Abstract
Vector-borne diseases transmitted by insect vectors such as mosquitoes occur in
over 100 countries and affect almost half of the world’s population. Dengue is currently
the deadliest arboviral disease but chikungunya and Zika show increasing prevalence
and severity. Vector control, mainly by the use of insecticides, play a key role in disease
prevention but the use of the same chemicals for more than 40 years, together with
the dissemination of mosquitoes by human activities, resulted in the global spread of
insecticide resistance. In this context, innovative tools and strategies for vector control
are urgently needed. Arboviruses transmitted by mosquitoes represent a major health
problem in EMRO countries. The main vector control activities include larviciding, space
spraying, impregnated bednet and indoor residual spraying. The susceptibility status of
the two main vectors of Arboviruses, Aedes aegypti and Ae. albopictus was evaluated in
different regions of EMRO and Indian subcontinent.
Resistance to different insecticide classes such as pyrethroids, organophosphate,
organochlorine, carbamates used as imagocide and larvicide were evaluated using WHO
guidelines. An intensive search of scientific literature was done in “PubMed”, “Web of
Knowledge”, “Scopus”, “Google Scholar”, “SID”, etc. Results showed a wide variety of
susceptibility/resistance status to these chemicals according to the location. Historical
context of pesticide used, genetic background of vectors, age and abdominal conditions of
adults may play a role in the susceptibility status of these species to different insecticides.
Monitoring and mapping of resistance in countries should be carried out for appropriate
vector control.
Keywords: Resistant; Arboviruses; Insecticides; Eastern Mediterranean Region
Introduction
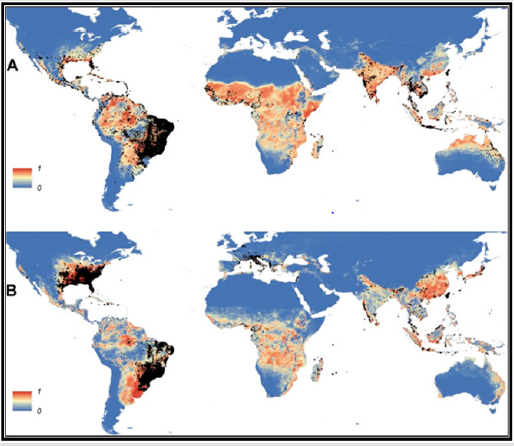
Diseases transmitted by mosquitoes include malaria, dengue, West Nile virus, chikungunya, yellow fever, filariasis, tularemia, dirofilariasis, Japanese encephalitis, Saint Louis encephalitis, Western equine encephalitis, Eastern equine encephalitis, Venezuelan equine encephalitis, Ross River fever, Barmah Forest fever, La Crosse encephalitis, Zika fever, Keystone virus and Rift Valley fever: Usutu virus. Currently dengue is spreading worldwide, placing at risk around 40% of the global population [1]. To date, no specific drugs are available and dengue treatment is restricted to supportive care. The major dengue vector is Aedes aegypti and Aedes albopictus which their distribution is shown in Figure 1. An estimated 50 million dengue infections occur annually and approximately 2.5 billion people live in dengue endemic countries. In addition, the recent chikungunya and Zika virus dispersion throughout the globe. Actions against dengue are mostly focused on the reduction of mosquito densities, and vector control can be accomplished through mechanical, biological, and chemical approaches [2].
Dengue is the most rapidly spreading mosquito-borne viral
disease in the world. In the last 50 years, incidence has increased
30-fold with increasing geographic expansion to new countries and,
in the present decade, from urban to rural settings [3]. According to
the WHO guideline several insecticides recommended for mosquito
control [4]. Insecticide Resistance is recognized by the World
Health Organization (WHO) as an important threat to arboviral
disease control and prevention. There is an urgent need to identify
the countries and regions where resistance could challenge vector
control and to accelerate the deployment of innovative tools for
vector control. Better understanding of the strength and dynamics
of insecticide resistance will help to develop a global strategy for
insecticide resistance containment in arbovirus vectors. Outbreaks
of dengue have been documented in the Eastern Mediterranean
Region possibly as early as 1799 in Egypt [5].
