Abstract
Atherosclerosis is a progressive process that leads to ischemic injury to body organs. The basis of the disease is lipid accumulation in the arterial wall that results in narrowing of the artery lumen. Several pathogenetic mechanisms are involved in the development of atherosclerosis. Chronic inflammation and increased oxidative stress induce structural and functional damage of the vascular endothelium and contribute to atherosclerosis progression, where several antioxidants exert varying extents of anti-atherogenic effects. Heparan sulfate proteoglycans are cell surface and extracellular matrix macromolecules that play a key role in maintaining tissue integrity, besides cellular-extracellular matrix communication, towards keeping adequate structure and function of tissues and organs. These macromolecules undergo cleavage by the enzyme heparanase- the enzyme responsible for the degradation of heparan sulfate chains. Recent studies implicated heparanase in atherosclerosis development and progression, and pathophysiologic pathways have been suggested.
In this manuscript, we shed light on the knowledge regarding involvement of heparanase in atherosclerosis and focus on how heparanase inhibition may constitute an effective strategy to attenuate atherosclerosis progression.
Keywords:Heparanase; Heparan Sulfate Proteoglycans; Atherosclerosis; Oxidative Stress; E0 Mice
Abbreviations: AS: Atherosclerosis; Ox- LDL: Oxidized LDL; ECM: Extra-Cellular Matrix; OS: Oxidative Stress; HSPGs: Heparan Sulfate Proteoglycans; HFD: High Fat Diet; AKI: Acute Kidney Injury; TFPI: Tissue Factor Pathway Inhibitor
Atherosclerosis- Knowledge and Pathogenesis
Atherosclerosis (AS) is the process in which lipid particles,
mainly oxidized LDL (Ox-LDL) accumulate in the luminal side of the
arterial walls, then enclosed with a fibrous cap, which is composed
of hypertrophic endothelial cells, collagen and extra-cellular matrix
(ECM) proteins. Together, the lipid core and the surrounding fibrous
cap form the atherosclerotic plaques, which cause progressive
narrowing of the arterial lumen, resulting in impaired blood flow
and oxygen supply, leading to ischemic injury in the affected organs
[1]. Being a multifactorial process, AS is accelerated in the presence
of several cardiovascular risk factors, such as diabetes mellitus,
hypertension, hyperlipidemia, complex genetic susceptibility to
the disease, and tobacco abuse. In these diseases, higher levels
of oxidative stress (OS) occur frequently, which lead to advanced
disease and a higher rate of target organ damage and diseaserelated
complications. OS augments the inflammatory response
in the vascular endothelium, which results in vascular endothelial
structural and functional damage, thus leading to accelerated AS
[2,3].
A proof for the role of OS in the progression of AS is that
multiple endogenous and exogenous antioxidants had been
proven to attenuate AS progression [4-6]. Heparan sulfate
proteoglycans (HSPGs) are macromolecules that are composed of
glycosaminoglycan chains covalently bound to a protein core and
are either embedded in cell membranes or located in the ECM
[7]. HSPGs exert important functions in cell-ECM interaction [8-
10] and play key roles both in normal biologic processes [11-16] as well as in several pathologic processes [7,11,12,17-23], and
their normal structure and function are crucial for maintaining
normal tissue structure, integrity and function [24]. Heparan
sulfate endoglycosidase heparanase (Heparanse), the only enzyme
in mammalians that degrades HS chains in the HSPGs [19,25-27],
is implicated in the tight regulation of HSPG turnover, through
both intracellular and extracellular roles [28-30]. As HSPGs exist
in all the body organs and systems, heparanase inhibition is
under extensive investigation in various diseases. Recent studies
implicate heparanase in AS development and progression. In their
study, Blich, et al. demonstrated intense staining for heparanase in
the intima of vulnerable atherosclerotic plaques in human coronary
arteries compared to a weak staining in stable plaques [31].
