Abstract
Head and Neck cancer (HNC) is a complex and heterogeneous disease associated with high incidence of disseminated metastasis. Traditional oncologic treatment resulted ineffective for a total regression of the disease. Nanomedicine is emerging as innovative and useful therapeutic approach able to overcome limitations of current treatments. A variety of Nanomaterials (NMs) are under investigation in preclinical and clinical trials to validate and guarantee the safety and effective use of NMs against Head and neck metastatic cancer.
Abbreviations: HNC: Head and Neck Cancer; NMs: Nanomaterials; AuNRs: Gold Nanorods; AuNPs: Gold Nanoparticles; SPION: Superparamagnetic Iron Oxide Nanoparticle
Mini Review
Head and Neck Cancer (HNC) is a complex multifactorial disease that originates in the epithelial layer of mucosa affecting oral cavity, pharynx and larynx. HNC is an invasive disease able to infiltrate surrounding lymph nodes with high incidence of ipsilateral and cervical lymph node metastasis (40%), and tissues with greater incidence in lungs (66%), bone (22%), liver (10%), skin, mediastinum and bone marrow [1]. Prognosis depends to several factors among which disease’s stage, loco-regional relapses and incidence of distant metastasis. Actually a median survival of about 10 months is associated with diagnosis of distant metastasis [2]. Patients with HNC are treated with surgical ablation combined with radiotherapy and chemotherapy but patients showing HNC recurrence and spread metastasis are not responsive to these treatments. Cancer metastasis resistance is largely due to different and heterogeneous metastatic cells subpopulation in which they modify gene expression, growing rate, cell surface properties and functions, compared to primary tumor cells; these instability and consequent mutations are the causes of resistance against common drugs therapy. Moreover, drugs inability to reach sites of metastasis contributes to the poor outcome of current therapies [3].
Nanotechnology based therapy has emerged as promising oncology approach able to overcome limitations of current treatments. Nanomaterials (1-100nm) can act as carriers for drugs and targeting ligands to reach selectively cancer cells. Main properties that allow nanomaterials to exploit their favorable actions are: large surface area, structural surface properties and long circulation time; moreover due to their controlled drug load and selective delivery showed excellent biocompatability, biodistribution and biodegradation resulting in lower systemic toxicity [4]. The incoming application of nanotechnology- based approaches in medicine are bringing advantageous opportunities for the detection, prevention and treatment of cancer metastasis due to their ability to direct specific response against defined target [3,5]. In this mini-review were summarized properties and applications of nanomaterials for treatment and detection of Head and Neck metastatic cancer and more recent studies under clinical trials validation.
Nanotechnology- Based Therapy
Nanomaterials (NMs) can be classified according to their material properties, shape and application. Concerning materials used, NMs can be categorized in organic (gold and metals nanoparticles, quantum dots and fullerenes) and inorganic (liposomes, dendrimers and polymeric nanoparticles). NMs were primarily used as drug cover- protection to avoid premature degradation and/or clearance and at the same time prolong drug circulating time. Drug can be loaded inside the NMs structure (encapsulated drug) or surface absorbed/conjugated, depending on type of release desired [6]. NMs can exploit active or passive targeting strategy to perform their activity and reach desired site. Basically active targeting exploits the high affinity of ligand-receptor mechanism; by functionalizing the NMs surface with selective ligands; conversely passive targeting is based on NMs physicochemical properties suitable to exploit EPR effect (Enhanced Permeability and Retention) that guarantees accumulation of NMs in the tumor site due to an increased vascular permeability [7]. Moreover NMs can act themself as therapeutic agents exploit specific material properties. Exist NMs that are susceptible to externalinduced physical triggers and endogenous- induced triggers to perform their activity. External-induced physical triggers NMs are sensitive to photo- thermal specific light wavelength or to magnetothermal selective magnetic field. When these materials are excited by the external physical triggers, they release vibrational energy as heat to ablate malignant cells. Depending on the temperature induced (hyperthermia), it’s possible caused cells apoptosis (42-46 C) or cells necrosis (> 46 C) [8].
