areas, animals plague mostly occurred in woodland, cultivation or bush while human plague occurred by uncertain reason, which all above have been caused great burden of local residents [8,9].
The Jianchuan plague focus was identified in 1974, until now animal plague was reported many times but no human cases reported. The main reservoir are Apodemus chevrieri and Eothenomys miletus, and Ctenophthalmus (Sinoctenophthalmus) quadrates and Neopsylla specialis specialis are as the major vector to transmit Y. pesti s [9]. The Yulong plague focus is located to the north of Jianchuan plague focus and is connected with the Jianchuan plague focus. On October 25, 2005, five cases of severe pneumonia plague and two deaths were reported among residents of the same village in Yulong County [8], later the plague focus was confirmed in Yulong county in 2006 by Y. pestis culture, but the composition and distribution of small mammals, fleas and microenvironment in this focal area is unclear until now [10]. On April, 2017, one small mammal (Rattus tanezumi) and two fleas (Neopsylla specialis specialis and Frontopsylla spadix spadix) were successfully isolated Y. pestis in Jianchuan focus implied the animal plague epidemic is ongoing and the outbreak of human plague may occur at any time in the focal area [9].
Comparing to Jianchuan plague focus, however, limited researches of small mammals and fleas in the Yulong plague focus may cause us lose the initiative to prevent and control plague before the disease occurred in human. Once the environment variables are changing, they will be affecting the abundance of small mammals and fleas and plague will be re-mergence in human again. Therefore, we need to make efforts concentrated on composition of small mammals and parasitic fleas in the Yulong plague focus, and comparing to Jianchuan plague to evaluated structure and characteristics of Yulong plague foci clearly, where is a new relatively active plague natural focus in Yunnan province. Plague mainly depends on the animal hosts and vector fleas. The animal hosts parasitized by fleas plays an important role in plague transmission and conserving Y. pestis [11]. In a plague natural focus, one of the main approaches to controlling the incidence of the disease is by controlling the dynamics of host and flea populations [12]. However, previous study was mostly analyzed the characteristics of the Yulong focus from the Y. pestis [13]. It is still unclear whether the dominant small mammals and vector fleas are similar to the adjacent Jianchuan focus. In this study, we aim to investigate the distribution and composition of small mammals and parasitic fleas in the Yulong plague focus by comparing with those in the Jianchuan plague focus. This finding will be better understanding of the composition and distribution of small mammals and fleas may contribute to developing earlier warning methods for human plague outbreaks.
Materials and Methods
Study Design
This is a cross-sectional study based on the epidemiological information for the plague natural foci in the Yunnan province, two wild rodent plague foci included Yulong and Jianchuan were selected [8]. Small mammals were captured and parasitic fleas collected seasonally in these foci from December 2015 to October 2016.
Study Setting
The Yulong and Jianchuan plague foci are all located in the middle of the Hengduan Mountains with similar geographical environment, but separated by the low-lying valley in the center of the two regions. The Yulong plague focus is located in the northwest Yunnan province (latitude, 26°42’47’’ N to 26°56’13’’ N, longitude, 99°48’37’’ E to 100°16’39’’ E). The landscape is cultivation with alpine pine forest or mixed forest mountain area between 2900~3300 meter, having a warm temperate climate. The Jianchuan plague focus is closing to Yulong focus (latitude, 26°10’ N to 26°45’ N, longitude 99°28’ E to 99°56’ E). The landscape is cultivation with pine forest mountain area between 2100–3000 meter, having a cold temperate climate [14]. Previously studies showed Yulong focus ever had human plagues with two people died but Jianchuan focus has long plague history only animals plagues reported [8,9,15].
Sampling Period
The sampling was separately conducted in each focus in each season. The four seasons are winter (which started from 30th Nov to 31st Dec, 2015), spring (6th Mar to 29th Mar, 2016), summer (7th July to 16th July, 2016), autumn (9th Oct to18th Oct, 2016).
Collection of Small Mammals and Parasitic Fleas
In this study, the areas for wild trapping setting were selected and suggested by the experts from Jianchuan Station for Endemic Disease Control and Prevention of Jianchuan and Yulong CDC in different landscapes (such as woodland, cultivated, bush, etc.), They provide the setting where small mammals captured were the most or plague had occurred based on annual small mammals monitoring data and plague record data. Dead traps were used with one or two peanuts and all traps were placed the whole night started from the evening of the day to next early morning. Among them, one Dead trap were placed with no repetition in every 20 meters in per setting, this arrangement yielded at least 200 traps in one night of each focus. The captured small mammals were euthanized and brought to Jianchuan Station for Endemic Disease Control and Prevention and Yulong CDCs’ laboratory for species identification based on morphological characteristics (such as head, length of body, length of tail, hind foot, ear, fur and weight, etc.) [16]. Then, small mammals were anesthetized by ether, the parasitic fleas on their body also were anesthetized and collected using a toothbrush to comb [17]. The fleas were stored in 75% ethanol for preservation and later identification under a light microscope based on the characteristic of fleas [18].
