Abstract
Women infertility is a serious problem in an increasing number of couples. Carriers of structural chromosomal rearrangements such as Robertsonian or reciprocal translocations have an increased risk of spontaneous abortion and producing offspring with genetic abnormalities. We report an infertile woman with abnormal karyotype uncommon Robertsonian translocation (RT) 45, XX, rob (13;13) (10q;10q) chromosomal constitution, while the male partner showed normal 46, XY karyotype. Peripheral blood was taken from patients, then performed with lymphocyte culture and stained by binded using Giemsa-banding method. Cytogenetic study was performed on the first-degree relatives of our proband whose mother, father, if any, and their living children, who can be reached, in order to reveal whether the translocation was de novo or familial in our case. According to the a-CGH was made for Uniparental disomi(UPD), but no similar repeat fields above 10 Mb were found the mother and father of the case were found to have normal karyotype.
Keywords: Robertsonian Translocations; Carrier; Chromosomal Rearrangements; Abortion; Uniparental Disomy
Introduction
The most common cause of pregnancy losses is chromosomal anomalies. Fetal chromosomal anomalies are found in 90% of preembryonic losses, 50% of first trimester abortions, 2/3 of malformed embryos, and 1/3 of malformed fetuses. Considering all pregnancy losses, the rate of chromosomal anomalies is 50%. It is accepted that the chromosomal rearrangements detected in the parents are an important etiological factor in spontaneous abortion, stillbirth, or birth of a baby with malformation. Chromosomal abnormalities, according to the results of research done all over the world; It is mostly characterized by infertility, habitual failures or birth of children with unbalanced karyotype. It is among the indications for postnalate and prenatal diagnosis as one of the most important causes of infertility [1]. Robertsonian translocations (ROBs) are common structural chromosome rearrangement in humans and ROBs are created by fusion of two ac-rocentric chromosomes at or near the centromere. The gametes of Robertson type translocation carriers display 1/3 normal, 1/3 carrier and 1/3 unbalanced new structure. Gametes with unbalanced chromosomes will cause monosomic/trisomic fetuses following fertilization. Monosomic fetuses end in miscarriage; trisomic fetuses will cause trisomy 21,13,14,15 or trisomy22, depending on their chromosome structure. Most of them will end in miscarriage and some will reach term [2,3].
Abnormal phenotype is not observed in these translocations, but the real danger is that the silent translocation carriers will encounter varying degrees of risk for spontaneous abortions and the birth of children with anomalies, which they will encounter in their desired adulthood. The level of risk in question depends on which acrocentric chromosomes are involved in RT carriers and their sex. The higher risk is RTs between homologous acrocentric chromosomes 13(13q;13q) or 21(21q;21q), and RT carriers with homologous chromosomes have a chance to give birth to children with Patau or Down syndrome, but never give birth to normal children [4].
Case Report
A non-consanguineous couple of age 25 years (male) and 22 years old (female) were admitted in the Afyonkarahisar Health Science University of Medical Genetics Department with a history of repeated abortions for cytogenetics evaluation. They were not related. The wife had had four spontaneous abortions during the first trimester. The proband was phenotypically normal. His wife had normal menstrual cycles. In the marriage story of our proband, there was a miscarriage that recurred twice in the last two years. Her first abortion resulted in a fetus characterized by edematous villi structures with decidua, four months after conception. There was no history of such recurrent abortions in other family members. Parents of proband were made cytogenetics analyses also. Consent form was obtained from the patients since cytogenetic analysis and a-CGH procedures were studied from peripheral blood. Genetic counseling was given to the family. It was explained that it is not possible to give birth to a healthy child under these conditions. It was explained that the only option could be with the egg donation method.
