Abstract
Background: Congenital anomalies of the urogenital system are some of the most common birth defects affecting children. The aim of this study was to evaluate the prevalence and types of congenital genitourinary anomalies seen in the 2 teaching hospitals in Enugu, Nigeria.
Materials and Methods: This was an observational study carried out on babies at the two teaching hospitals in Enugu. The newborns were examined for congenital genitourinary anomalies and the study covered a period of 5 years.
Results: There were 17,241 babies delivered in the 2 teaching hospitals during the study period. Overall, 328 babies had at least one congenital anomaly which gave an overall incidence of all congenital anomalies of 1.9% or 19 babies per 1000 live births. Out of the 328 babies that had congenital anomalies, 93 babies had a genitourinary anomaly. This gave an incidence of 0.5% or 5 babies per 1000 live births. There was female predominance and majority of the babies were preterm and weighed less than 2.5 kilograms. Hypospadias was the most common genitourinary anomaly and only a few of the anomalies were diagnosed prenatally. Half of the mothers took herbal concoction during their pregnancy and about one-fifth of them had gestational diabetes/ hypertension.
Conclusion: Genitourinary anomaly is a common birth defect that is may be associated with some morbidity. Hypospadias is the most common abnormality in the present study and half of the mothers took herbs.
Keywords: Anomalies; Congenital; Genitourinary; Hypospadias, Hospital
Abbreviations: UNTH: University of Nigeria Teaching Hospital; ESUTH: Enugu and Enugu State University Teaching Hospital; SPSS: Statistical Package for Social Science
Introduction
Congenital genitourinary anomalies represent 35-45% of birth defects and include a wide range of structural abnormalities of the urinary and reproductive tracts, whose collective occurrence reflects their embryologic origin [1,2]. Congenital anomalies of the urogenital system are some of the most common birth defects affecting newborns and encompass a wide range of structural abnormalities of the kidneys, collecting systems, urinary bladder, urethra, testes, scrotum and penis [3]. The clinical spectrum of congenital genitourinary anomalies ranges from minor anomalies such as glanular hypospadias to severe conditions such as cloacal exstrophy: It may be isolated or associated with other anatomic defects (syndromic) [4]. The etiology may be genetic or non-genetic, or may be unknown [5]. There are wide variations in the incidence of congenital anomalies of the urogenital system due to clinical heterogeneity, use of different inclusion criteria and coding [4]. Severe congenital genitourinary anomalies may be associated with stillbirths and spontaneous abortions [4]. Impaired renal function is a common sequel of congenital urinary anomalies resulting from kidney malfunction [4]. Prenatal diagnosis congenital genitourinary anomaly provides an opportunity for prenatal intervention and prompt treatment in the early postnatal period. Congenital urogenital abnormalities are clinical and epidemiological challenge; its management should be multidisciplinary due to the complex surgical, endocrine, genetic, social, ethical and psychological issues [4,6]. The aim of this study was to evaluate the prevalence and types of observable congenital genitourinary anomalies seen in the 2 teaching hospitals in Enugu, Nigeria.
Materials and Methods
This was an observational study carried out at the two teaching hospitals in Enugu, namely: University of Nigeria Teaching Hospital (UNTH) Enugu and Enugu State University Teaching Hospital (ESUTH), Enugu, South East, Nigeria. The hospitals serve the whole of Enugu State, which according to the 2016 estimates of the National Population Commission and Nigerian National Bureau of Statistics, has a population of about 4 million people and a population density of 616.0/km2. The hospitals also receive referrals from its neighboring states. Ethical approval was obtained from the ethics and research committees of the 2 teaching hospitals and informed consent was obtained from the patients’ mothers. For the purposes of this study, our interest was on clinically obvious and observable abnormality of genital and urinary systems which was present at birth or noticed a few days after birth. All the live babies born in UNTH and ESUTH during the period of this study were included. Stillborns were excluded from this study. This study covered a period of 5 years, from January 2014 to December 2018. The protocol of this study conforms to the declaration of Helsinki. All the consecutive children who were born in both teaching hospitals during the study period had a thorough physical examination (general and systemic) performed by a pediatrician and a pediatric surgeon at birth and at 6 weeks postnatal visit. This 2-stage physical examination minimized the error of missing any congenital anomalies of the genitourinary system. Diagnosis of congenital anomalies of the urogenital system was based only on clinical evaluation of the babies by the pediatrician and pediatric surgeon. Investigations such as radiography, ultrasonography, and computed tomography scan and magnetic resonance imaging of the abdomen and pelvis were not performed.