Statistical Analysis
Data were encoded and digitized using the EpiData software(3.1 version) and analyzed using the R software(4.0.2 version) [19]. Information on small mammals and parasitic fleas was summarized using descriptive statistical methods. The proportion of each species of small mammals and parasitic fleas was determined as the proportion of captured individuals to the total number of captures: (Dr)=Ni/N×100, where Ni represents the number of individuals of that species captured and N represents the total number of individuals (of all species) captured [20,21]. A proportion of more than 10% was considered to indicate “dominant” species, 1% to 10% “common” species, and less than 1% “rare” species. The following international definitions of various parasitic indicators were adopted [22,23]: flea prevalence (%) = (number of fleainfected small mammals/total number of small mammals captured) ×100, and flea index = (total number of fleas/total number of small mammals captured). Commonly used Chinese definitions were used for the capture efficiency rate and small mammal density, as follows: capture efficiency (%) = (number of collected traps/number of traps placed)×100, small mammal density (%) = (number of small mammals captured/number of collected traps)×100 [24].
The capture efficiency rate and flea prevalence in different focus were compared by the Chi-squared test. The compositions of small mammal species and parasitic fleas in different focus were compared by the Fisher’s exact test. The value of P<0.05 was considered to indicate statistical significance.
Results
In this present study, 10,120 traps were placed and 9,937 were recovered, with a capture efficiency rate of 98.20%. A total of 1953 small mammals were captured, the total small mammals captured density was 19.65%. The small mammals represented 24 species, 14 genera, 6 families, and 3 orders, of which 40.86% were Apodemus chevrieri, 25.40% were Eothenomys miletus, 14.50% were Apodemus draco, and 2.97% were Eothenomys proditor. In addition, 514 small mammals were infected by fleas, the total flea prevalence was 26.32%. Overall, 1102 parasitic fleas were collected, the total flea index was 0.56. The parasitic fleas represented 11 species, 7 genera, 3 families, and 6 subfamilies, of which 30.94% were Neopsylla specialis specialis, 37.48% were Ctenophthalmus (Sinoctenophthalmus) quadrates, 6.90% were Macrostylophora (M.) euteles, and 5.35% were Ctenophthalmus (Sinoctenophthalmus) yunnanus. The difference in capture efficiency rate between the Yulong and Jianchuan plague focus was not significant (Chi-squared test, P=0.1258). The small mammal density in the Jianchuan plague focus was significantly higher than that in the Yulong plague focus (Chi-squared test, P<0.01).
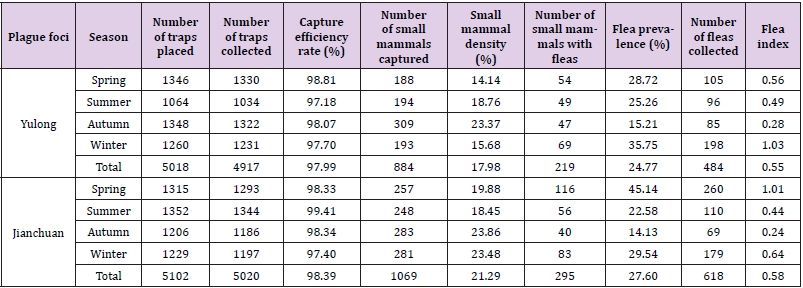
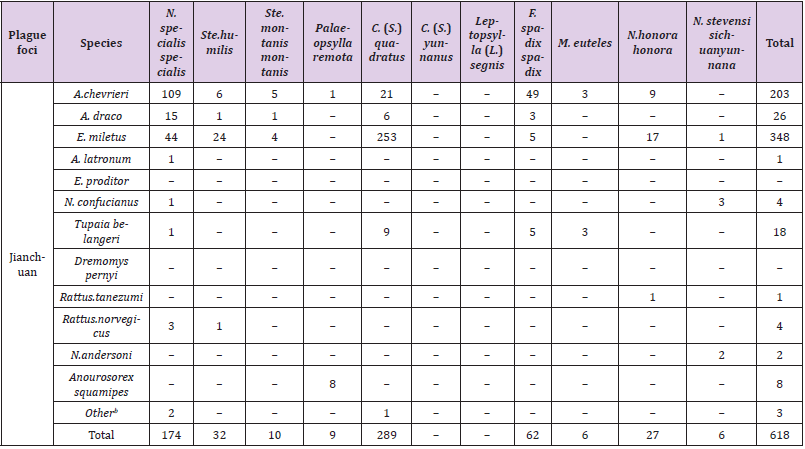
Table 1 showed the capture of small mammals and parasitic fleas in the Yulong and Jianchuan plague foci during the four seasons. Overall, 884 small mammals were captured and 484 parasitic fleas were collected in the Yulong plague focus, with the total flea prevalence of 24.77%, the flea prevalence and flea index were highest in winter (198, 35.75%, and 1.03 respectively) and lowest in autumn (85, 15.21%, and 0.28, respectively). By comparison, 1069 small mammals were captured and 618 parasitic fleas were collected in the Jianchuan plague focus. The total flea prevalence was 27.60%, the flea prevalence and flea index were highest in spring (260, 45.14%, and 1.01, respectively) and lowest in autumn (69, 14.13%, and 0.24, respectively). The difference in total flea prevalence among small mammals in the two plague foci were not significant (Chi-squared test, P=0.159). The numbers of seasonal composition on small mammals and parasitic fleas in the two plague foci were significantly different, respectively. (small mammals, Chi-squared test, P<0.01, parasitic fleas, Chi-squared test, P<0.05).
Table 1: Small mammals and parasitic fleas captured in the Yulong and Jianchuan plague focus in the Yunnan province.