Chromosome Analysis
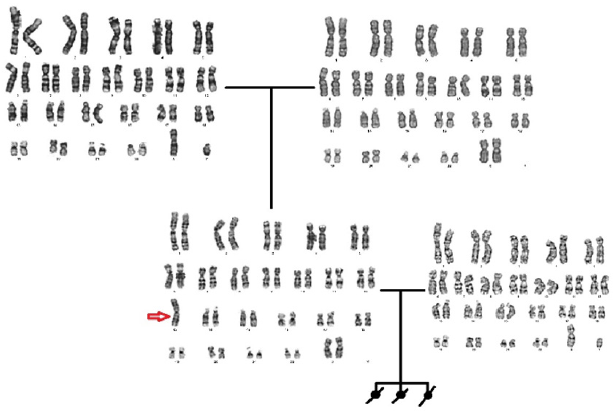
For cytogenetic analysis for karyotyping analysis, 2 ml peripheral blood samples with sodium heparin were collected from both partners. RPMI 1640 medium was prepared for lymphocyte culture, supplemented with 0.2 ml PHA, and cultured blood samples were incubated at 37 °C for 72 hours. After fixation using fixative (3: 1 methanol-acetic acid), G-banded metaphases were prepared for cytogenetic analysis and chromosome study was performed. Twenty-five metaphases for each partner were analyzed and the karyotype was interpreted using Applied Imaging Software. Chromosomes were identified and classified according to the International System for Human Cytogenetic Nomenclature (ISCN, 2016) guidelines, karyotyping, and numerical and structural abnormalities recorded. The karyotype of the proband was 45, XX, rob (13;13)( 10q;10q) as seen in (Figure 1). Belongs to husband and parents of proband were 46, XX and 46, XY normal karyotype as seen in Figure 1. According to the a-CGH was made for UPD, but no similar repeat fields above 10 Mb were found the mother and father of the case were found to have normal karyotype.
Discussion
In this case report, we present a patient with de novo 45, XX, rob t(13;13)(10q;10q) balanced robertsonian translocation carrier a female. Our case is an extremely special and rare case with a clinical message. It has been reported that the chromosomal irregularities detected in first trimester abortions are caused by unbalanced gametes that occur in the parents of balanced translocation carriers. Chromosomal aberrations in human cell nuclei with 23 pairs of chromosomes cause reduced fertility in both men and women [5]. It is accepted that chromosomal rearrangements in parents are an important etiological factor in spontaneous abortion, stillbirth or birth of a baby with malformation. Since approximately 15% of pregnancies mostly result in spontaneous abortion in the first trimester, chromosome analysis during this period is extremely important. The most important cause of spontaneous abortions occurring with an incidence of 50% is chromosomal abnormalities [6,7]. Reciprocal and Robertsonian type translocations involving the exchange of different chromosomal parts are commonly observed between chromosomes. Robertsonian translocation is considered to be the most common structural chromosomal abnormality in the population, with an incidence of 1.23/1000 live births [2].
Reardan et al. Described a female case with 2 abortions with a Robertsonian translocation, similar to our case. First of all, they emphasized that there was no mosaicism in their cases, similar to our patient. They also suggested that the detected translocation occurred at the stage of chromosome rearrangement, very early stages of zygote development, and immediately after the fusion of pronuclei during fertilization. In our case, because of the absence of mosaicism and the same translocation, we think that robertsonian translocation occurred in the postzygotic stage in the etiology of the event [8]. According to the a-CGH result of our patient for UPD, but it was found that the mother and father of the patient who did not have similar repeat areas above 10 Mb had normal karyotype. Chromosomal microarray usually (sequence comparative genomic hybridization, aCGH) analysis is useful for detecting clinically significant copy number abnormalities in patients with phenotypic features suggesting a congenital chromosome rearrangement and is currently recommended by the American College of Medical Genetics as a first trimester test for specific patients. Transmission of homologous Robertsonian translocations from normal surrogate mothers to normal surrogate daughters was the first indicator of UPD with no apparent phenotypic effect [9]. Although both novo and familial rearrangements between acrocentric chromosomes resulted in UPD, it was not compatible with UPD in our case [10].
