Abstract
Codonopsis (Codonopsis pilosula (Franch.) Nannf.) is a common medicinal and edible plant with a variety of biological functions, including enhancing gastrointestinal motility, anti-inflammatory, antioxidant, regulating glucose and lipid metabolism, regulating immune response and anti-tumor. Aspirin is a widely used clinical drug with multiple therapeutic effects. However, long-term use of aspirin could also bring some side effects, including gastrointestinal discomfort, liver and kidney damage, asthma, and allergies. The present study is aimed to investigate the effect of Codonopsis decoction on aspirin-induced gastric injuries. Cell proliferation was evaluated by MTT assay and cell apoptosis was investigated by flow cytometry. Gastric mucosal injury index and HE staining were used to evaluate the pathological changes of gastric tissue. The levels of cyclooxygenase-1 (COX-1), cyclooxygenase-2 (COX-2), tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6) and transforming growth factor-α (TGF-α) in GES-1 cells culture supernatant were detected by ELISA kits. The levels of cyclooxygenase (COX), tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6) and TGF-α in gastric tissue and serum were detected by ELISA kits. Results showed that Codonopsis decoction significantly attenuated the aspirin-induced cytotoxicity and apoptosis of GES-1 cells. Codonopsis decoction significantly increased the levels of COX-1, COX-2 and TGF-α in GES-1 cells treated by aspirin. Codonopsis decoction could also increase the expression levels of COX and TGF-α in vivo. Codonopsis decoction significantly reduced the levels of TNF-α and IL-6 both in vivo and in vitro. In addition, Codonopsis decoction could increase the daily weight gain of rats taking aspirin, and significantly reduce the gastric tissue damage and gastric mucosal injury index. These data indicated that Codonopsis decoction may possess potential to reduce the side effects caused by oral aspirin and may help the safe use of aspirin.
Keywords: Codonopsis Decoction; Aspirin; Gastric Mucosal Injury; COX; COX-1; COX-2;TNF-α;IL-6; TGF-α s
Introduction
Aspirin is the first non-steroidal drug used clinically in humans
with a wide range of pharmacological effects [1]. Because aspirin
has the effect of anti-thrombosis in the body, it is widely used in
the treatment of cardiovascular and cerebrovascular diseases
[2,3]. However, long-term use of aspirin in the process of treating
cardiovascular and cerebrovascular diseases can cause gastric
mucosal damage, gastrointestinal dysfunction, and eventually show
side effects such as nausea, vomiting and gastrointestinal bleeding
[4]. TGF-α and COX are important molecules for gastric tissue
damage repair. In the process of gastric mucosal injury repair, TGF-α
molecules can promote the migration and proliferation of ulcer
border cells, promote the regeneration of gastric epithelial tissue,
and promote the formation of granulation tissue and angiogenesis
[5]. COX is a catalytic enzyme for the metabolism of arachidonic acid. COX-1 is a physiological enzyme that catalyzes the synthesis
of prostaglandin (PG) involved in normal physiological functions,
and maintains the integrity of the gastrointestinal mucosa [6].
COX-2 is an inducible enzyme produced rapidly by stimulation,
which can induce the synthesis of PG in inflammation sites and
participate in damage. The metabolite PG compounds have an
important protective effect on gastric mucosa [7]. Previous studies
showed that aspirin cause mucosal damage, ulceration and ulcer
complication throughout the gastrointestinal tract by inhibiting PG
synthesis [8]. As important inflammatory cytokines TNF-α and IL-
6, which participate in the process of gastric mucosal damage [9].
Codonopsis is a commonly used Chinese herbal medicine
and is also used as a food ingredient. Traditional Chinese medical
theory believes that Codonopsis can be used to treat syndrome
of qi deficiency of spleen and lung, syndrome of anorexia, and
syndrome of palpitation [10]. Pharmacological studies showed
that Codonopsis extracts exhibit a wide range of pharmacological
activities, including gastrointestinal function regulation, endocrine
function regulation, immune function regulation, hematopoietic
function improvement, cardiovascular protection, anti-aging and
antioxidant, and it has not been found obvious toxicity or side
effects [11-13]. However, the effect of Codonopsis decoction on
aspirin-induced gastric mucosal injury has not been reported yet.
This study will investigate the relieving effect and mechanism of
Codonopsis decoction on gastric mucosal injury induced by oral
aspirin in rats.
Materials and Methods
Preparation of Codonopsis Decoction
The Codonopsis used in this study is the dried root of Codonopsis pilosula, a plant of Campanulaceae. To prepare Codonopsis decoction, slices of Codonopsis (Tongrentang, Beijing, China) weighing 300 g were soaked in 1 L distilled water for 25 min, then heated and boiled for 30 min. Then the decoction was filtered with 3 layers of gauze and centrifuged at 3000 rpm/min for 20 min. Finally, the supernatants were collected and concentrated to 300 mL using a rotary evaporator.