For each patient, the following data were collected: gender, age of the baby at the time of diagnosis, maternal age, gestational age of the pregnancy before delivery (term/preterm), baby’s birth weight and mode of delivery. Baby’s birth weight greater or equal to 2.5 kilograms (kg) were considered to be normal while birth weight less than 2.5kg were considered as low birth weight. Babies born at less than 37 completed weeks, calculated from the first day of last menstrual period, were considered preterm while babies born at or after 37 completed weeks were considered term. Other data collected include family history of congenital anomaly, maternal comorbidities such as diabetes/hypertension and use of drugs and herbs.
Data Analysis
Statistical Package for Social Science (SPSS) for Windows version 23 (IBM Corp., Armonk, NY) was used for data entry and analysis. Data were expressed as percentages, medians and means.
Results
Patents’ Demographics
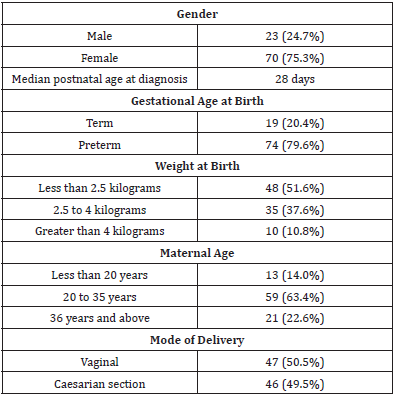
There were 17,241 babies delivered in the 2 teaching hospitals during the study period; 11,494 babies were delivered in ESUTH (61 babies had an anomaly of the urogenital system) while 5,747 babies were delivered in UNTH (32 babies had an anomaly of the urogenital system). Overall, 328 babies had at least one congenital anomaly which gave an overall incidence of all congenital anomalies of 1.9% or 19 babies per 1000 live births. Out of the 328 babies that had congenital anomalies, 93 babies had a genitourinary anomaly. This gave an incidence of 0.5% or 5 babies per 1000 live births. Considering the congenital anomalies system wise, congenital anomalies of the urogenital system accounted for 27.8% of all the systems. Other demographic features are shown in Table 1.
Distribution of Congenital Genitourinary Anomalies
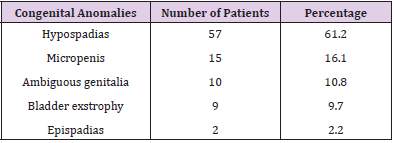
These are shown in Table 2.
Possible Associated Risk Factors of Genitourinary Anomalies
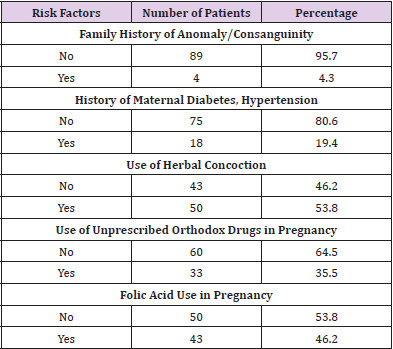
Possible associated risk factors are depicted in Table 3.
Prenatal Diagnosis of Congenital Urogenital Anomalies
Nineteen (20.4%) patients had their genitourinary anomaly diagnosed prenatally using maternal ultrasound scan.
Discussion
Embryologically, the urinary and genital systems are closely related so dividing them into separate entities is often artificial [7]. The genitourinary system arises from the intermittent mesoderm which forms the urogenital ridge on either side of the aorta. From the urogenital ridge, the urinary system and genitals are formed through the process of epithelial-mesenchymal induction [8]. Congenital anomalies are a global health problem and contribute significantly to childhood mortality and hospital admissions [9]. Congenital anomalies are the leading cause of mortality and morbidity especially in developing countries [9,10]. The wide range of causes of birth defects means that prevention approaches is needed. However, preventive measures are available in only about 60% of the cases [11]. In the present study, the general incidence of all congenital anomalies was 1.9% or 19 babies per 1000 live births. This is comparable to the report of Sarkar, et al. [12]. However, it is at variance to the report of other series on congenital anomalies [10,13]. The differences in incidence may be due to the cohort of patients recruited by the different studies. For instance, the studied population may be live births, still births, patients on hospital admission or population based. In the current study, the prevalence of genitourinary anomalies was 0.5%. This is similar to the findings of Dolk, et al. [14]. Howbeit, variable prevalence of genitourinary anomalies has been reported [10,12,13]. The setting of the studies and the investigations performed may affect the reported prevalence of specific genitourinary anomalies. For instance, posterior urethral valve which is a congenital urinary anomaly can only be confirmed by investigation.