Similarly, Osterholm, et al. demonstrated a 6.6 fold increased
heparanase mRNA levels in atherosclerotic plaques in human
carotid arteries in comparison to non-atherosclerotic iliac arteries
[23]. Likewise, Baker, et al. documented elevated heparanase levels
and activity associated with coronary atherosclerosis progression
in diabetic hyperlipidemic swine [22]. In their review, Vlodavsky,
et al. reported in detail the role of heparanase in atherosclerosis
and other vessel wall pathologies [32]. In line with these studies,
we demonstrated in apolipoprotein E deficient (E0) mice that
heparanase inhibition by PG545 (Pixatimod) significantly
decreased serum OS and lowered plasma lipid levels [33]. In
addition, we demonstrated in E0 mice placed on high fat diet (HFD)
that heparanase inhibition by PG545 significantly decreased serum
OS, along decreasing aortic wall thickness and atherosclerotic
plaque surface area. In the same study, we demonstrated that
PG545 significantly diminished the development of liver steatosis,
an issue under current investigation [34].
In biochemical staining and western blotting studies, we
demonstrated that PG545 caused significant reduction of IL-
1, TNF-α, and aKT, together with increasing FGF-2 and LC-III
expression, reflecting the fact that heparanase inhibition resulted
in anti-inflammatory effects, besides augmenting regeneration
process and increased autophagy, in an attempt of the liver to
repair injured liver tissue. All these effects, together with lowering
serum OS and lipid levels, can be suggested as the pathogenetic
mechanisms by which heparanase inhibition exerts the beneficial
anti-atherosclerotic and anti-steatosis effects (Figure 1). Moreover,
we also studied the effect of Roneparstat (SST0001) on OS, AS, and
liver steatosis in E0 mice placed on HFD for eight weeks. SST0001
showed metabolic effects similar to PG545 (decreased serum
lipids levels and OS, and significantly attenuated the development
of liver steatosis), but had no effect against the development of
atherosclerosis in the aortas of the mice. Like PG545, also SST0001
demonstrated neither heptotoxicity nor renal toxicity, and did
not affect blood pressure. In contrast to PG545, which caused
prominent weight loss in mice despite minimal effect on food
intake, SST0001 affected neither food intake nor mice body weight
(data not published).
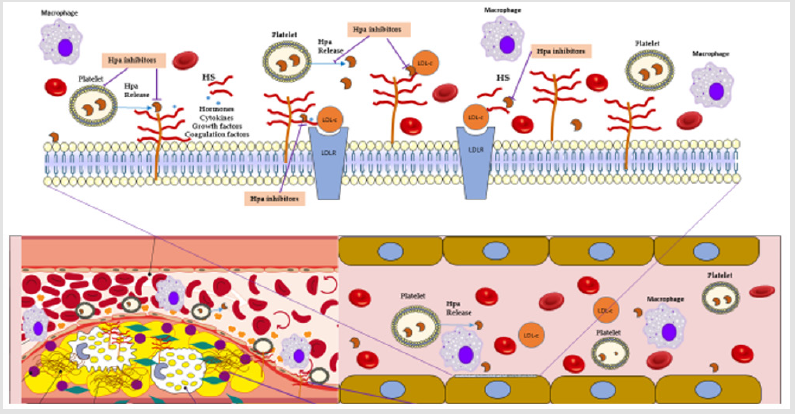
Figure 1: A schematic presentation presenting a long-cut view for an artery. Endothelial cell layer are shown, with a magnification of the luminal endothelia cell membrane, showing components of cell wall- including embedded LDL receptors and heparan sulfate proteoglycan molecules, as well as heparanase molecules and intra-luminal blood cellular components.
Heparanase Inhibition in Acute Kidney Injury
In ischemia-reperfusion acute kidney injury (AKI) rat model, pre-treatment with PG545 significantly attenuated the development of AKI, which was expressed by lower serum creatinine and blood urea nitrogen levels in the treated mice. Histologic studies of the kidneys in the sham-control mice revealed the development of acute tubular necrosis, with tubular lysis, loss of brush border and the accumulation of debris in the tubular lumen. Electron microscopy analyses revealed mitochondrial distortion in the sham-control mice, compared to normal ultrastructure of the mitochondria in mice pre-treated with the heparanase inhibitor [35].