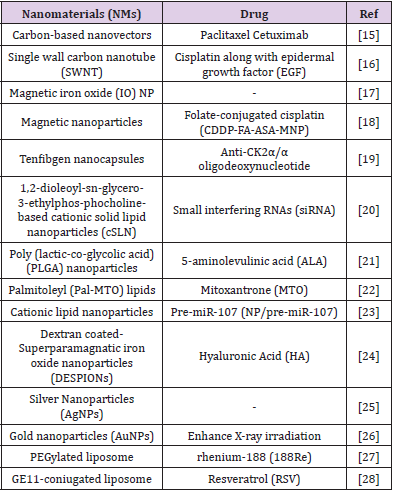
The most used nanomaterials in hyperthermia treatments are: Gold Nanorods (GNRs), Gold Nanoparticles (GNPs) and Superparamagnetic Iron Oxide Nanoparticle (SPION) [9] These NMs are also used in clinic as contrast agents for imaging (magnetic resonance imaging-MRI) due to their ability to become detectable under magnetic field and distinguish diseased tissues from healthy ones [10]. Iron oxide NPs are used is Magnetic Resonance Imaging MRI to detect liver metastases, metastatic lymph nodes, inflammatory and degenerative diseases at an early stage when other contrast agents are unable to visualize them [11]. Instead Endogenous- induced triggers NMs are sensitive to endogenous environment changing such as pH, temperature, hypoxia and enzyme concentration. Using these environment variations NMs are able to achieve high drug concentration in the tumor region saving normal cells [12]. In vivo NMs biodistribution is correlated to their size and shape; remaining in the nanosize range, little dimension variations could influence NMs retention, uptake and pharmacologic activity [12]. Was reported that uptake of spherical gold nanoparticles with approx diameters of 50 nm was higher than that of particles showing lower (14nm) or higher (74 nm) diameters. Concerning NMs shape, comparison between different gold nanomaterials (spherical nanoparticles, rod, wire, hollow) confirmed that different cellular up-take by endothelial cells was observed and more in specific, uptake of spherical nanoparticles was favored [13]. In Table 1 we choose some representative examples to have an overview of nanomaterials and drugs tested in preclinical studies for head and neck cancer and metastasis therapy [14].
Nanotechnology in Clinical Trials
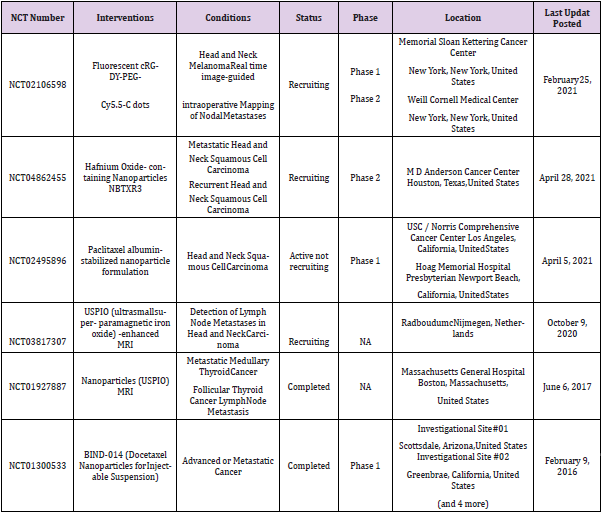
A fair number of nanotechnology drug formulations tested both for diagnostic and/or therapeutic purposes are under investigation in clinical trials for HNC and metastatic treatments in order to evaluate in vivo their effects for future approval in clinic applications [15-20]. In Table 2 are summarized only the most recent clinical trials focused on Head and Neck cancer metastasis found on clinicatrials.gov [21-23].
Table 2: Nanomaterials tested in clinical trials for Head and Neck metastatic cancer.
Note: NA: Not Applicable.