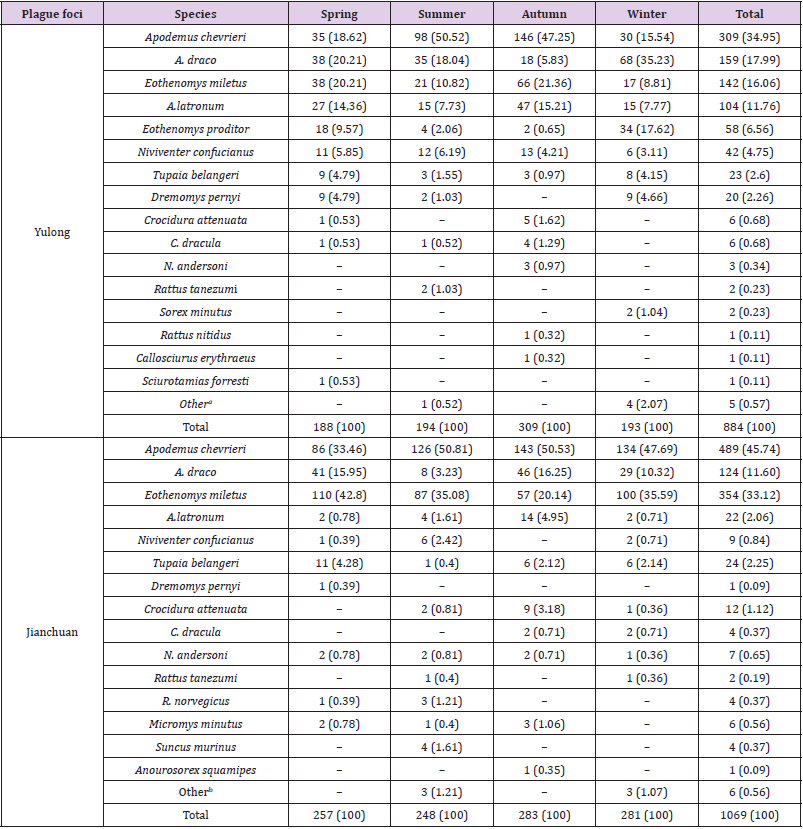
Table 2 showed the specific composition and distribution of small mammals in the Yulong and Jianchuan plague foci during four seasons. 18 species of small mammals were captured in the Yulong plague focus, representing 14 genera, 6 families, and 3 orders. A. chevrieri (34.95%), A. draco (17.99%), E. miletus (16.06%), and A latronum (11.76%) were the dominant species. 17 species of small mammals were captured in the Jianchuan plague focus, representing 10 genera, 5 families, and 3 orders. A. chevrieri (45.74%), E. miletus (33.12%), and A. draco (11.60%) were the dominant species. Further, 58 specimens of E. proditor were captured in the Yulong plague focus (6.56% of all specimens) but not captured in the Jianchuan focus. During the four seasons, the total numbers of first-three dominant species of small mammals (A. chevrieri, A. draco, E. miletus) in the Yulong and Jianchuan plague focus were significantly different (Chi-squared test, P<0.01). In addition, in the Yulong plague focus, the majority of small mammals (309) were captured in autumn (34.95% of all small mammals captured in Yulong focus), but the numbers of small mammals captured during the other three seasons were not much difference. By contrast, the numbers of small animals captured in the Jianchuan plague focus during the four seasons were similar
Table 2: Seasonal distribution of small mammals in the Yulong and Jianchuan plague focus in the Yunnan province [n (%)].
Note: aFour specimens of Soriculus leucopus in winter and one specimen of Vernaya fulva in summer. bTwo specimens of Eothenomys melanogaster and one specimen of Rattus rattus/sladeni in summer; one specimen of Deficiency in winter.
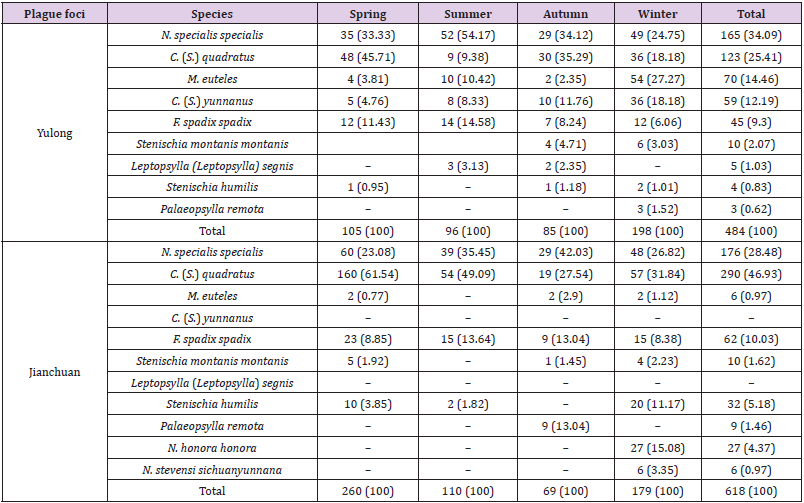
Table 3 showed the composition and distribution of parasitic fleas in the Yulong and Jianchuan plague foci during four seasons. 9 species of parasitic fleas were collected in the Yulong plague focus, representing 7 genera, 3 families, and 6 subfamilies. N. specialis specialis (34.09%), C. (S.) quadrates (25.41%), M. euteles (14.46%), and C. (S.) yunnanus (12.19%) were the dominant parasitic fleas. By contrast, 9 species of parasitic fleas were collected in the Jianchuan plague focus, representing 6 genera, 3 families, and 5 subfamilies. C. (S.) quadrates (46.93%), N. specialis specialis (28.48%), and Frontopsylla (F.) spadix spadix (10.03%) were the dominant parasitic fleas. C. (S.) yunnanus was not collected in Jianchuan focus. The numbers of dominant species of parasitic fleas (N. specialis specialis, C. (S.) quadrates, M. euteles, and C. (S.) yunnanus) in the Yulong and Jianchuan plague focus were significantly different in all seasons (Fisher’s exact test, P<0.01). Furthermore, in the Yulong plague focus, the majority of parasitic fleas (198) were collected in winter (40.91% of overall fleas in Yulong focus), and the numbers collected during the other three seasons were not much difference. In the Jianchuan plague focus, the majority of parasitic fleas (260) were collected in spring (42.07% of overall fleas in Jianchuan focus), with the second highest collection in winter (28.96% of overall fleas in Jianchuan focus). The numbers collected during the other two seasons were not much difference.