Cell Culture
The human gastric epithelial cells (GES-1) were cultured in RPMI 1640 medium (Gibco, Thermo Fisher Scientific, Inc.) supplemented with 10% FBS (Gibco, Therm Fisher Scientific, Inc.) and 1% penicillin G/streptomycin at 37˚C in a humidified atmosphere containing 5% CO2. GES-1 cells were seeded at 5x105 cells per 25 cm2 culturing flask and were sub‑cultured every time they reached 80% confluence.
MTT Assay
MTT assay was used to examine the effects of Codonopsis decoction on the viability and proliferation of GES-1 cells. Cells were seeded in 96‑well plates at 5x104 cells per well in 100 μL 1640 and cultured at 37˚C for 12 h. Cells were subsequently divided into control group (1640 only) and Codonopsis decoction groups (0.488, 0.9765625, 1.953125 , 3.90625, 7.8125, 15.625 , 31.25, 62.5, 125, 500 and 1000 μg/mL Codonopsis decoction). Cells in the groups were treated with Codonopsis decoction subsequently cultured at 37˚C for 24 h. Following this, 10 μL of MTT solution (5 mg/mL; Sigma-Aldrich; Merck KGaA) was added to each well and incubated at 37 ˚C for 4 h. The culture medium was removed, dimethylsulfoxide (DMSO, Sigma-Aldrich; Merck KGaA) was added to each well and cells were incubated at 37˚C for 10 min, following which the absorbance (A) of MTT was measured at 490 nm using a microplate reader (Bio-Rad Laboratories, Inc., Hercules, CA, USA). According to the above test results: cells were subsequently divided into control group (1640 only), positive control group (3.3 mg/mL aspirin +10 mg/mL OME), aspirin injury group (3.3 mg/ mL) and Codonopsis decoction groups (3.3 mg/mL aspirin + 0.05, 0.1 and 0.2 mg/mL Codonopsis decoction) [14]. Cells in the groups were treated with aspirin or Codonopsis decoction subsequently cultured at 37˚C for 12 and 24 h. All experiments were repeated 3 times.
Flow Cytometry Analysis
Apoptosis rate was determined by fluorescein isothiocyanate (FITC)-labeled Annexin V/propidium iodide (PI) double staining according to the manufacturer’s instructions. Cells in each group were centrifuged at 1000 g for 5 min and subsequently washed twice with PBS. Then cells were resuspended (1 ×106 cells/mL) in 195 μL Annexin V-FITC binding buffer. Aliquots containing 1 ×106 cells in 195 μL buffer were stained with 10 μL PI solution and 5 μL Annexin V-FITC for 25 min. The apoptosis rates were detected by flow cytometry. The experiments were performed in triplicate.
Animals and Model
Male Sprague Dawley rats weighing 200±20 g were selected to establish a gastric mucosal injury model. First, the rats were weighed and fasted for 12 h. Subsequently, rats were given 60 mg aspirin dissolved in 0.5% carboxymethyl cellulose solution (CMC) for 7 consecutive days.
Grouping Situation
The rats used in the study were randomly divided into 5 groups (Table 1): control group (rats were given 0.5% CMC for 7 d followed by distilled water for another 7 d), aspirin group (rats were given 300 mg/kg aspirin for 7 d followed by distilled water for another 7 d), positive control group (rats were given 300 mg/kg aspirin followed by 15 mg/kg OME for another 7 d), low-dose Codonopsis decoction group (rats were given 300 mg/kg aspirin followed by Codonopsis decoction for another 7 d), medium-dose Codonopsis group (rats were given 300 mg/kg aspirin followed by Codonopsis decoction for another 7 d), and a high-dose Codonopsis group (rats were given 300 mg/kg aspirin followed by Codonopsis decoction for another 7 d).
Body Weight
The body weight of rats in each group of were respectively weighed at 0 d, 7 d and 14 d. And the daily weight gain of each group was calculated.
\Anatomical Lesion
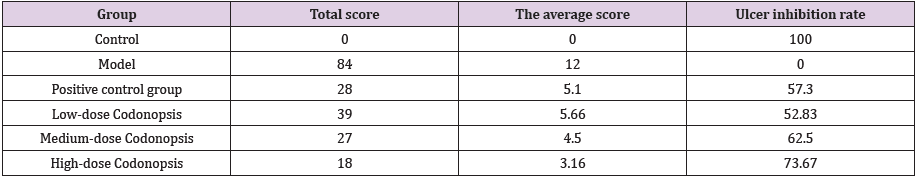
Rats in each group were anesthetized and sacrificed at 14 d. Then stomachs were opened along the greater curvature and washed with saline. According to a previous report, the number and severity of gastric lesions were assessed for overall pathology based on a 0-5 scoring system: [15] 0 points means no lesions, 1 point means small round hemorrhagic lesions, 2 points means that the diameter of the lesion is less than 2 mm, 3 points means that the diameter of the lesion is 2-3 mm, 4 points means that the diameter of the lesion is 3-4 mm, 5 points means that the diameter of the diseased area is greater than 4mm. When the diameter is greater than 1 mm, multiply the score by 2. The average score represents the gastric mucosal injury index (UI). The damage inhibition rate is calculated by the formula: (1-UICodonopsis/UIModel)×100%.