Recent outbreaks of suspected dengue have been recorded in
Saudi Arabia, Sudan and Yemen, 2005-2006 [5,6]. Yemen is also
affected by the increasing frequency and geographic spread of
epidemic dengue in 2005. Since the first case of DHF died in Jeddah
in 1993, Saudi Arabia has reported three major epidemics in 2011
[5]. The frequency of reported outbreaks continues to increase,
with outbreaks for example in Sudan in 1985 [6] and in Djibouti
in 1991 [7], and Somalia [8]. In 2015 an outbreak in Egypt that
occurred following a decade, long absence of reported cases from
that country [9]. Pakistan may be represent the highest burden
of dengue in EMRO region, since 2006, dengue epidemics have
occurred every year and the range has extended to most cities in
Pakistan. Dengue now affects thousands of people and has caused
hundreds of deaths. It has become a major health problem in
Pakistan, and it is likely to become an even greater health problem
in the coming years [10].
On 1 May 2019, in response to increasing numbers of dengue
fever cases in Pakistan and India, health authorities in Afghanistan
heightened monitoring for the disease. The 14 cases were
reported from six provinces. This is the first report of people with
autochthonous, meaning locally acquired cases of dengue fever in
Afghanistan. Both Aedes albopictus and Aedes aegypti are present
in Afghanistan.
Results and discussion
All the results were followed as defined by WHO (1981,
1992, 1998) (11-13): susceptible when mortality was 98% or
higher, possible resistant when mortality was between 97 and
90%, and resistant when the mortality was lower than 90%. An
excel sheet was created for insecticide resistance based on the
applied insecticide at diagnostic dosage recommended by WHO. Deltamethrin and cypermethrin were used to evaluate resistance/
susceptible status of field collected adult females of Aedes aegypti
from Slum area of Misri Shah Lahore (Pakistan), the result indicated
that Aedes females were more resistant to deltamethrin as
compared to cypermethrin [11-14]. In Pakistan the results showed
varying degrees of resistance in field populations. Resistance in Ae.
albopictus to chlorpyrifos was generally very high while moderate
to high levels of resistance were found with organophosphate,
pyrethroid, new chemicals and two from carbamates [15].
In 2010 in Lahor resistance status of Aedes aegypti was
evaluated against pyrethroid insecticide. Results indicated that Ae.
aegypti field collected population from Government Islamia College
for Women Cooper Road, Lahore was resistant to Deltamethrin.
However, field population of Ae. aegypti from Government College
University, Lahore was found susceptible [16]. In Sonitpur district
of Assam insecticide susceptibility assays were performed on wildcaught
adult female Aedes albopictus mosquitoes. Ae. albopictus
was resistant to DDT (4%) in all study sites except Gohpur. The
species was found to be 100% susceptible to deltamethrin (0. 05%)
in all study sites [17]. A survey was performed in Delhi on Aedes
albopictus, Gurgaon (Haryana), Hardwar (Uttarakhand), Guwahati
(Assam) and Kottayam (Kerala). Results revealed High resistance
against DDT in Uttarakhand and Haryana population, whereas
Delhi, Kerala and Assam populations showed tolerance.
Delhi population showed 97% mortality for deltamethrin and
Kerala population showed 96% mortality against permethrin.
All other populations studied were fully susceptible against both
pyrethroids [18]. The status of resistance in Ae. aegypti from Lahore
(Pakistan) was evaluated against pyrethroids insecticide. Results
indicated that Aedes aegypti field population from Government
Islamia College for Women Cooper Road, Lahore (GICW) was
resistant to Bifenthrin. Whereas the field collected population of
Ae. aegypti from Government College University, Lahore (GCU) was
found susceptible as compared to laboratory reared population
[19]. A survey in Asam (India) showed that both St.albopictus and
St.aegypti were fully resistant to DDT in all the study locations.