Table 3: Seasonal distribution of parasitic fleas in the Yulong and Jianchuan plague focus in the Yunnan province [n (%)].
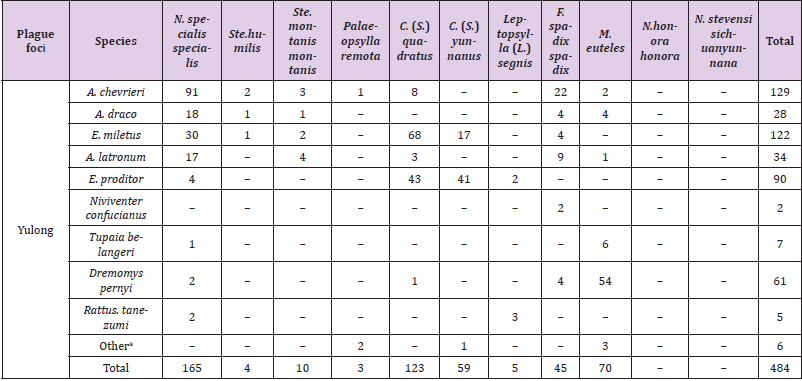
Table 4 showed the combinations distribution of each small mammal species infected per parasitic flea species in the Yulong and Jianchuan plague foci. 3 species of small mammals had the most parasitic fleas in the Yulong plague focus: A. chevrieri (129), E. miletus (122), and E. proditor (90). Two species of small mammals had the most parasitic fleas in the Jianchuan plague focus: A. chevrieri (203) and E. miletus (348). The most dominant parasitic fleas were N. specialis specialis and C. (S.) quadrates in the two plague foci. Simultaneously, N. specialis specialis was most frequently found on A. chevrieri, and C. (S.) quadratus was most frequently found on E. miletus. However, in the Yulong plague focus, the most frequent parasitic flea was N. specialis specialis (165), followed by C. (S.) quadrates (123). The opposite was the case in the Jianchuan plague focus, with 289 specimens of C. (S.) quadrates and 174 specimens of N. specialis specialis. Of note, 59 specimens of C. (S.) yunnanus were collected in the Yulong plague focus but not in the Jianchuan, and 70 specimens of M. euteles were collected in the Yulong plague focus but only 6 were collected in the Jianchuan plague focus (Table 5).
Table 4: Distribution of parasitic fleas on small mammals in the Yulong and Jianchuan plague focus in the Yunnan province.
Note: aTwo specimens of Palaeopsylla remota on Sorex minutus; one specimen of Ctenophthalmus (Sinoctenophthalmus) yunnanus on Sorex minutus; two specimens of Macrostylophora euteles on Callosciurus erythraeus and one on Sciurotamias forresti.
Table 5: Distribution of parasitic fleas on small mammals in the Yulong and Jianchuan plague focus in the Yunnan province (Continued).
Note: bTwo specimens of Neopsylla specialis specialis on Micromys minutus and one on Suncus murinus; one specimen of Ctenophthalmus (Sinoctenophthalmus) quadratus on Suncus murinus.
Discussion
In this study, we analyzed the composition and distribution of small mammals and parasitic fleas in two plague foci in the Yunnan Province. A. chevrieri was the dominant species of small mammals in the two focal areas, most frequently captured in autumn. N. specialis specialis was the dominant parasitic flea in the Yulong plague focus, most collected in summer. C. (S.) quadratus was the dominant parasitic flea in the Jianchuan plague focus, most collected in spring. It suggests that the two foci have the same dominant host, but the dominant flea species are slightly different. The results are consisted with previously studies, Zhang et al found that N. specialis specialis was the dominant parasitic flea in the Yulong plague focus by 2006-2017 surveillance data and Guo et al found C. (S.) quadratus was the dominant parasitic flea in the Jianchuan plague focus. The main flea difference in the two foci, however, which may be caused by their independent ecological circle [9,25]. This also implied that Yulong focus has a different composition of small mammals and fleas even adjacent to Jianchuan focus, further we need to effort long-term monitoring data to determine whether this composition is accidental or long-term, which to confirm whether impact occurrence of plague.
Furthermore, the flea index in Yulong focus was the most in winter but Jianchuan focus reached the peak in spring, implying small mammals in winter of Yulong focus and in spring of Jianchuan focus had higher average number of parasitic fleas on per small mammal individuals may contribute to higher risk for the occurrence of plague than that in other seasons. A. chevrieri was the dominant species of small mammals in the two foci, most frequently captured in autumn. A. chevrieri, A. draco, and E. miletus account for most small mammals in the two plague foci. The results are consisted with previously studies, A. chevrieri and E. miletus are dominant species of small mamamls in the two focal areas [26]. Additionally, it is worth noting that A. draco was the dominant species, the captured number was even more than those in E. miletus in Yulong focus, previously studies showed that A. draco is an invasive species but E. miletus is the native species in the middle of the Hengduan Mountains, in order to adapt and survive, A. draco needs to constantly adjust its physiological characteristics such as basal metabolic rate [27].