HE Staining
The lesions of the rat stomach were selected and fixed in 4% paraformaldehyde for 24h. HE staining was performed according to the reported method [16]. The pictures was taken using a microscope (Olympus Corporation, Tokyo, Japan).
Elisa Assay
Levels of COX-1, COX-2 ,TNF-α, IL-6 and TGF-α in GES-1 culture supernatant and COX, TNF-α, IL-6 and TGF-α in vivo were detected by ELISA kits. Cells in the groups were treated with aspirin or Codonopsis decoction subsequently cultured at 37˚C for 12 and 24 h. Following this, cell culture supernatants were collected by sterile tube and centrifuged at 1000 g for 20 min (4℃). Blood in each group were collected at 14 d. The serum were separated by centrifugation at 500 g for 15 min. The gastric tissue (0.5 mm×0.5 mm) of each group of rats was selected and washed with PBS 3 times. Then the gastric tissues were thoroughly ground in 1.5 mL PBS and centrifuged at 500 g for 15 min.
Statistical analysis
Results were expressed as means ± standard deviations (SD). The statistical significance of differences was determined using the Student’s t-test or one-way analysis of variance (ANOVA) as appropriate. All the statistical tests were performed using GraphPad Prism software 8.2 (GraphPad software, USA). The p-value < 0.05 was considered statistically significant.
Results
The Effects of Codonopsis Decoction on the Viability of GES-1 Cells
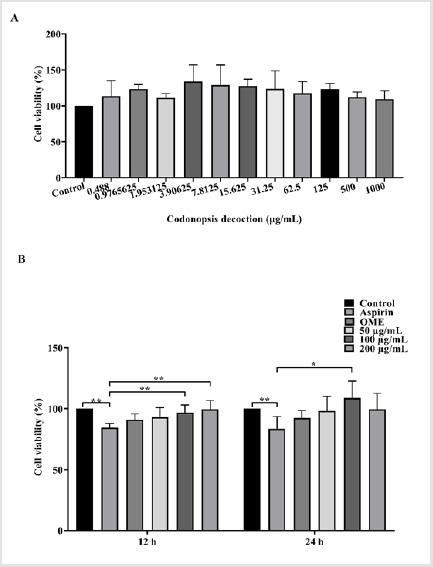
As shown in Figure 1, Codonopsis is safe and non-cytotoxic to GES-1 cells. Compared to the control group, aspirin significantly decreased the viability of GES-1 cells (P<0.01). Positive control group and Codonopsis decoction at concentrations of 100 and 200 μg/mL significantly attenuated the aspirin-induced cytotoxicity.
The Effects of Codonopsis Decoction on the Apoptosis Rate of GES-1 Cells
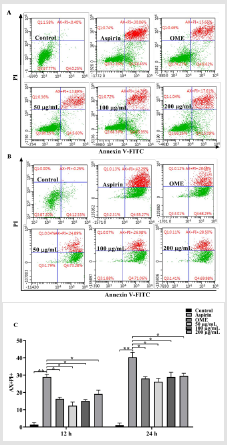
Flow cytometry analysis were performed to determine the effect of Codonopsis decoction on apoptosis of GES-1 cells. As shown in Figure 2, aspirin significantly induced the apoptosis of GES-1 cells (P<0.01) compared with the control group. Positive control group and Codonopsis decoction significantly attenuated the aspirin-induced apoptosis (P<0.05).
Effect of Codonopsis Decoction on Levels of COX-1, COX-2, TNF-α, Il-6 And TGF-α in GES-1 Cells Culture Supernatant
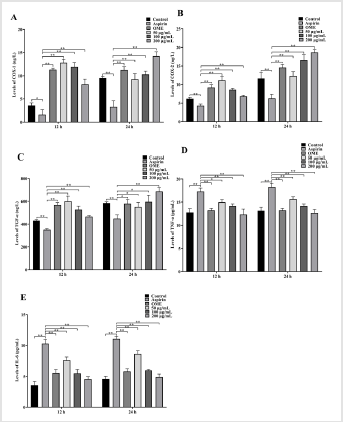
The levels of COX-1, COX-2 and TGF-α were significantly decreased compared with the control group following 12 h and 24 h treatment with aspirin (P<0.01, Figures 3A-3C). The levels of TNF-α and IL-6 were significantly increased compared with the control group following 12 h and 24 h treatment with aspirin (P<0.01, Figures 3D-3E). However, this trend was significantly ameliorated by Codonopsis decoction incubation at 50, 100 and 200 μg/mL and positive control group at 12 or 24 h.
