Abstract
Objective: The most important problem associated with Nipple-Sparing Mastectomy (NSM) and Skin-Sparing Mastectomy (SSM) for breast cancer patients is the risk of local recurrence. Local recurrence is associated with concurrent or following distant metastasis and mortality. Patients also suffer from second operation and worse quality of life if local recurrence or distant metastasis occurs. Long-term follow-up data regarding the oncologic safety of NSM and SSM with cancer recurrence at the Nipple-Areola-Complex (NAC) and survival are limited. This is a single center retrospective review and reported as an observational result.
Methods: A retrospective review of 168 female breast cancer patients who had undergone SSM or NSM with immediate breast reconstruction in Chang Gung Memorial Hospital, Linkou, from January 1, 2014 to November 2, 2018, was performed. The focus of this review is simply to determine the local recurrence rate after SSM or NSM in breast cancer patients with immediate reconstruction.
Results: Overall, 172 mastectomies (59 SSM and 113 NSM) of 168 female breast cancer patients were performed. Among these 172 mastectomies, 150 were immediate reconstruction with autologous flap or one-stage prosthesis implantation, 22 were two-stage tissue expander breast reconstructions followed by prosthesis implantation months later. Median follow-up duration was 51 months, ranged from 24 months to 78 months. Local recurrence was found in only one patient. The recurrence location was below NAC (Nipple-Areola-Complex) and it was 45 months after first surgery. There were two patients with distant metastasis during the follow-up period. However, these two patients with distant metastasis had no evidence of local recurrence.
Conclusion: SSM and NSM with immediate reconstruction are both safe surgical intervention methods compared with traditional simple mastectomy and breastconserving surgery for breast cancer patients regarding to local recurrence rate and longterm overall survival.
Keywords: Breast Cancer; Metastasis; Radiotherapy; Breast Reconstruction; Tumor Stage; Computed Tomography
Abbreviations: NSM: Nipple-Sparing Mastectomy; SSM: Skin-Sparing Mastectomy; NAC: Nipple-Areola-Complex; DIEP: Deep Inferior Epigastric Perforator; SIEA: Superficial Inferior Epigastric Artery Flap
Introduction
It is breast cancer that has the highest incidence among all female cancer types in Taiwan. The age at cancer diagnosis in Taiwan female breast cancer patients is much earlier than that in European and American countries. The range of age at diagnosis is between 45 and 64 years old. Young female patients care much about their appearance after therapeutic surgery. With the advancement of radiotherapy and anti-cancer drugs, the proportion of breast-conserving surgery is increasing. Breast preservation surgery accounts for two thirds of all breast cancer surgery in Taiwan. Meanwhile, the issue of oncologic safety, local recurrence, and distant metastasis in the long following life also bother these young female patients. Both SSM and NSM offer these young female patients another choice in surgical options. In 1991, SSM was first described by Toth and Lappert [1] with removing all the breast tissue and nipple-areola-complex while preserving the skin envelope [2]. The first Nipple-Sparing Mastectomy(NSM) report was from Hinton et al. [3], who mentioned that NSM achieved the same local recurrence rates and survival rates compared to radical mastectomy [1]. Many prospective and retrospective studies [4-6] of NSM have shown the oncologic and surgical safety of NSM as well as the superior aesthetic outcomes and improved quality of life achieved when NSM is with immediate breast reconstruction. Present National Comprehensive Cancer Network guidelines state that NSM is an acceptable surgical option for carefully selected patients with breast cancer [7].
Nevertheless, the application of NSM for breast cancer remains controversial because of the limited long-term follow-up data, such as the rate of local recurrence rate and distant metastasis, and mortality rate after local recurrence or distant metastasis [8]. Because increasing numbers of patients with breast cancer are selecting NSM [9,10], it is important to identify the incidence of cancer recurrence at the NAC after NSM, describe the associated factors, and determine its association with prognosis. Previous studies [11-15] have reported cancer recurrence incidence at the NAC from 0% to 3.7% after NSM. This study is to review the local recurrence rate after Nipple-Sparing Mastectomy(NSM) and Skin- Sparing Mastectomy(SSM) with immediate breast reconstruction.
Methods
A retrospective data was collected including all patients who underwent SSM and NSM followed by immediate or immediate 2-stage tissue expander breast reconstruction in our hospital from January 1, 2014 to November 2, 2018. If breast reconstruction was applied with implant, silicone Style 410 implants by Allergan (Parsippany-Troy Hills, NJ, USA) were used for subpectoral augmentation. The first choice of autologous tissue reconstruction was Deep Inferior Epigastric Perforator (DIEP) flap. If DIEP flap was not suitable, Perfundus Artery Perforator (PAP) flap would be harvested. The third flap choice was Superficial Inferior Epigastric Artery Flap (SIEA). The age at diagnosis, type of surgery, tumor stage, node stage, histologic grade, subtype, and tumor histologic type were reviewed. Tumor and node staging were conducted according to the 8th edition of the American Joint Committee on Cancer Cancer Staging Manual [16]. These 168 patients all have regular followups. The patients were regularly followed up postoperatively every 3 to 6 months for the first 5 years and 6 to 12 months 5 years after surgery if no recurrence or metastasis occurs. Local recurrence or distant metastasis was identified by clinical physical examination, breast ultrasonography, mammography, or chest radiography. Abnormal clinical findings were evaluated through further studies, including computed tomography of the chest, bone scan, and liver ultrasonography. Core needle biopsy was performed to evaluate suspicious lesion below the NAC.