More females were affected in the index study. Li et al and Abdou et al, however, documented male predominance with regards to congenital genitourinary anomalies [15,16]. The reason for the gender difference is not known. The specific type of urogenital anomaly may determine the gender that is mostly affected. For example, hypospadias occur only in males. Twenty-eight days was the median age at diagnosis of genitourinary anomaly in the current study. One study from Ibadan, Nigeria reported that about 50% of the congenital genitourinary abnormalities were diagnosed at neonatal age [17]. The severity and obvious nature of the abnormality may determine when it is diagnosed. About threequarters of our patients were delivered preterm and about 50% of the patients had low birth weight. Bhat et al reported that infertility treatment, increased maternal age and parity greater than 2 increased the risk of low birth weight and preterm birth resulting in a statistically significant increase in genitourinary anomalies [18]. Majority of the mothers whose babies had congenital genitourinary anomalies were between the ages of 20 and 35 years. Although other authors have reported advanced maternal age as a risk factor for increased incidence of congenital anomalies particularly of the renal and genitourinary systems [19,20]. Goetzinger, et al. reported that up to the age of 35 years, advanced maternal age is not associated with increased risk of congenital anomalies [21]. About half of the patients who had genitourinary anomalies were delivered vaginally. The choice of the method of delivery of babies with congenital anomalies may be based on obstetric grounds and the belief that one method of delivery offers the fetus some benefit over the other [22].
Hypospadias was the most common genitourinary abnormality recorded in the present study. Hypospadias is one of the most common congenital anomalies in males [23]. Other series on congenital anomalies also reported hypospadias as the most common anomaly [24,25]. However, Marshal et al and Braga et al reported undescended testis as the most common genitourinary anomaly [7,26]. Hypospadias is an arrested development of the urethra, foreskin, and ventral surface of the penis whereby the urethral opening may be anywhere along the shaft, within the scrotum or in the perineum. In the current study, there is no family history of urogenital anomalies in most of the patients. Although some forms of urogenital anomalies are part of the syndrome or are associated with a positive family history, most cases are sporadic and isolated [27]. Maternal hypertension and diabetes are known risk factors for congenital anomalies. Howbeit, only about onefifth of the mothers gave a history of hypertension or gestational diabetes. Mounika and Nitsch reported that up to 14% of congenital urinary anomalies could be eliminated if gestational diabetes is prevented [28]. Some non-prescribed orthodox drugs taken during pregnancy can have negative impacts on the outcome of the pregnancy. About one-third of the mothers in the present study gave a history of intake of non-prescribed drugs in early pregnancy. Offor et al. documented the impact of drug treatment of maternal illnesses during pregnancy in the etiology of congenital anomalies [29]. Only about one-fifth of the patients in the present study had a prenatal diagnosis of their pathology. However, Gupta et al reported that genitourinary anomalies comprise 30-50% of all anomalies detected on prenatal scan [30].
Conclusion
Genitourinary anomalies are a common birth defect that is may be associated with some morbidity. Hypospadias is the most common abnormality in the present study and half of the mothers accepted taking herbal concoction during their pregnancy. Awareness and early recognition of these genitourinary anomalies are required for prompt referral and treatment of these genitourinary anomalies.
Limitations of the Study
This was a hospital based observational study. Babies with urogenital anomalies delivered outside the teaching hospital were not captured. There is a small number of patients. A larger number would have availed better analysis. Only the clinically obvious urogenital anomalies were reported. Investigations such as ultrasound were not done.
References
- Patwardhan N, Woodward M, Nicholls G (2010) Disorders of Sex Development. In: Sinha C, Davenport M (Eds.)., Handbook of Pediatric Surgery: Springer, London, pp. 321-326.
- Connell M, Owen C, Segars J (2013) Genetic Syndrome and Genes Involved in the Development of the Female Reproductive Tract. A Possible Role for Gene Therapy. J Genet Syndr Gene Ther 4: 127.
- Li ZY, Chen YM, Qiu LQ, Chen DQ, Hu CG, et al. (2019) Prevalence, types and malformations in congenital anomalies of the kidney and urinary tract in newborns: a retrospective hospital based study. Ital J Pediatr 45(1): 50
- Gazzaneo IF, De Queiroz CM, Goes LC, Lessa VJ, De Omena Filho RL, et al. (2015) Profile of patients with genitourinary anomalies treated in a clinical genetics service in the Brazilian unified health system. Pediatria 34(1): 91-98.
- Hughes IA (2008) Disorders of sex development: a new definition and classification. Best Pract Res Clin Endocrinol Metab 22(1): 119-134.
- Guerra Junior G, Maciel Guerra AT (2007) The role of pediatrician in the management of children with genital ambiguities. J Pediatr 83(5): S184-SI91.
- Marshal FF (1978) Embryology of the Lower Genitourinary Tract. Urologic Clinic of North America 5(1): 3-15.