Both the species were completely susceptible to deltamethrin
and malathion except for St. albopictus at Sotia which displayed
low level of resistance to malathion [20]. The result of insecticide
susceptibility tests in Delhi (India) shows high resistance against
DDT and moderate level of resistance to pyrethroids (deltamethrin;
permethrin mortalities) [21].
The survey in Delhi revealed that adult Ae. aegypti was resistant
to DDT and dieldrin, tolerant to propoxur and fenitrothion,
but was susceptible to malathion, deltamethrin, permethrin
and lambdacyhalothrin. However, the larvae were found to be
susceptible to all the three larvicides tested, viz. temephos, fenthion
and malathion [22]. In the Jazan Region of Saudi Arabia adults Ae.
aegypti mosquitoes were found to be susceptible only to Cyfluthrin,
whereas variable resistances were observed from Lambdacyhalothrin,
Deltamethrin, Permethrin, Fenitrothion, Bendiocarb
and DDT insecticides. The Ae. aegypti larvae were resistant to
Temephos and showed high susceptibility to Methoprene than
Diflubenzuron. The larvae were more susceptible to Methoprene
than Diflubenzuron [23]. In Port Sudan City, Ae. aegypti were found
to be susceptible to Deltamethrin, Bendiocarb, tolerant to Lambdacyhalothrin
and resistant to DDT and Malathion [24].
A study in Republic of Yemen revealed that the mosquito
larvae of Ae. aegypti were more susceptible to the OP insecticides
sumithion than acifon, actellic and of onac respectively, while the
pyrethroids fendona was more effective against larvae than aralin.
Data indicated that adult mosquitoes of the field strain Ae. aegypti
were resistant to the insecticides lambdacyhalothrin, malathion
and fenitrothion but were tolerant to deltamethrin, permethrin
and cyfluthrin [25]. The few insecticide resistance data available
revealed widespread resistance to DDT in Ae. aegypti across the
country while resistance to organophosphates appeared more
frequent in southern India. Pyrethroid resistance in Ae. aegypti
and Ae. albopictus was reported in Delhi and Kerala regions [26-
29]. Challenges for the control of arboviral diseases in India include
the development of vector surveillance and resistance monitoring
programs and the implementation of rational vector control
strategies throughout the country.
There are several reports of resistant status and mechanism of
insecticide resistance in Ae. aegypti and Ae. albopictus to different
groups of insecticides in the world [30-54]. WHO (2020) [55]
recommended several measures for prevention and control of
Dengue including: preventing mosquitoes from accessing egglaying
habitats by environmental management and modification;
disposing of solid waste properly and removing artificial manmade
habitats, covering, emptying and cleaning of domestic
water storage containers on a weekly basis, applying appropriate
insecticides to water storage outdoor containers; using of
personal household protection such as window screens, longsleeved
clothes, insecticide treated materials, coils and vaporizers,
improving community participation and mobilization for sustained
vector control, applying insecticides as space spraying during
outbreaks as one of the emergency vector-control measures, active
monitoring and surveillance of vectors should be carried out to
determine effectiveness of control interventions.
Conflict of Interest
The authors declare that there is no conflict of interest.
Acknowledgment
This research is supported by Ministry of Health and Medical Education under code number of NIMAD 982984
References
- (2014) World Health Organization. A global brief on vector-borne diseases.
- (2009) World Health Organization. Dengue: Guidelines for Diagnosis, Treatment, Prevention and Control, WHO Press, Geneva, Switzerland.
- (2008) World Health Organization. Dengue and dengue fever. World Health Organization, Geneva.
- (2006) World Health Organization. Pesticides and their application: for the control of vectors and pests of public health importance.
- (2005) WHO/EMRO. Regional Office for the Eastern Mediterranean; Division of Communicable Disease Control. World Health Organization, Newsletter L: 7-8.