Results
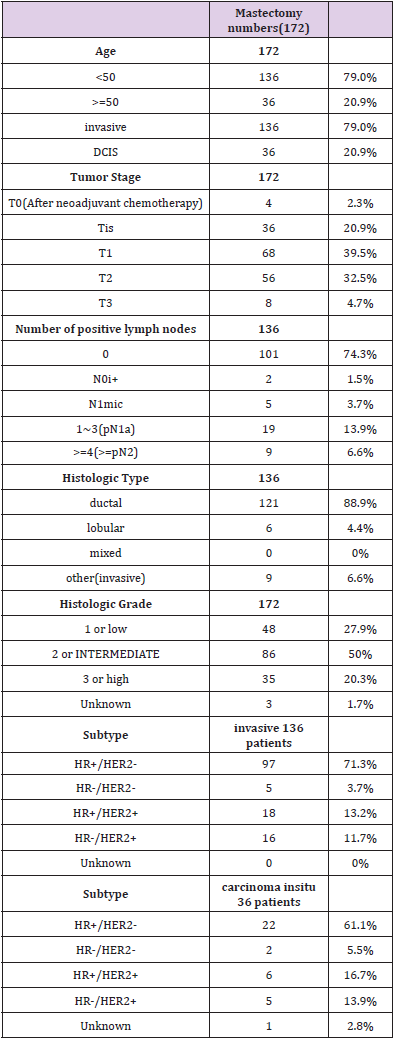
Overall, 172 mastectomies (59 SSM and 113 NSM) of 168 female breast cancer patients were performed. Among these 172 mastectomies, 150 were immediate reconstruction with permanent autologous tissue or one-stage prosthesis implantation, 22 were two-stage tissue expander breast reconstruction followed by prosthesis implantation months after adjuvant chemotherapy and radiation therapy. Median follow-up duration was 51 months, ranged from 24 months to 78 months. Local recurrence was found in only one patient. The recurrence location was below NAC (nipple-areola-complex) and it was 45 months after first surgery. There were two patients who developed distant metastasis during the follow up period. However, these two patients with distant metastasis had no evidence of local recurrence. The basic characteristics of these 168 breast cancer patients are shown in Table 1. The median age of patients at diagnosis was 44 years (range, 27-76 years). One hundred and thirty-six patients were less than fifty years old. Thirty-six patients were older than fifty years old. One hundred and thirty-six patients had invasive carcinoma and thirty-six patients had carcinoma in situ. Among the 136 patients with invasive disease, 101 were node negative, 2 had malignant cells in regional lymph node(s) no greater than 0.2 mm(pN0i+), 5 had micrometastases lymph nodes(N1mic), 19 had pN1a, 9 had more than pN1. As regard to histologic type, among the 136 patients with invasive disease, 121 had invasive ductal carcinoma, 6 had invasive lobular carcinoma. There were 9 patients had other invasive types including invasive tubular carcinoma and invasive mucinous carcinoma. Most of the histologic grade is intermediate grade or grade 2. In all 172 mastectomies, 48 were low grade or grade 1, 86 were intermediate grade or grade 2, 35 were high grade or grade 3, and 3 were unknown Table 2.
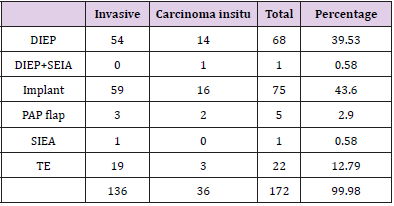
As for reconstruction type, most of patients chose prosthesis implantation. In 172 mastectomies with reconstructions, 75(43.6%) had prosthesis implantation, 68(39.5%) had DIEP flap reconstruction, 5(2.9%) had PAP flap reconstruction, 1(0.58%) had SIEA flap reconstruction, and 1(0.58%) had DIEP plus SIEA flap reconstruction. The only one patient who later developed local recurrence was a 38-year-old lady. In March 2014, she had a diagnosis of DCIS (ductal carcinoma in situ). Following NSM, she had immediate DIEP flap reconstruction. She had a histologic type of negative estrogen receptor, negative progesterone receptor, and positive type 2 Human Epidermal Growth Factor Receptor (HER2). She did not receive anti-HER2 therapy after first mastectomy because the histologic type was DCIS. But later in December 2017, she had a mass right below the NAC. After wide excision, it was proved that she had an invasive ductal carcinoma with pathologic T2 (tumor over than 2 centimeters) status this time. The duration from surgery to cancer recurrence at the NAC was 45 months. Sentinel lymph node excision was negative for metastasis in both frozen section and permanent pathology. This time, she had a histologic type of positive estrogen receptor, negative progesterone receptor, and still positive type 2 human epidermal growth factor receptor (HER2). Therefore, adjuvant therapy for this patient included anti-hormone therapy with Tamoxifen (10mg BID), chemotherapy with twelve times of weekly Paclitaxel, and eighteen times of anti- HER2 target therapy with Trastuzumab. Because she had ductal carcinoma in situ in 2014 when the first time she was diagnosed with breast DCIS, strictly speaking, she had a new cancer instead of recurrence in 2017. There were two distant metastasis patients but there was no evidence of local recurrence.