In this study, the mostly increased number of A. draco shows maybe its further adapted well to the geographical climate of the Hengduan Mountains, but whether the increase in number will increase the occurrence of animal plague requires long-term monitoring and judgment. Furthermore, E. proditor was captured in the Yulong plague focus but not in the Jianchuan plague focus, it might be a secondary host or a popular host, but not be as the dominant host in the Yulong focus. We also observed some seasonality in species distribution, with the dominant species most abundant in autumn, which may be related to the fall grain harvest. The maturity of food in autumn can provide an ideal habitat and food source for the small mammals, leading to the massive reproduction of small mammals. In general, there were a certain variety similarity between the two plague foci, which A. chevrieri, A. draco, and E. miletus were accounted for most, however, there were obviously differences in the dominant fleas and some small mammals like C. (S.) yunnanus and E. proditor may be associated with geography, local climate, and human interference.
The two main species of parasitic fleas collected in the two plague foci were C. (S.) quadratus and N. specialis specialis. N. specialis specialis was the dominant parasitic flea in the Yulong plague focus but C. (S.) quadratus was the dominant parasitic flea in the Jianchuan plague focus. The results consisted with previously studies [25,28]. Further, while 358 specimens of the top-three species of parasitic fleas (N. specialis specialis, C. (S.) quadratus, and M. euteles) in the Yulong plague focus accounted for 73.96% of all parasitic fleas collected in Yulong focus, they were slightly more prevalent in the Jianchuan plague focus, accounting for 76.38% of all parasitic fleas in Jianchua foucus. In addition, C. (S.) yunnanus species only were collected in the Yulong plague focus, and mostly parasite on E. proditor. Although the dominant parasitic fleas and seasonal distribution of parasitic fleas in the two plague foci were different, in general, the total representation species were similar. That may be because the two foci formed a small independent environment, respectively.
We observed seasonality of parasitic flea abundance, with the dominant flea species in the Yulong plague focus reaching a peak in winter, and the dominant flea species in the Jianchuan plague focus reaching a peak in spring. The parasitic fleas in the Yulong and Jianchuan plague focus showed different seasonality, indicating that a seasonal variation in vectors may be closely related to the local climate, which is in line with the seasonal distribution of plague in the Yunnan province. In addition to acceleration of other anthropogenic changes, acceleration of the climate change is an important factor in the current rise in the spread of infectious diseases and the risk of global outbreaks [29-31]. Simultaneously, while Y. pestis is essential for plague occurrence, the bacterium only establishes itself in areas with a suitable landscaping and climatic conditions, with sufficient small mammals and parasitic fleas to allow epizootic transmission [32]. Recent studies have identified the different vegetation, climate variables and landscape of the plague focus, and to define regions that the risk will increase when human exposure to plague bacteria in the plague focus [33-40].
The main predictors of plague outbreaks in Uganda and Tanzania are elevation and vegetation, as revealed by local spatialscale studies [41]. The growth of vegetation, precipitation and climatic variables are varying with the season changing, this could indicate that the seasonal variation of host animals and vectors may be closely related to the local climate and precipitation, and these factors are closely linked to the local geographical environment in the Yulong and Jianchuan plague foci. The two plague foci are located close to one another. However, while similar geographical environment may result in a similar small mammal composition, the difference in climate may lead to differences in small mammal density. Further, the vegetation in the Yulong plague focus is mainly alpine pine forest or mixed forest. Because of the complex environment in the area, it has its own special climate, and the temperature and humidity changes are little, protecting small mammals from the cold. The vegetation is denser providing abundant food for small mammals. Additionally, the average altitude in Yulong focus is higher than Jianchuan focus, human interference in high-altitude areas with rich vegetation, and high diversity and evenness indices is relatively low, with obvious boundary effects and community stability [21].
Hence, the Yulong plague focus hosted more small mammal species than the Jianchuan plague focus. Further, high altitude and low population density may have created a suitable environment for small mammals to live and reproduction. The difference of altitude and the low-lying valley in the center of the two focal areas may have been led to the formation of unique environment in the two plague foci, with differences in precipitation. Precipitation is used to affects the growth of primary vegetation, thus influencing the potential food source of small mammals and habitat environment [42,43]. With an increasing or decreasing number of small mammals, flea infection rates rise or fall, accordingly. In addition, an increasing in soil moisture may increase the daily survival rate of fleas, increasing the likelihood of infectious fleas and the contact rate among susceptible hosts, thereby increasing the likelihood of the occurrence of plague [44,45]. The evidence of a link between meteorological variables, small mammal density, and risk of infection outbreaks is compelling [46].