Conclusion
Many strategies are under investigation in order to improve HNC cancer metastasis treatments and therapies. Nanomaterials, thanks to their suitable properties such as small size, selective ligands functionalization, drug protection and vehiculation, suitable retention in the diseased site and modulated responses according to the applied stimulus (endogenous or exogeneous), are becoming increasingly used both in therapy and diagnostic (theranostic application) [24-26]. NMs have the potential to enhance chemotherapy efficiency without increasing toxicity showing many encouraging results demonstrated by preclinical and clinical trials for HNC cancer and metastasis treatments. Despite these new achievements, are still not enough for a complete eradication of metastatic cancer diseases and knowledge of cancer spreading mechanisms [27,28]. For a more in deep reading we suggest the review “Metastatic Disease in Head and neck cancer” that provide a complete and multidisciplinary update on distant metastases in Head and Neck oncology [29].
Conflict of Interest
None.
References
- Noguti J, De Moura CF, De Jesus GP, Da Silva VH, Hossaka TA, et al. (2012) Metastasis from oral cancer: an overview. Cancer genomics & proteomics 9(5): 329-335.
- De Lima JM, Bonan PR, Da Cruz Perez DE, Hier M, Alaoui-Jamali MA, et al. (2020) Nanoparticle-Based Chemotherapy Formulations for Head and Neck Cancer: A Systematic Review and Perspectives. Nanomaterials (Basel) 10(10): 1938.
- Banerjee D, Cieslar-Pobuda A, Zhu GH, Wiechec E, Patra HK (2019) Adding Nanotechnology to the Metastasis Treatment Arsenal. Trends in pharmacological sciences 40(6): 403-418.
- Awasthi R, Roseblade A, Hansbro PM, Rathbone MJ, Dua K, et al. (2018) Nanoparticles in Cancer Treatment: Opportunities and Obstacles. Current drug targets 19(14): 1696-1709.
- Schroeder A, Heller DA, Winslow MM, Dahlman JE, Pratt GW, et al. (2012) Anderson, Treating metastatic cancer with nanotechnology. Nature Reviews Cancer 12(1): 39-50.
- Schroeder A, Heller DA, Winslow MM, Dahlman JE, Pratt GW, et al. (2011) Anderson, Treating metastatic cancer with nanotechnology. Nature Reviews Cancer 12(1): 39-50.
- Greish K (2010) Enhanced permeability and retention (EPR) effect for anticancer nanomedicine drug targeting, Methods in molecular biology (Clifton NJ) 624: 25-37.
- Brace C (2011) Thermal tumor ablation in clinical use. IEEE Pulse 2(5): 28-38.
- Chatterjee DK, Diagaradjane P, Krishnan S (2011) Nanoparticle-mediated hyperthermia in cancer therapy. Therapeutic delivery 2(8): 1001-1014.
- Debouttière PJ, Roux S, Vocanson F, Billotey C, Beuf O, et al. (2006) Design of Gold Nanoparticles for Magnetic Resonance Imaging. Adv Funct Mater 16(18): 2330-2339.
- Gobbo OL, Sjaastad K, Radomski MW, Volkov Y, Prina-Mello A (2015) Magnetic Nanoparticles in Cancer Theranostics. Theranostics 5(11): 1249-1263.
- Huang DY, Hou YL, Yang SM, Wang RG (2014) Advances in nanomedicine for head and neck cancer. Frontiers in bioscience (Landmark edition) 19: 783-788.
- Bartczak D, Muskens OL, Nitti S, Sanchez-Elsner T, Millar TM, et al. (2012) Interactions of human endothelial cells with gold nanoparticles of different morphologies. Small 8(1): 122-130.
- Zhao Y, Chen H, Chen X, Hollett G, Gu Z, et al. (2017) Targeted nanoparticles for head and neck cancers: overview and perspectives. WIREs Nanomedicine and Nanobiotechnology 9(6): e1469.
- Sano D, Berlin JM, Pham TT, Marcano DC, Valdecanas DR, et al. (2012) Noncovalent assembly of targeted carbon nanovectors enables synergistic drug and radiation cancer therapy in vivo. ACS Nano 6(3): 2497-2505.