Although our case is a balanced ROB carrier, she has a normal phenotype, but she cannot has never a healthy child. This situation was expressed in the publication of Wei Weiz Zang et al. In a way that supports our study; “Balanced ROB carriers are well known to generally have a normal phenotype, but they can have oligospermia in male adults, abortion in female adults, or infertility associated with translocation,” they noted. Although rare, various abnormal phenotypes have been described in some balanced ROB carriers [11].
Conflict of Interest
Regarding this study, the authors and / or their family members do not have a scientific and medical committee membership or relationship with their members, consultancy, expertise, working status in any company, shareholding or similar situations that may have a potential conflict of interest.
Authorship Contributions
Idea/Concept: Serap Tutgun Onrat; Muhsin Elmas; Design: Serap Tutgun Onrat; Muhsin Elmas; Supervision/Consultancy: Serap Tutgun Onrat, Data Collection and/or Processing: Kamuran Avcı Analysis and / or Interpretation: Serap Tutgun Onrat; Kamuran Avcı Source Search: Serap Tutgun Onrat; Muhsin Elmas; Manuscript Writing: Serap Tutgun Onrat; Critical Review: Serap Tutgun Onrat.
Source of Finance
During this study, any pharmaceutical company that has a direct connection with the subject of the research, a company that supplies and / or produces medical tools, equipment and materials, or any commercial company, during the evaluation process of the study, financial and / or no moral support was received.
References
- Alp MN, Oral D (2006) Genetic studies in couples with recurrent spontaneous abortions. Dicle Medical Journal 33 (2): 71-80.
- Bugge M, DeLozier-Blanchet C, Bak M, Brandt CA, Hertz JM, et al. (2005) Trisomy 13 due to rea(13q;13q) is caused by I (13) and not rob(13;13) (q10;q10) in the Majority of Cases. American Journal of Medical Genetics 132 A (3): 310-313.
- Kolgeci S, Kolgeci J, Azemi M, Shala R, Dakas A, et al. (2013) Reproductive risk of the silent carrier of Robertsonian translocation. Medicinski arhiv 67(1): 56-59.
- Scriven PN, Flinter FA, Braude PR, Ogilvie CM (2001) Robertsonian translocations - Reproductive risks and indications for preimplantation genetic diagnosis. Human Reproduction 16(11): 2267-2273.
- Ahmadi Shadmehri A, Darbouy M, Tabatabaiefar MA, Tavakkoly Bazzaz J (2020) A 15; 15 Translocation in a couple with Repeated Abortions: Case report. International Journal of Research in Applied and Basic Medical Sciences 6(1): 14-17.
- Nielsen J, Wohlert M (1991) Chromosome abnormalities found among 34910 newborn children: results from a 13-year incidence study in Århus, Denmark. Human Genetics 87(1): 81-13.
- Venkateshwari, Ananthapur Srilekha, Avvari Sunitha, Tella Pratibha, Nallari Jyothy A, et al. (2010) Recurrent Abortions: A Case Report 11(3): 197-200.
- Reardon PC, Greenstein RM, Howard RO, Gianacopolos EK, Breg WR, et al. (1981) Unusual mosaicism of De novo structural abnormalities and ocular anomalies in a male with 13 trisomy syndromes. American Journal of Medical Genetics. 10(2): 113-118.
- Kirkels VGHJ, Hustinx Th WJ, Scheres JMJC (1980) Habitual abortion and translocation (22q;22q): unexpected transmission from a mother to her phenotypically normal daughter. Clinical Genetics 18(6): 456-461.
- Shaffer LG, McCaskill C, Adkins K, Hassold TJ (1998) Systematic search for uniparental disomy in early fetal losses: The results and a review of the literature. American Journal of Medical Genetics 79(5): 366-372.
- Zhao WW, Wu M, Chen F, Jiang S, Su H, et al. (2015) Robertsonian translocations: An overview of 872 Robertsonian translocations identified in a diagnostic laboratory in China. PLoS ONE [Internet]. 2015 May 1 [cited 2021 Mar 10]; 10(5): e0122647.

 Case Report
Case Report