Heparanase Inhibition in Malignant Diseases
The role of heparanase in the development of malignant diseases was well established, and the effect of heparanase inhibition in the treatment of different malignancies has been extensively studied. Recent studies demonstrated favorable effects of the heparanase inhibitor SST0001 (Roneparstat) when added to standard treatment protocols in patients with multiple myeloma [36-38] or in different kinds of human pediatric osteosarcoma and other sarcoma models [39,40], as well as in cases of mesothelioma [41] and different pediatric malignancies [42], and thus heparanase inhibitors are under consideration as an additional modality for treating malignant diseases [43-45].
Heparanase Inhibitors and the Coagulation System
Impaired endothelial dysfunction is an early feature of formation
of the atherosclerotic lesion. Acute cardiac ischemic syndromes,
such as unstable angina pectoris or myocardial infarctions, occur
following the abrupt rupture of an atherosclerotic plaque, an
event that leads to exposure of the highly thrombogenic lipid core
constituents of the plaque to the intra-vascular content, resulting
in activation of the coagulation system, which further accelerates
the atherosclerotic plaque formation [46-48]. Heparan sulfate
molecules play key roles in activation of the coagulation system.
In a book chapter, Nadir Y. had described in detail the role
of heparanase in the activation of the coagulation system and
reported increased heparanase level and activity in several clinical
settings associated with hypercoagulability, such as in women
using oral contraceptives, cases of diabetic foot, women at delivery,
after orthopedic surgeries, and in patients with lung cancers and
other malignancies [49]. Former studies have reported in detail the
role of heparanase in increasing coagulation via three mechanisms,
including enhancement of tissue factor activity, upregulation of
tissue factor expression in endothelial cells, and by releasing the
single chain polypeptide tissue factor pathway inhibitor (TFPI)
from cell surface [50], and heparanase inhibition by peptides
derived from TFPI-2 was proved to inhibit the procoagulant activity
of heparanase and to attenuate sepsis in mouse model [51].
Conclusion
There is a large evidence regarding the involvement of
heparanase in several disease processes, supported by the fact
that higher heparanase levels and activity are associated with
more advanced and complicated diseases, as was shown in
several malignancies, diabetes mellitus, nephropathies of variable
etiologies, and infectious and inflammatory processes. Likewise,
several recent studies had implicated heparanase in atherosclerosis
development and progression. Many basic science and clinical
studies were held and others being ongoing, to explore the effect
of heparanase inhibition on malignant diseases. Conversely, only
few studies have reported the effect of heparanase inhibition
on atherosclerosis formation and progression. Undoubtedly,
heparanase inhibition exerts favorable effect towards attenuating
atherosclerosis progression, in spite of the contradictory results
obtained so far.
The effect of heparanase inhibition on AS is expressed by
significant attenuation in the development of aortic atherosclerotic
plaques and reducing aortic wall thickness, through several
possible mechanisms, which include lowering of serum lipid values,
decreasing serum oxidative stress, and anti-inflammatory effects.
However, lack of pathogenic mechanism and effect of long term use
of heparanase inhibitors in humans limit the possibility to apply
using heparanase inhibitors in humans for prevention or treating
atherosclerosis, and further research is absolutely warranted. In
the literature, additional heparanase inhibitors are available, where
their effect was not examined neither on AS nor on liver steatosis. In
face of the existing non-consistent results demonstrated so far, it is
highly warranted that the effect of additional heparanase inhibitors
on atherosclerosis prevention and treatment be studied.
References
- Jacob Fog Bentzon, Fumiyuki Otsuka, Renu Virmani, Erling Falk (2014) Mechanisms of plaque formation and rupture. Circ Res 114(12): 1852-1866.
- Halliwell B (1994) Free radicals, antioxidants, and human disease:curiosity, cause, or consequence?. TheLancet 344(8924): 721-724.
- Kaplan, Aviram M (1999) Oxidized low density lipoprotein:Atherogenic and proinflammatory characteristics during macrophage foam cell formation. An inhibitory role for nutritional antioxidants and serum paraoxonase. Clinical Chemistry and Laboratory Medicine 37: 777-787.