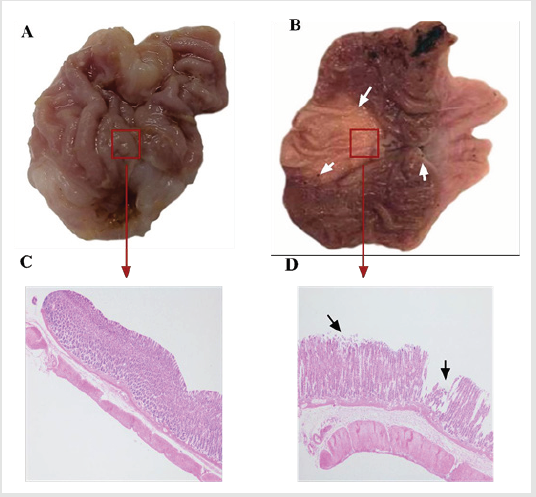
Effect of Oral Aspirin on Gastric Mucosa
As shown in Figure 4, the gastric mucosa of the rats in the control group was intact without obvious erosion, bleeding or ulcer formation (Figure 4A). In contrast, obvious erosions or ulcers were seen on the gastric mucosal of model rats, and the gastric mucosal wrinkles disappeared (Figure 4B). Results of HE staining showed that the mucosal structure of the gastric tissue of the control group was complete, the mucosal glands were arranged neatly, and there was no bleeding and no inflammatory cell infiltration (Figure 4C). In the gastric tissue of the model group, mucosal structural defects, mucosal edema, and interstitial edema can be seen, and there are exudates and necrotic tissue on the surface of the mucosa (Figure 4D).
Figure 1: Effects of Codonopsis decoction on the viability of GES-1 cells
A. Results of different concentrations of Codonopsis decoction on the viability of GES-1 cells.
B. Effects of Codonopsis decoction on GES-1 cells against aspirin-induced cytotoxicity at 12 h and 24 h.
Figure 2: Effects of Codonopsis decoction on apoptosis of GES-1 cells
A. Results of Codonopsis decoction on GES-1 cells against aspirin-induced apoptosis for 12 h.
B. Results of Codonopsis decoction on GES-1 cells against aspirin-induced apoptosis for 24 h.
C. Results of Codonopsis decoction on apoptosis of GES-1 cells.
Figure 3: Levels of COX-1, COX-2, TNF-α, IL-6 and TGF-α in GES-1 culture supernatant
A. Levels of COX-1 at 12 h and 24 h.
B. Levels of COX-2 at 12 h and 24 h.
C. Levels of TGF-α at 12 h and 24 h.
D. Levels of TNF-α at 12 h and 24 h.
E. Levels of IL-6 at 12 h and 24 h.
Figure 4: Effect of aspirin on the gastric mucosa of rats. The rats in the control group and the model group were given distilled water or aspirin by gavage for 7d. The gastric lesions were observed at 7 d. HE staining was used to detect pathological changes of gastric mucosa
A. The gastric lesions of rats in the control group.
B. Gastric lesions of rats in the model group.
C. Histopathology of the gastric mucosa of rats in the control group.
D. Histopathology changes of gastric mucosa in model rats.
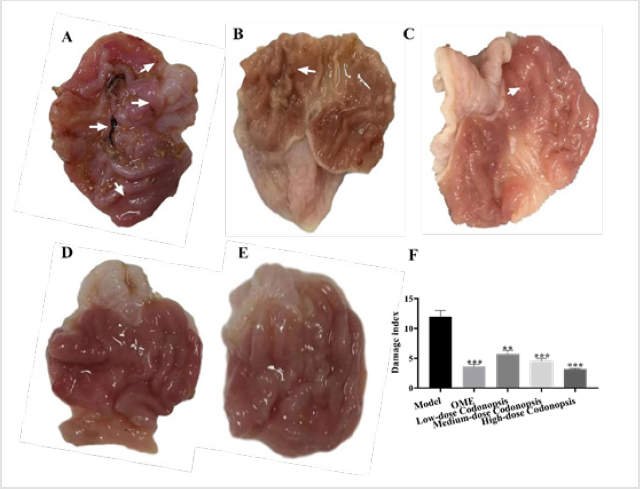
Codonopsis Decoction vould Significantly Reduce Gastric Mucosal Injury Index
As shown in Figure 5, compared with the rats in the model group, the gastric mucosal folds of the rats in the low-dose Codonopsis decoction group were basically intact, and the diameter of gastric mucosal lesions in some rats was reduced. The gastric mucosal folds of rats in the medium-dose Codonopsis decoction group and positive control group were intact, and the number of lesions was significantly reduced. The gastric mucosal folds of the rats in the high-dose Codonopsis group were intact, and only one rat had minimal lesions in the gastric mucosa. The statistical results showed that the UI values of high-dose, medium-dose and low-dose Codonopsis decoction groups and positive control group were significantly increased (P<0.01), and the UI values of highdose Codonopsis decoction was significantly higher than that of medium-dose group and low-dose group (Table 2).