Consequently, because of the difference in seasonal precipitation brought about by the respective unique environments, the parasitic fleas in the Yulong and Jianchuan plague focus were different. The observation that C. (S.) yunnanus (mainly on E. proditor) specimens were collected in the Yulong plague focus (mainly in winter) and not in the Jianchuan plague focus, indicated that this species may be the dominant species of E. proditor, but this notion requires further research. In the two plague foci, more specimens of M. euteles were collected in the Yulong plague focus than that collected in the Jianchuan plague focus. That could be because, first, M. euteles mainly parasitized Dremomys pernyi, predominantly captured in the Yulong plague focus, and, second, M. euteles might be better adapted to survival in the geographical landscape in the Yulong plague focus than that in the other focus. However, this also requires further research. Small mammals and parasitic fleas are the inevitable factors maintain Y.pestis in a plague focus, while the habitat/landscape, climate variables and pathogenic characteristics determine the properties and structures of the plague focus [47]. Guo, Song, Dong, et al reported that the Yulong plague focus is adjacent to the Jianchuan plague focus, almost forming a single focus. However, a Y. pestis strain isolated in the Yulong plague focus can utilize glycerin and maltose via glycolysis, which is quite different from that of the wild-type Y. pestis strain in the Yunnan province [10,48,49]. Further, Y. pestis strain which isolated from the Yulong is similar to that found on Marmota (Ma.) himalayana in Tibet, and the plague focus of Ma. himalayana and the Yulong plague focus are both located in mountainous regions [48].
Previously, we demonstrated that the Y. pestis strains in the Yulong plague focus are different from the Jianchuan plague focus strains, with a similar geographical landscape, and that the Yulong strains are highly similar to those in the Qinghai Tibetan Plateau plague focus [10,13,50-52]. Therefore, Y. pestis from the Yulong plague focus may have migrated from Ma. himalayana in Tibet via small mammals, and the difference in Y. pestis strains may have led to subtle differences in the properties and structures of the Yulong and Jianchuan plague focus.
This present study has certain strength. First of all, we compared the composition and seasonal distribution of small mammals and parasitic fleas in two similar and adjacent wild rodent foci, but found that although the two places are similar, the existence of species in each season is obviously different. This may be due to the two place is separated by a canyon geographically, which has led to climate change, but further research is still needed. Secondly, our research is different from previous studies on plague foci. We focus more on observing the population of small mammals and fleas in the four seasons, and we can find the number and changes of small mammals and fleas in different seasons. This also provides a scientific basis for local government to effectively implement small mammals and flea control. We also have certain limitations, however, because we are a cross-sectional study, we cannot prove the causality. Secondly, we have only investigated for a year, and the conclusions we got need more time to monitor to be corroborated. This time we only roughly divided into four seasons to collect small mammals and parasitic fleas, but did not carefully analyze the differences in temperature, humidity, and food in each season. These may affect the composition of small mammals and fleas. Our team next step will consider take these factors into further analysis.
Conclusion
The dominant species of small mammals in the two plague foci were identical, with a similar variety of species, suggesting similar small mammal composition in the two foci. A. chevrieri, A. draco, and E. miletus were the dominant small mammals in the two plague foci. In Yulong focus, A. draco thrives better and E. proditor was only a secondary host or popular host. The dominant species of parasitic fleas were different in the two plague foci, but the total variety of species was similar, N. specialis specialis and C. (S.) quadratus were the common dominant vectors. From epidemiological perspective, Yulong focus is adjacent to Jianchuan focus, but there are differences of composition and distribution on small mammals and parasite fleas, these indicating that complicated and uncertainty on wild rodents plague focus, We need to fully understand the proportions and structure of the Yulong focus, especially the distribution of small mammals and parasite fleas may contribute to prevent and control the spread from animal plague to humans.
Ethics Statement
The study has been approved the Medical Ethics Committee of Dali University and performed in accordance with the Rules for Implementation of Laboratory Animal Medicine (1998) from the Ministry of Health, China. The captured of small mammals and parasitic fleas were allowed and no painful.
Consent for publication
Not available.
Availability of Data and Materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Competing Interests
The authors declare that they have no competing interests.
Funding
The study was supported by the National Natural Science Foundation of China (No. 81860565; No. 81460485), The Scientific and Technological Innovation team of Natural Focal Diseases Epidemiology in Universities of Yunnan province, The Innovation Team of Natural Focal Diseases Epidemiology in Dali University (No. ZKPY2019201). The funders had no role in the study design, data collection and analysis, decision to publish or preparation of the manuscript.
Acknowledgement
The author would like to express thankfulness to Jianchuan Station for Endemic Disease Control and Prevention and Yulong Center for Disease Control and Prevention.
Author’s Contribution
JXY conceived the project and obtained the funding, QFZ, ZFW, DDX, MDW, YZ ZXL, MH and RDH collected the data, YYL, QFZ, STL and QNH analyzed the data, YYL and QFZ wrote the paper, JXY revised the paper. All authors read and approved the final manuscript.
References
- Xu L, Stige LC, Kausrud KL, Ben AT, Wang S, et al. (2014) Wet climate and transportation routes accelerate spread of human plague. Proc Biol Sci 281(1780): 20133159.
- Stenseth NC, Atshabar BB, Begon M, Belmain SR, Bertherat E, et al. (2008) Plague: past, present, and future. PLoS medicine 5(1): e3.
- English AI (2005) Plague. Wkly Epidemiol Rec 80(15): 138-140.
- Vallès X, Stenseth NC, Demeure C, Horby P, Mead PS, et al. (2020) Human plague: An old scourge that needs new answers. PLoS neglected tropical diseases 14(8): e0008251.
- VK N, C P-R, EA H-V (2018) The 2017 plague outbreak in Madagascar: Data descriptions and epidemic modelling. Epidemics 25: 20-25.
- Wang X, Wei X, Song Z, Wang M, Xi J, et al. (2017) Mechanism study on a plague outbreak driven by the construction of a large reservoir in southwest china (surveillance from 2000-2015). PLoS neglected tropical diseases 11(3): e0005425.