- Bhirde AA, Patel V, Gavard J, Zhang G, Sousa AA, et al. (2009) Targeted killing of cancer cells in vivo and in vitro with EGF- directed carbon nanotube-based drug delivery. ACS nano 3(2): 307-316.
- Zhao Q, Wang L, Cheng R, Mao L, Arnold RD, et al. (2012) Magnetic Nanoparticle-Based Hyperthermia for Head &Neck Cancer in Mouse Models. Theranostics 2(1) (2012) 113-121.
- Wang D, Fei B, Halig LV, Qin X, Hu Z, et al. (2014) Targeted iron-oxide nanoparticle for photodynamic therapy and imaging of head and neck cancer. ACS Nano 8(7): 6620-6632.
- Brown MS, Diallo OT, Hu M, Ehsanian R, Yang X, et al. (2010) CK2 modulation of NF-kappaB, TP53, and the malignant phenotype in head and neck cancer by anti-CK2 oligonucleotides in vitro or in vivo via sub-50-nm nanocapsules. Clinical cancer research: an official journal of the American Association for Cancer Research 16(8): 2295-2307.
- Yu YH, Kim E, Park DE, Shim G, Lee S, et al. (2012) Cationic solid lipid nanoparticles for co-delivery of paclitaxel and siRNA. Eur J Pharm Biopharm 80(2): 268-273.
- Wang X, Shi L, Tu Q, Wang H, Zhang H, et al. (2015) Treating cutaneous squamous cell carcinoma using 5-aminolevulinic acid polylactic-co- glycolic acid nanoparticle-mediated photodynamic therapy in a mouse model. International journal of nanomedicine 10: 347-355.
- Chang RS, Suh MS, Kim S, Shim G, Lee S, et al. (2011) Cationic drug-derived nanoparticles for multifunctional delivery of anticancer siRNA. Biomaterials 32(36): 9785-9795.
- Piao L, Zhang M, Datta J, Xie X, Su T, et al. (2012) Lipid-based nanoparticle delivery of Pre-miR-107 inhibits the tumorigenicity of head and neck squamous cell carcinoma. Mol Ther 20(6): 1261-1269.
- Thompson A, Thapa R, Galoforo S, Hang X, Buelow K, et al. (2017) Targeting Stem Cells in Head and Neck Cancer Using Superparamagnetic Iron Oxide Nanoparticles. International Journal of Radiation Oncology, Biology, Physics 99(2): E621.
- Singh J, Moore W, Fattah F, Jiang X, Zheng J, et al. (2019) Activity and pharmacology of homemade silver nanoparticles in refractory metastatic head and neck squamous cell cancer. Head & Neck 41(1): E11-E16.
- Teraoka S, Kakei Y, Akashi M, Iwata E, Hasegawa T, et al. (2018) Gold nanoparticles enhance X‑ray irradiation‑induced apoptosis in head and neck squamous cell carcinoma in vitro. Biomed Rep 9(5): 415-420.
- Chang CY, Chen CC, Lin LT, Chang CH, Chen LC, et al. (2018) PEGylated liposome-encapsulated rhenium-188 radiopharmaceutical inhibits proliferation and epithelial–mesenchymal transition of human head and neck cancer cells in vivo with repeated therapy. Cell Death Discovery 4(1): 100.
- Zheng T, Feng H, Liu L, Peng J, Xiao H, et al. (2019) Enhanced antiproliferative effect of resveratrol in head and neck squamous cell carcinoma using GE11 peptide conjugated liposome. International journal of molecular medicine 43(4): 1635-1642.
- Pisani P, Airoldi M, Allais A, Aluffi Valletti P, Battista M, et al. (2020) Metastatic disease in head & neck oncology. Acta otorhinolaryngologica Italica: organo ufficiale della Societa italiana di otorinolaringologia e chirurgia cervico-facciale 40(1): S1-S86.

 Mini Review
Mini Review