Figure 5: The effect of Codonopsis decoction on aspirin-induced gastric mucosal injury. Rats were given aspirin orally for 7 d, and the rats were given high-dose, medium-dose and low-dose Codonopsis decoction for another 7 d. Then the gastric mucosa lesions were observed
A. Gastric mucosal lesions of rats in model group.
B. Gastric mucosal lesions in rats in low-dose Codonopsis group.
C. Gastric mucosal lesions of rats in medium-dose Codonopsis group.
D. Gastric mucosal lesions in rats in high-dose Codonopsis group.
E. Gastric mucosal lesions in rats in positive control group.
F. Injury index of rat gastric mucosa.
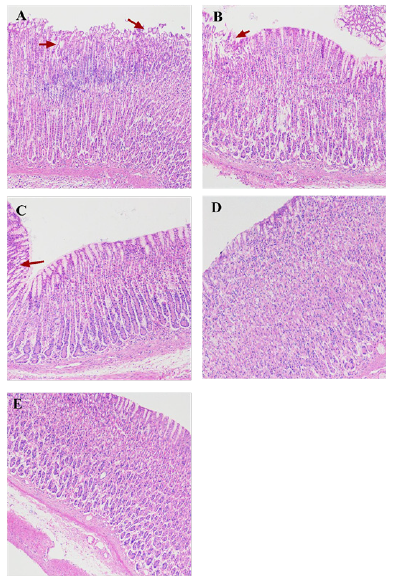
Codonopsis Decoction Could Significantly Reduce the Stomach Histopathology Induced by Aspirin
As shown in Figure 6, the pathological section of the model group showed that the surface mucosa was damaged, superficial erosion and some necrotic tissue, the boundary between the mucosa and submucosa was blurred, interstitial edema and inflammation Infiltration of sex cells occurs, glands around the ulcer damaged, structure unclear, arrangement disordered, and there are epithelial cell shedding. Pathological sections of rats in each group of Codonopsis and positive control group showed that mucosal surface exudate decreased, inflammatory exudate decreased, granulation tissue increased, and epithelial cells were generated and covered on the mucosal surface.
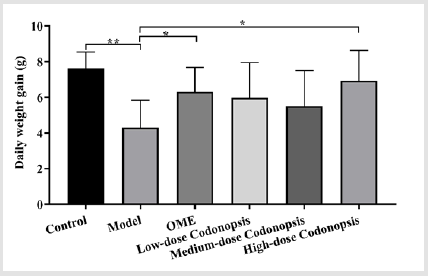
Effect of Codonopsis Decoction on Body Weight of Rats
The average daily gain of rats in the model group was significantly lower than that of the control group (P<0.01) (Figure 7). Compared with the model group, the average daily gain of rats in the high-dose Codonopsis decoction group and positive control group were increased significantly (P<0.05), while the average daily weight gain of the middle-dose and low-dose Codonopsis decoction groups was not increase significantly.
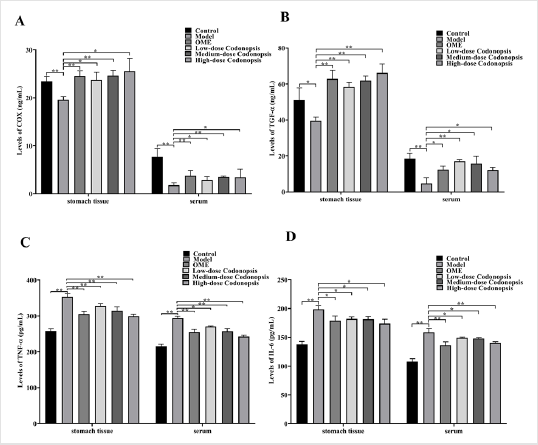
Effect of Codonopsis Decoction on Levels of COX, TNF-α, IL-6 and TGF-α in Serum and Stomach Tissue
As shown in Figure 8, the levels of COX and TGF-α in serum and gastric tissues of rats taking aspirin were significantly lower than those in the control group (P<0.01, Figures 8A-8B). After administration of Codonopsis decoction and OME, the levels of COX and TGF-α in serum and gastric tissue of rats increased significantly. Compared with the control group, the levels of TNF-α and IL-6 were significantly increased following treatment with aspirin (P<0.01, Figures 8C-8D). However, after administration of Codonopsis decoction and OME, the levels of TNF-α and IL-6 in serum and stomach tissue of rats were decreased significantly.
Figure 6: The effect of Codonopsis decoction on histopathology of gastric mucosa
A. Histopathology of the gastric mucosa of rats in model group.
B. Histopathology of gastric mucosa of rats in low-dose Codonopsis group.
C. Histopathology of gastric mucosa of rats in medium-dose Codonopsis group.
D. Histopathology of gastric mucosa of rats in high-dose Codonopsis group.
E. Histopathology of gastric mucosa of rats in positive control group
Figure 7: Daily weight gain of rats. The body weight of each rat was measured at 0 and 14 d. The daily weight gain is calculated by the following formula: (Body weight14d- body weight0d) / 14.