- Yin JX, Geater A, Chongsuvivatwong V, Dong XQ, Du CH, et al. (2008) Predictors for presence and abundance of small mammals in households of villages endemic for commensal rodent plague in Yunnan Province, China. BMC ecology 8: 18.
- Luo H, Dong X, Li F, Xie X, Song Z, et al. (2013) A Cluster of Primary Pneumonic Plague Transmitted in a Truck Cab in a New Enzootic Focus in China. American Journal of Tropical Medicine & Hygiene 88(5): 923-928.
- Guo Y, Gao Z, Duan C, Zhang H, Wang P (2019) Composition and risk analysis of hosts and vectors in new wild plague foci of Jianchuan county, Yunnan province, China. Chin J Vector Biol & Control 30(01): 31-34.
- Song ZZ, Xia LX, Liang Y (2008) Confirmation and study of Plague Natural Foci for Yulong County and Guchengqu in Yunnan Province. Chinese Journal of Control of Endemic Diseases 23(1): 3-7.
- Qin CY, Xu L, Zhang RZ, Liu QY, Li GC, et al. (2012) Eco-geographic landscapes of natural plague foci in China v. biological characteristics of major natural reservoirs of Yesinia pestis. Zhonghua liu xing bing xue za zhi = Zhonghua liuxingbingxue zazhi 33(7): 692.
- Koshel EI, Aleshin VV, Eroshenko GA, Kutyrev VV (2014) Phylogenetic Analysis of Entomoparasitic Nematodes, Potential Control Agents of Flea Populations in Natural Foci of Plague. Biomed Res Int 2014(1): 135218.
- Wang P, Shi L, Zhang F, Guo Y, Zhang Z, et al. (2018)Ten years of surveillance of the Yulong plague focus in China and the molecular typing and source tracing of the isolates. PLoS neglected tropical diseases 12(3): e0006352.
- Zhang CG, Gao ZH, Song ZZ (2015) The research advancement on the structure and property of Yulong natural plague foci. China Tropical Medicine 015(012): 1531-1533.
- Cai WF, Luo JL, Su LQ, Zhang SX, Li SQ (2019) Community diversity characteristics of small mammals in plage natural focus of Jianchuan County, Yunnan, 2008-2017. China Tropical Medicine 19(05): 440-443.
- Zheng ZM, Jiang ZK, Chen AG (2008) GLIRES. Shanghai Jiaotong University Press.
- Yin JX, Cheng XO, Luo YY, Zhao QF, Wei ZF, et al. (2020) The relationship between fleas and small mammals in households of the Western Yunnan Province, China. Scientific reports 10(1): 16705.
- Xie BQ, Zeng JF (2000) THE SIPHONAPTERA OF YUNNAN. YUNNAN SCIENCE AND TECHNOLOGY PRESS.
- Gentleman R, Huber W, Carey VJ (2011) R Language. Springer Berlin Heidelberg.
- Tang L, Zhang CG, Shao ZT, Duan XD, En-Nian PU, et al. (2017) The community structures and species diversity of fleas in Yulong plague focus,Yunnan province. Chinese Journal of Vector Biology & Control.
- Liu JY, Hong DU, Tian GB, Pin-Hong YU, Wang SW, et al. (2008) Community Structure and Diversity Distributions of Small Mammals in Different Sample Plots in the Eastern Part of Wuling Mountains. Zoological Research 29(6): 637-645.
- Yin JX, Geater A, Chongsuvivatwong V, Dong XQ, Du CH, et al. (2011) Predictors for abundance of host flea and floor flea in households of villages with endemic commensal rodent plague, Yunnan Province, China. Plos Neglected Tropical Diseases 5(3): e997.
- Ko S, Kim HC, Yang Y-C, Chong S-T, Richards AL, et al. (2011) Detection of Rickettsia felis and Rickettsia typhi and Seasonal Prevalence of Fleas Collected from Small Mammals at Gyeonggi Province in the Republic of Korea. Vector-Borne and Zoonotic Diseases 11(9): 1243-1251.
- Liu Q, Lu L, Meng F, Guo T, Huo X, et al. (2009) Surveillance methods for vector density-Rodent.
- Zhang ZF, Zhang FX, Yand-Tian HE, Chen ZJ, Chen FM, Gan YZ, et al. (2012) Research on the six‐year monitoring and control measures in epidemic wild rodent plague foci in Lijiang. Foreign Medical Sciences (Section of Medgeography) 33(03): 154-158.
- Li GC, Cai WF, Zhang FX, Lu L, Liang Y, Wang GL, et al. (2009) Surveilance on the host and vector in Yulong plague focus in Yunnan Province. Disease Surveilance 24(2): 114-117.
- Li XT, Wang R, Wang P, Meng LH, Liu CY, Wang ZK, et al. (2009) Thermoregulation and evaporative water loss in Apodemus draco from the Hengduan Mountains region. Acta Theriologica Sinica 29(3): 302-309.
- Wei ZF, Yin JX (2015) Research of natural plague foci in Yunnan Province. Bull Dis Control Prev 30(6): 84-86.
- Karesh WB, Dobson A, Lloyd-Smith JO, Lubroth J, Dixon MA, et al. (2012) Ecology of zoonoses: natural and unnatural histories. Lancet (London, England) 380(9857): 1936-1945.