- Hyams KC (1986) Evaluation of febrile patients in Port Sudan, Sudan: isolation of dengue virus. American Journal of Tropical Medicine and Hygiene 35: 860-865.
- Rodier GR, Gubler DJ, Cope SE, Cropp CB, Soliman AK, et al. (1996) Epidemic dengue 2 in the city of Djibouti 1991-1992. Transaction Royal Society of Tropical Medicine and Hygiene 90: 237-240.
- Kyobe H, Bosa HK, Montgomery JM, Kimuli I, Lutwama J et al. (2014) Dengue fever outbreak in Mogadishu, Somalia 2011: Co-circulation of three dengue virus serotypes. International Journal of Infectious Diseases 21(S1): 3.
- (2015) World Health Organization. Dengue Fever in Egypt.
- Rasheed SB, Butlin RK, Boots M (2013) A review of dengue as an emerging disease in Pakistan. Public Health 127(1): 11-17.
- (1992) World Health Organization. Vector resistance to pesticides. Fifteenth report of the WHO expert committee on vector control. WHO Tech Rep Ser.
- (1981) World Health Organization. Instruction for determining the susceptibility or resistance of adult mosquito to organochlorine, organophosphorine and carbamate insecticides- diagnostic test.
- (1998) World Health Organization. Report of the WHO informal consultation. Test procedures for insecticide resistance monitoring in malaria vectors, bio-efficacy and persistence of insecticides on treated surfaces.
- Jahan N, Shahid A (2013) Evaluation of resistance against deltametrin and cypermethrin in dengue vector from Lahore, Pakistan. Journal of Animal and Plant Sciences 23(5): 1321-1326.
- Khan HAA, Akram W, Shehzad K, Shaalan EA (2011) First report of field evolved resistance to agrochemicals in dengue mosquito, Aedes albopictus (Diptera: Culicidae), from Pakistan. Parasites and Vector 4: 146-151.
- Jahan N, Mumtaz N (2010) Evaluation of resistance against deltamethrin in Aedes mosquitoes from Lahore, Pakistan Biologia 56 (1&2): 9-15.
- Das M, Dutta P (2014) Status of insecticide resistance and detoxifying enzyme activity of Aedes albopictus population in Sonitpur district of Assam, India. International Journal of Mosquito Research 1(4): 35-41.
- Kushwah RBS, Mallick PK, Ravikumar H, Dev V, Kapoor N, et al. (2015) Status of DDT and pyrethroid resistance in Indian Aedes albopictus and absence of knockdown resistance (kdr) mutation. Journal of Vector Borne Diseases 52(1): 95-98.
- Jahan N, Sadiq A (2012) Evaluation of resistance against Bifenthrin in dengue vector from Lahore, Pakistan. Biologia (Pakistan) 58 (1&2): 13-19.
- Yadav K, Rabha B, Dhimas S, Veer V (2015) Multi- insecticide susceptibility evaluation of Dengue vector Stegomia albopictus and St. aegypti in Assam, India. Parasites and Vectors 8: 1-8.
- Kushwah RBS, Dykes CL, Kapoor N, Adak T, Singh OP (2015) Pyrethroid-Resistance and presence of two knockdown resistance (kdr) mutations, F1534C and a novel mutation T1520I, in Indian Aedes aegypti. PLoS Neglected Tropical Diseases 9(1): e3332.
- Katyal R, Tewari P, Rahman SJ, Pajni HR, Kumar K, et al. (2001) Susceptibility status of immature and adult stages of Aedes aegypti against conventional insecticides in Delhi, India. Dengue Bulletin 25: 84-87.
- Alsheikh AA, Mohammed WS, Noureldin EM, Daffalla OM, Shrwani KJ, et al. (2016) Resistance status of Aedes aegypti to Insecticides in the Jazan Region of Saudi Arabia. Bioscience Biotechnology. Research of Asia (1): 155-162.