Figure 8: Levels of COX, TNF-α, IL-6 and TGF-α in rat stomach tissue and serum
A. Levels of COX in stomach tissue and serum.
B. Levels of TGF-α in stomach tissue and serum.
C. Levels of TNF-α in stomach tissue and serum.
D. Levels of IL-6 in stomach tissue and serum.
Discussion
Codonopsis is a classical traditional Chinese herbal, sometimes
used as a substitute for Ginseng to widely replenish Qi deficiency,
strengthen the immune system, enhance the body’s metabolism,
improve poor GI function, alleviate gastric ulcers, and improve
appetite [17,18]. To the best of our knowledge, there have been no
previous studies investigating the improving effect of Codonopsis
decoction on aspirin-induced gastric lesions. In the present study,
we investigated the effect of Codonopsis decoction on aspirininduced
gastric damage both in vivo and in vitro. Results showed
that aspirin incubation significantly inhibited the proliferation
and promoted apoptosis of the GES-1 cells in vitro. However, after
intervention with Codonopsis decoction, the cells proliferation
activity were obviously restored. And the rate of apoptosis is
significantly reduced. Aspirin-induced the low levels of COX-1,
COX-2 and TGF-α in GES-1 culture supernatant were significantly
attenuated by Codonopsis decoction at all doses. Aspirin-induced
the high levels of TNF-α and IL-6 in GES-1 culture supernatant were
also significantly attenuated by Codonopsis decoction at all doses.
The in vivo study investigated the alleviating effect of Codonopsis
decoction on gastric mucosal injury in rats caused by oral aspirin.
Results show that oral aspirin can induce obvious gastric mucosal
damage in rats, which is in accordance to previous studies [19-21].
At the same time, after oral administration of aspirin, the daily gain
of rats was significantly reduced, the expression levels of COX and
TGF-α in serum and stomach tissue were significantly reduced, and
the expression levels of TNF-α and IL-6 in serum and stomach tissue
were significantly increased. However, after oral administration
of Codonopsis decoction, the daily gain of model rats increased,
the number of gastric mucosal lesions was significantly reduced,
and the pathological changes of gastric mucosa were significantly
reduced. It shows that Codonopsis decoction can significantly
reduce the gastric injury induced by oral aspirin.
TGF-α is a regulatory peptide of the EGF family, also known
as “mucosal intact peptide”. It is an endogenous protective factor
in cell proliferation and promotes a variety of cells by binding to
the receptor epidermal growth factor receptor (EGFR) of mitosis
[22-24]. TGF-α plays an important role in maintaining the integrity
of the mucosa, can regulate the proliferation of gastric mucosal
epithelial cells, and participate in the regulation of mucosal
epithelial renewal and repair after injury [25,26]. As a commonly
used drug, aspirin has antipyretic and analgesic, anti-inflammatory
and anti-rheumatic effects [27]. The main mechanism of aspirin
anti-inflammatory is to inhibit the synthesis of prostaglandin
by inhibiting COX. However, because aspirin does not have the
characteristics of targeting, oral aspirin will also inhibit the level
of COX in the gastric mucosa [28-30]. COX is a key enzyme for the
formation of PG, and PG has a protective effect on gastric tissue
[31]. This study found that the concentrations of COX in the gastric
tissue and serum of rats after oral administration of aspirin were
significantly reduced (P<0.01). This result further confirmed the
effect of aspirin on the expression of COX. At the same time, we
found that the levels of TGF-α in the stomach tissue and serum of
rats that were orally taken aspirin were also significantly reduced.
It can be speculated that inhibiting the damage and repair function
of gastric mucosal epithelium may be a way for aspirin to cause
gastric damage. On the contrary, the levels of TGF-α and COX in
serum and stomach tissue of rats that were given Codonopsis
decoction after oral administration of aspirin significantly
increased. Correspondingly, the gastric mucosal lesions of rats in
the Codonopsis decoction group were significantly reduced. TNF-α
and IL-6 are cytokines with a wide range of biological effects, which
have an important regulatory role in the immune and inflammatory
process of the body [32]. These increase in their expression levels
usually mean the aggravation of body damage. IL-6 can activate the
central nervous system to regulate injury and defense, and at the
same time regulate immune response and acute phase response,
which is significantly increased in patients with gastric ulcer [33].
TNF-α is a kind of endogenous multidirectional inflammatory cell
factors that can activate neutrophils and continue to damage the
gastric mucosa, causing ulcers to be difficult to heal [34]. Aspirininduced
the high levels of TNF-α and IL-6 in serum and stomach
tissue of rats were also significantly attenuated by Codonopsis
decoction at all doses. These data suggest that the Codonopsis
decoction may relieve aspirin-induced gastric mucosal damage by
reducing the inhibitory effect of aspirin on COX and TGF-α, and the
high expression levels of TNF-α and IL-6.
Pyrazole drugs are usually used to treat gastric mucosal damage
caused by aspirin, but pyrazole drugs can also bring side effects
such as hypomagnesemia and skin damage [35,36]. Codonopsis is
a traditional Chinese medicine that has been used for hundreds of
years, and it is also a plant with the same medicine and food [37].
It is enough to ensure the safety of Codonopsis. It should be noted
that more than one hundred compounds have been isolated and
identified from Codonopsis, including polysaccharides, alkaloids,
lactone compounds, volatile oils, etc. [38]. These compounds endow
Codonopsis with a variety of biological effects. It may be one of the
ingredients that plays a specific role, or it may be a combination of
several ingredients. In addition, the results of this study indicate
that Codonopsis decoction has effects on COX, COX-1, COX-2, TNF-α,
IL-6 and TGF-α. Combined with the multi-target characteristics
of traditional Chinese medicine, [39] we believe that the effect of
Codonopsis decoction may be better than that of a single compound.