- Nigdikar SV, Williams NR., Griffin BA, Howard AN (1998) Consumption of red wine polyphenols reduces the susceptibility of low- density lipoproteins to oxidation in vivo. American Journal of Clinical Nutrition 68(2): 258-265.
- Shadi Hamoud, Tony Hayek, Nina Volkova, Judith Attias, Danit Moscoviz, et al. (2014) Pomegranate extract (POMx) decreases the atherogenicity of serum and of human monocyte-derived macrophages (HMDM) in simvastatin-treated hypercholesterolemic patients: A double-blinded, placebo-controlled, randomized, prospective pilot study. Atherosclerosis 232: 204-210.
- Vaya J, Belinky PA, Aviram M (1997) Antioxidant constituents from licorice roots: Isolation, structure elucidation and antioxidative capacity toward LDL oxidation. Free Radical Biology & Medicine 23(2): 302-313.
- Vlodavsky I, Blich M, Li JP, Sanderson RD, Ilan N (2013) Involvement of heparanase in atherosclerosis and other vessel wall pathologies.Matrix Biol 32(5): 241-251.
- Hamoud S, Kaplan M, Meilin E, Kaplan M, Torgovicky R, et al. (2013) Niacin administration significantly reduces oxidative stress in patients with hypercholesterolemia and low levels of high-density lipoprotein cholesterol. The American Journal of the Medical Sciences 345(3) : 195-199.
- Bernfield M, Götte M, Park PW, Reizes O, Fitzgerald ML, et al.(1999) Functions of cell surface heparan sulfate proteoglycans. Annual Review of Biochemistry 68: 729-777.
- Ilan N, Elkin M, Vlodavsky I (2006) Regulation, function and clinical significance of heparanase in cancer metastasis and angiogenesis. The International Journal of Biochemistry & Cell Biology 38(12): 2018-2039.
- Vlodavsky I, Friedmann Y (2001) Molecular properties and involvement of heparanase in cancer metastasis and angiogenesis. J Clin Invest 108(3): 341-347.
- Parish CR, Freeman C, Hulett MD (2001) Heparanase: a key enzyme involved in cell invasion. Biochim Biophys Acta 1471(3) : M99-M108.
- Barash U, Cohen Kaplan V, Dowek I, Sanderson RD, Ilan N, et al. (2010) Proteoglycans in health and disease: new concepts for heparanase function in tumor progression and metastasis. FEBS J 277(19): 3890-3903.
- Sandwall E, O Callaghan P, Zhang X., Lindahl U, Lannfelt L, et al. (2010) Heparan sulfate mediates amyloid-beta internalization and cytotoxicity. Glycobiology 20(5): 533-541.
- Zetser A., Bashenko Y, Edovitsky E, Levy Adam F, Vlodavsky I, et al. (2006) Heparanase induces vascular endothelial growth factor expression: correlation with p38 phosphorylation levels and Src activation. Canc Res 66(3): 1455-1463.
- Vlodavsky I, Beckhove P, Lerner I, Pisano C, Meirovitz A, et al. (2012) Significance of heparanase in cancer and inflammation. Canc Microenviron 5(2): 115-132.
- Lygizos MI, Yang Y, Altmann CJ, Okamura K, Hernando AA, et al. (2013) Heparanase mediates renal dysfunction during early sepsis in mice. Phys Rep 1(6): e00153
- Goldberg R, Rubinstein AM, Gil N, Hermano E, Li JP, et al. (2014) Role of heparanase-driven inflammatory cascade in pathogenesis of diabetic nephropathy. Diabetes 63(12): 4302-4313.
- Shafat I, Ilan N, Zoabi S, Vlodavsky I, Nakhoul F (2011) Heparanase levels are elevated in the urine and plasma of type 2 diabetes patients and associate with blood glucose levels. PLoS One 6(2): e17312.
- Levidiotis V, Kanellis J, Ierino FL, Power DA (2001) Increased expression of heparanase in puromycin aminonucleoside nephrosis. Kidney Int 60(4): 1287-1296.