- Husham ASO, Abdalmagid MA, Brair M (2001) Status susceptibility of dengue vector; Aedes aegypti to different groups of Insecticides in Port Sudan City, Red Sea State. Sudan Joural of Public Health 5(4): 199-202.
- Alhag SKN (2013) Susceptibility status of dengue fever vector Aedes aegypti, (L.) in republic of Yemen. Bioscience Biotechnology Research Community 6(2): 138-141.
- Dhiman S, Rabha B, Yadav K, Baruah I, Veer V (2014) Insecticide susceptibility and dengue vector status of wild Stegomyia albopicta in a strategically important area of Assam, India. Parasites and Vectors.
- Kushwah RB, Mallick PK, Ravikumar H, Dev V, Kapoor N, et al. (2015) Status of DDT and pyrethroid resistance in Indian Aedes albopictus and absence of knockdown resistance (kdr) mutation. Journal of Vector Borne Diseases 52(1): 95-98.
- Muthusamy R, Shivakumar MS (2015) Involvement of metabolic resistance and F1534C kdr mutation in the pyrethroid resistance mechanisms of Aedes aegypti in India. Acta Tropica 148: 137-141.
- Muthusamy R, Shivakumar MS (2015) Susceptibility status of Aedes aegypti (L.) (Diptera: Culicidae) to temephos from three districts of Tamil Nadu, India. Journal of Vector Borne Diseases 52(2): 159-165.
- Suzuki T, Osei JH, Sasaki S, Adimazoya M, Appawu M, et al. (2016) Risk of transmission of viral haemorrhagic fevers and the insecticide susceptibility status of Aedes aegypti (Linnaeus) in some sites in Accra, Ghana. Ghana Medical Journal 50(3): 136-141.
- Mariappan T, Selvam A, Rajamannar V, Arunachalam N (2017) Susceptibility of Dengue/Chikungunya Vector, Aedes aegypti Against Carbamate, Organochlorine, Organophosphate and Pyrethroid Insecticides. Journal of Environmental Biolology 38(2): 251.
- Bharati M, Saha D (2018) Multiple insecticide resistance mechanisms in primary dengue vector, Aedes aegypti (Linn.) from dengue endemic districts of Sub-Himalayan West Bengal, India. PloS One 13(9): e0203207.
- Bharati M, Rai P, Saha D (2019) Insecticide Resistance in Aedes albopictus Skuse from Sub-Himalayan Districts of West Bengal, India. Acta Tropica 192: 104-111.
- Neng W, Yan X, Fuming H, Dazong C (1992) Susceptibility of Aedes albopictus from China to Insecticides, and Mechanism of DDT Resistance. Journal of American Mosquito Control Association 8(4): 394-397.
- Cui F, Raymond M, Qiao CL (2006) Insecticide Resistance in Vector Mosquitoes in China. Pest Management Science 62(11): 1013-1022.
- Yiguan W, Xin L, Chengling L, Su T, Jianchao J, et al. (2017) A Survey of insecticide resistance in Aedes albopictus (Diptera: Culicidae) During a 2014 dengue fever outbreak in Guangzhou, China. Journal of Economic Entomology 110(1): 239-244.
- Su X, Guo Y, Deng J, Xu J, Zhou G, et al. (2019) Fast Emerging Insecticide Resistance in Aedes albopictus in Guangzhou, China: Alarm to the Dengue Epidemic. PLoS Neglected Tropical Diseases 13(9): e0007665.
- Tangena J AA, Marcombe S, Thammavong P, Chonephetsarath S, Somphong B, et al. (2018) Bionomics and insecticide resistance of the arboviral vector Aedes albopictus in northern Lao PDR. PloS one 3(10): e0206387.
- Varón LS, Córdoba BC, Brochero HL (2010) Susceptibilidad de Aedes aegypti a DDT, Deltametrina y lambdacialotrina en Colombia. The Revista Panamericana de Salud Pública 27(1): 67.