In summary, the results of the study indicate that Codonopsis
decoction may target on COX, COX-1, COX-2, TNF-α, IL-6 and TGF-α
to reduce aspirin-induced gastric mucosal injury both in vivo and
in vitro. This suggests that Codonopsis may be a potential choice
to relieve or eliminate the side effects of aspirin. Of course, more
specific works still need to be carried out to clarify the exact pharmacological effects of the different ingredients in Codonopsis
decoction on the above aspects.
Acknowledgment
This work was supported by the National Key R&D Special Foundation of China (2017YFD0501501), Pig Industry Innovation Foundation of Beijing (BAIC02-2020) and Establishment of a detection system for the analysis of traditional Chinese medicine components by NMR spectrometer (TCVM-201804).
References
- TANG S Y, SIVAKUMAR M, NG A Min Parthasarathy S (2012) Anti-inflammatory and analgesic activity of novel oral aspirin-loaded nanoemulsion and nano multiple emulsion formulations generated using ultrasound cavitation [J]. Int J Pharm 430(1-2): 299-306.
- FERNANDEZ JIMENEZ R, WANG T J, FUSTER V (2019) Low-Dose Aspirin for Primary Prevention of Cardiovascular Disease: Use Patterns and Impact Across Race and Ethnicity in the Southern Community Cohort Study [J]. J Am Heart Assoc 8(24): e013404.
- ALMAS A, GHAZNI M S, HASHMANI S (2018) Aspirin in Primary Prevention of Myocardial Infarction/Angina and Stroke in Hypertensive Patients. J Coll Physicians Surg Pak 28(7): 574.
- HERNANDEZ C, BARRACHINA M D, VALLECILLO HERNANDEZ J, ALVARAZ A, DOLORES OM, et al. (2016) Aspirin-induced gastrointestinal damage is associated with an inhibition of epithelial cell autophagy. J Gastroenterol 51(7): 691-701.
- NUR AZLINA M F, QODRIYAH H M S, CHUA K H, (2017) Comparison between tocotrienol and omeprazole on gastric growth factors in stress-exposed rats. World J Gastroenterol 23(32): 5887-5894.
- KOUMAS L, PHIPPS R P (2002) Differential COX localization and PG release in Thy-1(+) and Thy-1(-) 341 human female reproductive tract fibroblasts. Am J Physiol Cell Physiol 283(2): C599-608.
- BRZOZOWSKI T, KONTUREK PC, KONTUREK SJ (2005) Role of prostaglandins in gastroprotection and gastric adaptation. J Physiol Pharmacol 56(5): 33-55.
- MAHMOUD Y I, ABD EL GHFFAR E A (2019) Spirulina ameliorates aspirin-induced gastric ulcer in 345 albino mice by alleviating oxidative stress and inflammation. Biomed Pharmacother 109: 314-321.
- NIE N, BAI C, SONG S, Zhang Y, Wang B, et al. (2020) Bifidobacterium plays a protective role in TNF-alpha-induced inflammatory response in Caco-2 cell through NF-kappaB and p38MAPK pathways. Mol Cell Biochem 464(1-2): 83-91.
- HOSSEN M J, KIM M Y, KIM J H, Cho JY (2016) Codonopsis lanceolata: A Review of Its Therapeutic Potentials. Phytother Res 30(3): 347-356.
- GAO SM, LIU J S, WANG M, Cao TT, Qi YD, et al. (2018) Traditional uses, phytochemistry, pharmacology and toxicology of Codonopsis: A review. J Ethnopharmaco 219: 50-70.
- ZOU YF, ZHANG YY, FU YP, Kari TI, Berit SP, et al. (2019) A Polysaccharide Isolated from Codonopsis pilosula with Immunomodulation Effects Both In Vitro and In Vivo. Molecules 24(20): 3632.
- SUI F, WANG R, WANG J, Li Y, Du Q, et al. (2005) Effects of Dangshen root extract serum on intracellular free 357 calcium in gastric parietal cells of dissociated rats. Zhong Yao Cai 28(10): 900-903.
- LIU C, DUAN Z, GUAN Y, Wu H, Hu K, et al. (2018) Increased expression of tight junction protein occludin is associated with the protective effect of mosapride against aspirin-induced gastric injury [J]. Exp Ther Med 15(2): 1626-1632.
- ARAB HH, SALAMA SA, OMAR HA, Arfa E, Maghrabi IA (2015) Diosmin protects against ethanol-induced gastric 362 injury in rats: novel anti-ulcer actions. PLoS One 10(3): e0122417.
- SERAFINI S, SANTOS MM, AOUN TANNURI AC, Zerbini MC, COELHO MC, et al. (2017) Is hematoxylin-eosin staining in rectal mucosal and submucosal biopsies still useful for the diagnosis of Hirschsprung disease? Diagn Pathol 12(1): 84.
- JING Y, LI A, LIU Z, Yang P, Wei J, et al. (2018) Absorption of Codonopsis pilosula Saponins by Coexisting Polysaccharides Alleviates Gut Microbial Dysbiosis with Dextran Sulfate Sodium-Induced Colitis in Model Mice. Biomed Res Int: 1781036.