- Masola V, Zaza G, Gambaro G, Onisto M, Bellin G, et al. (2016) Heparanase: a potential new factor involved in the renal epithelial mesenchymal transition (EMT) induced by ischemia/reperfusion (I/R) injury. PloS One 11(7): e0160074.
- Baker AB, Chatzizisis YS, Beigel R, Jonas M, Stone BV, et al. (2010) Regulation of heparanase expression in coronary artery disease in diabetic, hyperlipidemic swine. Atherosclerosis 213(2): 436-442.
- Osterholm C, Folkersen L, Lengquist, M, Ponten F, Renne T, et al. (2013) Increased expression of heparanase in symptomatic carotid atherosclerosis. Atherosclerosis 226(1): 67-73.
- Stephane Sarrazin, William C Lamanna, Jeffrey D Esko (2011) Heparan Sulfate Proteoglycans. Cold Spring Harb Perspect Biol 3(7): a004952.
- Levidiotis V, Kanellis J, Ierino FL, Power DA (2001) Increased expression of heparanase in puromycin aminonucleoside nephrosis. Kidney International 60(4) :1287-1296.
- Masola V, Zaza G, Gambaro G, et al. (2016) Heparanase: a potential new factor involved in the renal epithelial mesenchymal transition (EMT) induced by ischemia/reperfusion (I/R) injury. PloS ONE 11(7): e0160074.
- Kramer A, Van Den Hoven M, Rops A, Wijnhoven T, Heuvel L, et al. (2006) Induction of glomerular heparanase expression in rats with adriamycin nephropathy is regulated by reactive oxygen species and the renin-angiotensin system. Journal of the American Society of Nephrology 17(9): 2513-2520.
- Valentina Masola, Gloria Bellin, Giovanni Gambaro, Maurizio Onisto (2018) Heparanase: A Multitasking Protein Involved in Extracellular Matrix (ECM) Remodeling and Intracellular Events. Cells 7(12): 236.
- Jun Shu, Gaetano Santulli (2019) Heparanase in health and disease: The neglected housekeper of the cell? Atherosclerosis 283: 124-126.
- Rabelink T, van den Berg B, Garsen M, Wang G, Elkin M, et al. (2017) Heparanase: roles in cell survival, extracellular matrix remodelling and the development of kidney disease. Nat Rev Nephrol 13: 201-212.
- Blich M, Golan A, Arvatz G, Sebbag A, Shafat I, et al. (2013) Macrophage activation by heparanase is mediated by TLR-2 and TLR-4 and associates with plaque progression. Arterioscler Thromb Vasc 33(2): E56-65.
- Israel Vlodavsky, Miry Blich, Jin Ping Li, Ralph D Sanderson, et al. (2013) Involvement of heparanase in atherosclerosis and other vessel wall pathologies. Matrix Biol 32(5): 241-251.
- Shadi Hamoud, Rabia Shekh Muhammad, Niroz Abu Saleh, Ahmad Hassan, Yaniv Zohar, et al. (2017) Heparanase Inhibition Reduces Glucose Levels, Blood Pressure, and Oxidative Stress in Apolipoprotein E Knockout Mice. Biomed Res Int 2017: 7357495.
- Rabia Shekh Muhammad, Niroz Abu Saleh, Safa Kinaneh, Mohammad Agbaria, Edmond Sabo, et al. (2018) Heparanase inhibition attenuates atherosclerosis progression and liver steatosis in E0 Atherosclerosis 276: 155-162.
- Zaid Abassi, Shadi Hamoud, Ahmad Hassan, Iyad Khamaysi, Omri Nativ, et al.(2017) Involvement of heparanase in the pathogenesis of acute kidney injury: nephroprotective effect of PG545. Oncotarget 8(21): 34191-34204.
- Ritchie JP, Ramani VC, Ren Y, Naggi A, Torri G, et al. (2011) SST0001, a chemically modified heparin, inhibits myeloma growth and angiogenesis via disruption of the heparanase/syndecan-1 axis. Clin Cancer Res 17(6): 1382-1393.