- Yamada G, Suzuki K, Haraguchi R, Miyagawa S, Satoh Y, et al. (2006) Molecular genetic cascades for external genitalia formation an emerging organogenesis program. Dev Dyn 235(7): 1738-1752.
- Dastgiri S, Sheikhzadeh Y, Dastgiri A (2011) Monitoring of congenital anomalies in developing countries: a pilot model in Iran. Stud Health Technol Inform 164: 157-161.
- Ajao AE, Adeoye IA (2019) Prevalence, risk factors and outcome of congenital anomalies among neonatal admissions in Ogbomoso, Nigeria. BMC Pediatr 19: 88.
- Czeizel AE, Intody Z, Modell B (1993) What proportion of congenital abnormality can be prevented? BMJ 306(6876): 499-503.
- Sakar S, Patra C, Dasgupta MK, Nayek K, Karmakar PR (2013) Prevalence of congenital anomalies in neonates and associated risk factors in a tertiary care hospital in eastern India. J Clin Neonatol 2(3): 131-134.
- Mashhadi AH, Kargar MH (2014) Prevalence of congenital anomalies: a community-based study in the northwest of Iran. ISRN Pediatr, pp. 920940.
- Dolk H, Loane M, Garne E (2010) The prevalence of congenital anomalies in Europe. Adv Exp Med Biol 686: 349-364.
- Li ZY, Chen YM, Qiu LQ, Chen DQ, Hu CG, et al. (2019) Prevalence, types and malformations in congenital anomalies of the kidney and urinary tract in newborns: a retrospective hospital-based study. Ital J Pediatr 45(1): 50.
- Abdou MSM, Sherif AAR, Wahdan IMH, Ashour KSED (2019) Pattern and risk factors of congenital anomalies in a pediatric university hospital, Alexandria, Egypt. J Egypt Public Health Assoc 94(1): 3.
- Adeyemo AA, Gbadegesin RA, Omotade OO, Asinobi AO (1998) Major urogenital malformations in Nigerian children. African Journal of Nephrology 2: 39-42.
- Bhat A, Kumar V, Bhat M, Kumar R, Patni M, et al. (2016) Incidence of apparent congenital urogenital anomalies in North Indian newborns: A study of 20,432 pregnancies. African Journal of Urology 22(3): 183-188.
- Grossi M, Crawford S, Chandra S (2017) Advanced Maternal Age and incidence of Non-Chromosomal Congenital Anomalies: A Population-based Study. Obstetrics & Gynecology 129(5): 182S.
- Tennant PWG, Raza F, Bythell M, Rankin J (2010) Maternal age and the risk of structural congenital anomalies. Archives of Disease in Childhood-Fetal and Neonatal Edition 95(1): Fa4.
- Geotzinger KR, Shanks AL, Odibo AO, Macones GA, Cahill AG (2017) Advanced Maternal Age and Risk of Major Congenital Anomalies. Am J Perinatol 34(3): 217-222.
- McCurdy CM Jr, Seeds JW (1993) Route of delivery of infants with congenital anomalies. Clin Perinatol 20(1): 81-106.
- Van der Horst HJ, De Wall LL (2017) Hypospadias, all there is to know. Eur J Pediatr 176(4): 435-441.
- Fernandez N, Escobar R, Zarante I (2016) Craniofacial anomalies associated with hypospadias. Description of a hospital based population in South America. Int Braz J Urol 42(4): 793-797.
- Ogundoyin OO, Ogunlana DI, Lawal TA, Ademola SA (2017) Managemnent of hypospadias in a resource-poor setting: The Ibadan experience. Nigerian Journal of Plastic Surgery 13(2): 40-44.
- Braga LH (2017) Crytorchidism: A practical review for all healthcare providers. Can Urol Assoc 11(1-2Suppl1): S26-S32.
- Song R, Yosypiv IV (2011) Genetics of congenital anomalies of the kidney and urinary tract. Pediatr Nephrol 26(3): 353-364.
- Parimi M, Nitsch D (2020) A Systemic Review and Meta-Analysis of Diabetes During Pregnancy and Congenital Genitourinary Abnormalities. Kidney Int Rep 5(5): 678-693.
- Offor I, Awodele O, Oshikoya KA (2019) Drug-related teratogenic and pathologic causes of birth defects in a tertiary hospital in Southwestern Nigeria. Pharmacol Res Perspect 7(1): e00452.
- Gupta S, Mohi JK, Gambhir P (2020) Prenatal diagnosis of congenital anomalies of the genito-urinary system on fetal magnetic resonance imaging. Egypt J Radiol Nucl Med 51: 155.

 Research Article
Research Article