- LIANG W, ZENG LY, YU CX, LU DF, XIAO W (2018) Effect of flight transport stress on blood parameters in beagles and the anti-stress effect of dangshen. Animal Model Exp Med 1(2): 162-168.
- ADEFISAYO M A, AKOMOLAFE R O, AKINSOMISOYE O S, QUADRI Ak, LOAFE O, et al. (2018) Protective Effects of Methanol Extract of Vernonia amygdalina () Leaf on Aspirin-Induced Gastric Ulceration and Oxidative Mucosal Damage in a Rat Model of Gastric Injury. Dose Response 16(3):1559325818785087.
- FAN DD, LIN S, SONG YP, WANG ZW, LIU B, et al. (2016) Astragaloside IV protects rat gastric mucosa against aspirin-induced damage. Int Immunopharmacol 41: 47-55.
- VERMA S, KUMAR V L (2018) Artesunate affords protection against aspirin-induced gastric injury by targeting oxidative stress and proinflammatory signaling. Pharmacol Rep 70(2): 390-397.
- BULUT K, FELDERBAUER P, HOECK K, SCHMIDT EW, HOFFMAN P, et al. (2008) Increased duodenal expression of transforming 380 growth factor-alpha and epidermal growth factor during experimental colitis in rats. Eur J Gastroenterol Hepatol 20(10): 989-994.
- BRZOZOWSKI T, PTAK-BELOWSKA A, KWIECIEN S, MACZKA KG, STRZALKA M, et al. (2012) Novel concept in the mechanism of injury and protection of gastric mucosa: role of renin-angiotensin system and active metabolites of angiotensin [J]. Curr Med Chem 19(1): 55-62.
- SAKAMOTO N, OUE N, SENTANI K, Anami K, Uraoka A, et al. (2012) Liver-intestine cadherin induction by epidermal 386 growth factor receptor is associated with intestinal differentiation of gastric cancer. Cancer Sci 103(9): 1744-1750.
- KJELLEV S (2009) The trefoil factor family - small peptides with multiple functionalities [J]. Cell MoLife Sci 66(8): 1350-1369.
- SYAM A F, SADIKIN M, WANANDI S I (2009) Molecular mechanism on healing process of 391 peptic ulcer. Acta Med Indones 41(2): 95-98.
- DONG L, ANDERSON AJ, MALKOWSKI MG (2019) Arg-513 and Leu-531 Are Key Residues Governing Time-Dependent Inhibition of Cyclooxygenase-2 by Aspirin and Celebrex. Biochemistry 58(38): 3990-4002.
- LUCOTTI S, CERUTTI C, SOYER M (2019) Aspirin blocks formation of metastatic intravascular niches by inhibiting platelet-derived COX-1/thromboxane A2 [J]. J Clin Invest 129(5): 1845-1862.
- MABROK HB, MOHAMED MS (2019) Induction of COX-1, suppression of COX-2 and 398 pro-inflammatory cytokines gene expression by moringa leaves and its aqueous extract in 399 aspirin-induced gastric ulcer rats. Mol Biol Rep 46(4): 4213-4224.
- HAWKEY C J (2001) COX-1 and COX-2 inhibitors. Best Pract Res Clin Gastroenterol 15(5): 801-820.
- WALLACE JL (2008) Prostaglandins, NSAIDs, and gastric mucosal protection: why doesn't the stomach digest itself? Physiol Rev 88(4): 1547-1565.
- HONG S, LI Y, SU D (2019) Sevoflurane induces apoptosis of isolated placental trophoblast cells and stimulates expressions of TNF-alpha and IL-6. Am J Transl Res 11(11): 7084-7093.
- ERCAN G, ILBAR TARTAR R, SOLMAZ A (2020) Potent therapeutic effects of ruscogenin on gastric ulcer established by acetic acid. Asian J Surg 43(2): 405-416.
- MOUSA AM, EL SAMMAD NM, HASSAN SK (2019) Antiulcerogenic effect of Cuphea ignea extract against ethanol-induced gastric ulcer in rat0073. BMC Complement Altern Med 19(1): 345.
- JURAKIC TONCIC R, BALIC A, PAVICIC B (2019) Occupational Airborne Contact Dermatitis 412 Caused by Omeprazole [J]. Acta Dermatovenerol Croat 27(3): 188-189.
- HANSEN B A, BRUSERUD O (2016) Hypomagnesemia as a potentially life-threatening adverse effect of omeprazole. Oxf Med Case Reports 7: 147-149.
- LIU W, LV X, HUANG W (2018) Characterization and hypoglycemic effect of a neutral polysaccharide extracted from the residue of Codonopsis Carbohyd Polym 197: 215-226.
- LIAO Y, ZENG L, LI P (2017) Influence of Plant Growth Retardants on Quality of Codonopsis Radix. Molecules 22(10): 1655.
- HU Y, WANG J (2019) Interactions between clopidogrel and traditional Chinese medicine. J Thromb Thrombolysis 48(3): 491-499.

 Research Article
Research Article