- Gubler DJ, Reiter P, Ebi KL, Yap W, Nasci R, et al. (2001) Climate Variability and Change in the United States: Potential Impacts on Vector- and Rodent-Borne Diseases. Environmental Health Perspectives 109 Suppl 2(2): 223.
- Kilpatrick AM, Randolph SE (2012) Drivers, dynamics, and control of emerging vector-borne zoonotic diseases. Lancet (London, England) 380(9857): 1946-1955.
- Eisen RJ, Gage KL (2012) Transmission of Flea-Borne Zoonotic Agents*. Annual Review of Entomology. 57(57): 61-82.
- Collinge SK, Johnson WC, Ray C, Matchett R, Grensten J, et al. (2005) Landscape Structure and Plague Occurrence in Black-tailed Prairie Dogs on Grasslands of the Western USA. Landscape Ecology 20(8): 941-55.
- Davis S, Klassovskiy N, Ageyev V, Suleimenov B, Atshabar B, et al. (2007) Plague metapopulation dynamics in a natural reservoir: the burrow system as the unit of study. Epidemiology & Infection 135(5): 740-748.
- Eisen RJ, Enscore RE, Biggerstaff BJ, Reynolds PJ, Ettestad P, et al. (2007) Human plague in the southwestern United States, 1957-2004: spatial models of elevated risk of human exposure to Yersinia pestis. Journal of Medical Entomology 44(3): 530.
- Eisen RJ, Glass GE, Eisen L, Cheek J, Enscore RE, et al. (2007) A Spatial Model of Shared Risk for Plague and Hantavirus Pulmonary Syndrome in the Southwestern United States. American Journal of Tropical Medicine & Hygiene77(6): 999-1004.
- Eisen RJ, Griffith KS (2010) Assessing human risk of exposure to plague bacteria in Northwestern Uganda based on remotely sensed predictors. American Journal of Tropical Medicine & Hygiene 82(5): 904-911.
- Nakazawa Y, Williams R, Peterson AT, Mead P, Staples E, et al. (2007) Climate change effects on plague and tularemia in the United States. Vector Borne & Zoonotic Diseases 7(4): 529-540.
- Neerinckx SB, Peterson AT, Gulinck H, Deckers J, Leirs H (2008) Geographic distribution and ecological niche of plague in sub-Saharan Africa. International Journal of Health Geographics 7(1): 54.
- Winters AM, Staples JE, Ogenodoi A, Mead PS, Griffith K, et al. (2009) Spatial risk models for human plague in the West Nile region of Uganda. American Journal of Tropical Medicine & Hygiene 80(6): 1014-1022.
- Neerinckx S, Peterson AT, Gulinck H, Deckers J, Kimaro D, et al. (2010) Predicting potential risk areas of human plague for the Western Usambara Mountains, Lushoto District, Tanzania. American Journal of Tropical Medicine & Hygiene 82(3): 492-500.
- Eisen RJ, Holmes JL, Schotthoefer AM, Vetter SM, Montenieri JA, et al. (2008) Demonstration of Early-Phase Transmission of Yersinia pestis by the Mouse Flea, Aetheca wagneri (Siphonaptera: Ceratophylidae), and Implications for the Role of Deer Mice as Enzootic Reservoirs. Journal of Medical Entomology 45(6): 1160-1164.
- Parmenter RR, Yadav EP, Parmenter CA, Ettestad P, Gage KL (1999) Incidence of plague associated with increased winter-spring precipitation in New Mexico. American Journal of Tropical Medicine & Hygiene. 61(5): 814-821.
- Eisen RJ, Bearden SW, Wilder AP, Montenieri JA, Antolin MF, et al. (2006) Early-phase transmission of Yersinia pestis by unblocked fleas as a mechanism explaining rapidly spreading plague epizootics. Proceedings of the National Academy of Sciences of the United States of America 103(42): 15380-15385.
- Lorange EA, Race BL, Sebbane F, Hinnebusch BJ (2005) Poor Vector Competence of Fleas and the Evolution of Hypervirulence in Yersinia pestis. Journal of Infectious Diseases 191(11): 1907-1912.
- Stapp P (2007) Trophic Cascades and Disease Ecology. Ecohealth 4(2): 121-124.
- Zhang CG, Gao ZH, Song ZZ (2015) The research advancement on the structure and property of Yulong natural plague foci. China Tropical Medicine 15(12): 1531-1533.
- Dong XQ, Song ZZ, Liang Y, Wang GL, Yu GL, Ma YK (2009) The Epidemic Situation and Neighbour Relationship of Wild Rodent Plague in Yunnan Province. Journal of Kunming Medical University 30(8): 21-25.
- Guo Y, Wei DX, Liang Y, Su P, Yang ZM, Dong SS, et al. (2009) Comparative study on biochemical characteristic of Yersinia pestis from some natural focuses of plague in Yunnan Province 28(4): 373-375.
- Shen X, Wang Q, Xia L, Zhu X, Zhang Z, et al. (2010) Complete Genome Sequences of Yersinia pestis from Natural Foci in China. Journal of Bacteriology192(13): 3551-3552.
- Zhang Z, Zhang E, Xia L, Cai H, Yu D, Hai R, et al. (2009) Spatial variation of Yersinia pestis from Yunnan Province of China. American Journal of Tropical Medicine & Hygiene 81(4): 714-717.
- Zhang X, Hai R, Wei J, Cui Z, Zhang E, et al. (2009) MLVA distribution characteristics of Yersinia pestis in China and the correlation analysis. BMC microbiology 9(1): 205.

 Research Article
Research Article