- Fonseca González I, Quiñones ML, Lenhart A, Brogdon WG (2011) Insecticide resistance status of Aedes aegypti (L.) from Colombia. Pest Management Sciences 67(4): 430-437.
- Aponte A, Penilla RP, Rodríguez AD, Ocampo CB (2019) Mechanisms of Pyrethroid Resistance in Aedes (Stegomyia) aegypti from Colombia. Acta Tropica 191: 146-154.
- Mazzarri MB, Georghiou GP (1995) Characterization of resistance to organophosphate, carbamate, and pyrethroid insecticides in field populations of Aedes aegypti from Venezuela. Journal of the American Mosquito Control Association 11(3): 315-322.
- Mekuria Y, Gwinn T, Williams D, Tidwell M (1991) Insecticide Susceptibility of Aedes aegypti from Santo Domingo, Dominican Republic. Journal of the American Mosquito Control Association 7(1): 69-72.
- Canyon D, Hii J (1999) Insecticide susceptibility status of Aedes aegypti (Diptera: Culicidae) from Townsville. Australian Journal of Entomology 38(1): 40-43.
- Hamid PH, Prastowo J, Ghiffari A, Taubert A, Hermosilla C (2017) Aedes aegypti Resistance Development to Commonly Used Insecticides in Jakarta, Indonesia. PLoS One 12(12): e0189680.
- Marcombe S, Chonephetsarath S, Thammavong P, Brey PT (2018) Alternative Insecticides for Larval Control of the Dengue Vector Aedes aegypti in Lao PDR: Insecticide Resistance and Semi Field Trial Study. Parasites & Vectors 11(1): 1-8.
- Ping LT, Yatiman R, Gek L (2001) Susceptibility of adult field strains of Aedes aegypti and Aedes albopictus in Singapore to Pirimiphos-Methyl and Permethrin. Journal of the American Mosquito Control Association 17(2): 144-146.
- Koou SY, Chong CS, Vythilingam I, Lee CY, Ng LC (2014) Insecticide resistance and its underlying mechanisms in field populations of Aedes aegypti Adults (Diptera: Culicidae) in Singapore. Parasites & Vectors 7(1): 471.
- Lopez B, Ponce G, Gonzalez JA, Gutierrez SM, Villanueva OK, et al. (2014) Susceptibility to Chlorpyrifos in Pyrethroid-Resistant Populations of Aedes aegypti (Diptera: Culicidae) from Mexico. Journal of Medical Entomology 51(3): 644-649.
- Suter T, Crespo MM, de Oliveira MF, de Oliveira TS, de Melo Santos MA, et al. (2017) Insecticide Susceptibility of Aedes albopictus and Ae. aegypti from Brazil and the Swiss-Italian Border Region. Parasites & Vectors 10(1): 431.
- Paeporn P, Supaphathom K, Sathantriphop S, Chareonviritaphap T, Yaicharoen R (2007) behavioural responses of deltamethrin-and permethrin-resistant strains of Aedes aegypti when exposed to permethrin in an excito-repellency test system. Dengue Bulletin 31: 153-159.
- Rohani A, Chu W, Saadiyah I, Lee H, Phang S (2001) insecticide resistance status of Aedes albopictus and Aedes aegypti collected from urban and rural areas in major towns of Malaysia. Tropical Biomedicine 18(1): 29-39.
- Boyer S, Lopes S (2016) WHOPES Methods to test insecticide susceptibility of 4 aedes aegypti field populations in cambodia. Dengue Integrated Vector Management: Dissemination and Policy Uptake Workshop: Phnom Penh.
- Kawada H, Higa Y, Nguyen YT, Tran SH, Nguyen HT, et al. (2009) Nationwide investigation of the pyrethroid susceptibility of mosquito larvae collected from used tires in Vietnam. PLoS Neglected Tropical Disease 3(3): e391.
- (2020) World Health Organization (2020) Dengue and severe dengue.

 Research Article
Research Article