- Monica Galli, Manik Chatterjee, Mariella Grasso, Giorgina Specchia, Hila Magen, et al. (2018) Phase I study of the heparanase inhibitor roneparstat: an innovative approach for multiple myeloma therapy. Haematologica 103(10): e469-e472.
- Joseph P Ritchie, Vishnu C Ramani, Yongsheng Ren, Annamaria Naggi, Giangiacomo Torri, et al. (2011) SST0001, a chemically modified heparin, inhibits myeloma growth and angiogenesis via disruption of the heparanase/syndecan-1 axis. Clin Cancer Res 17(6): 1382-1393.
- Giuliana Cassinelli, Enrica Favini, Laura Dal Bo, Monica Tortoreto, Marcella De Maglie, et al. (2016) Antitumor efficacy of the heparan sulfate mimic roneparstat (SST0001) against sarcoma models involves multi-target inhibition of receptor tyrosine kinases. Oncotarget 7(30): 47848-47863.
- Itay Shafat, Myriam Weyl Ben Arush, Josephine Issakov, Isaac Meller, Inna Naroditsky, et al. (2011) Pre-clinical and clinical significance of heparanase in Ewing’s sarcoma. J Cell Mol Med 15(9): 1857-1864.
- Barash U, Lapidot M, Zohar Y, Loomis C, Moreira A, et al. (2018) Involvement of Heparanase in the Pathogenesis of Mesothelioma: Basic Aspects and Clinical Applications. J Natl Cancer Inst 110(10): 1102-1114.
- Itay Shafat, Ayelet Ben Barak, Sergey Postovsky, Ronit Elhasid, Neta Ilan, et al. (2007) Heparanase Levels Are Elevated in the Plasma of Pediatric Cancer Patients and Correlate with Response to Anticancer Treatment. Neoplasia 9(11): 909-916.
- Anna Shteingauz, Ilanit Boyango, Inna Naroditsky, Edward Hammond, Maayan Gruber, et al. (2015) Heparanase enhances tumor growth and chemo-resistance by promoting autophagy. Cancer Res 75(18): 3946-3957.
- Israel Vlodavsky, Preeti Singh, Ilanit Boyango, Lilach Gutter-Kapon, Michael Elkin, et al. (2016) Heparanase: from basic research to therapeutic applications in cancer and inflammation. Drug Resist Updat 29: 54-75.
- Ralph D Sanderson, Michael Elkin, Alan C Rapraeger, Neta Ilan, Israel Vlodavsky (2017) Heparanase regulation of cancer, autophagy and inflammation: New mechanisms and targets for therapy. FEBS J 284(1): 42-55.
- Sang-Min Kim, Jae Wan Huh, Eun Young Kim, Min Kyung Shin, Ji Eun Park, et al. (2019) Endothelial dysfunction induces atherosclerosis: increased aggrecan expression promotes apoptosis in vascular smooth muscle cells. BMB Rep 52(2): 145-150.
- Fernández-Ortiz A, Badimon JJ, Falk E, Fuster V, Meyer B, et al. (1994) Characterization of the relative thrombogenicity of atherosclerotic plaque components: implications for consequences of plaque rupture. J Am Coll Cardiol 23: 1562-1569.
- Rautou PE, Vion AC, Amabile N, Chironi G, Simon A, et al. (2011) Microparticles, vascular function, and atherothrombosis. Circ Res 109: 593-606.
- Nadir Y (2020) Heparanase in the Coagulation System. In: Vlodavsky I, Sanderson R, Ilan N (Eds) Heparanase. Advances in Experimental Medicine and Biology. Springer, Cham 1221.
- Yona Nadir (2014) Heparanase and Coagulation-New Insights. Rambam Maimonides Med J 5(4): e0031.
- Axelman E, Henig I, Crispel Y, Attias J, Li JP, et al. (2014) Novel peptides that inhibit heparanase activation of the coagulation system. Thromb Haemost 112(3): 466-477.

 Mini Review
Mini Review