Discussion
There is much more acceptance of skin-sparing mastectomy or nipple-sparing mastectomy with immediate breast reconstruction for the surgical treatment of breast cancer. Because skin-sparing mastectomy or nipple-sparing mastectomy with immediate breast reconstruction achieve almost excellent aesthetic outcomes and improved much in quality of life without compromising oncologic safety. However, SSM was first introduced in 1991[1], limited longterm follow-up data are rarely available regarding the oncologic safety. The data of NSM is even less. Patients who have undergone either mastectomy or breast-conserving surgery and Radiation Therapy (RT) are at risk for locoregional recurrence. For women treated for early breast cancer, the recurrence rate ranges from 4 to 7 percent after mastectomy or breast-conserving therapy, respectively [17]. If locoregional recurrence occurs within two years after primary treatment, distant metastatic disease is already present in 25 to 30 percent of cases [18]. Available studies [11- 15,19-22] have shown low rates of cancer recurrence at the NAC (0%-3.7%) after NSM. The recurrence rate (4%-7%) after NSM was even lower than that after traditional mastectomy or breastconserving surgery [17]. Most of these findings were obtained in a heterogeneous population of patients, including those with invasive and noninvasive disease, and reported variable follow-up durations. In a series by Jensen et al. [21], no cancer recurrence at the NAC was reported among 149 patients who underwent NSM during a mean follow-up of 60.2 months; however, 57% of these cases had only noninvasive disease. In a study by Wang et al. [22], no case of cancer recurrence at the NAC was found among 981 patients who underwent NSM; however, the follow-up evaluation was only 29 months, and 52% of the surgeries were performed for in situ disease [20]. During a median follow-up duration of 78 months, Sakurai et al. [13] reported a cancer recurrence rate at the NAC of 3.7% among 788 patients who underwent NSM without radiotherapy between 1985 and 2004. In one study [8], it included patients with invasive breast cancer who underwent NSM and immediate breast reconstruction between 2003 and 2015 and identified a 5-year local recurrence at the NAC of 3.5%.
The indication for adjuvant radiation therapy after breast cancer treatment surgeries in our hospital include breastconserving surgery, primary tumor larger than 5 centimeters, final pathology showing lymphovascular invasion, and more than two metastatic ipsilateral axillary lymph nodes found. So most of our study populations did not undergo adjuvant radiation therapy after SSM or NSM. It was because the whole breast tissue was removed. Only two studies investigated variables through a multivariate analysis. One of these two studies was published by Petit et al. [22]. There were 934 NSMs performed for invasive and intraepithelial breast cancer with a follow-up duration of 50 months. Eleven cases of local cancer recurrence at the NAC were found. The tumor size, receptor status, ERBB2 status, histologic grade, and Ki-67 proliferation index were associated with the risk of recurrence in a multivariate analysis. Regarding the surgical technique for NSM, Petit et al. [22] mentioned that they leaved a 5-mm-thick layer of glandular tissue below the NAC to avoid malperfusion and for the intraoperative delivery of electron-beam radiotherapy exclusively to the NAC to minimize the risk of local recurrence.
In the other study by Zhen-Yu Wu, et al. [8], in multivariate analysis, multifocality or multicentricity, negative hormonal receptor status, ERBB2-positive status, high histologic grade, and presence of extensive intraductal component were associated with an increased risk of cancer recurrence at the NAC after NSM. This study showed that multifocality or multicentricity and presence of extensive intraductal component has the strongest associations with cancer recurrence at the NAC. The association between tumornipple distance and cancer recurrence at the NAC did not show significance. Our data reveal our experience with the treatment and outcomes of cancer recurrence at the NAC after NSM and SSM. The only one patient with cancer recurrence at the NAC underwent wide local excision, and then received multimodal adjuvant treatment according to the biologic disease features, including hormonal therapy, chemotherapy, and target therapy. This patient has no more evidence of local cancer recurrence or distant metastasis after multimodal adjuvant treatment after cancer treatment surgery in 2017.
Conclusion
Nipple-sparing mastectomy and skin-sparing mastectomy are both safe surgical treatment options for breast cancer patients compared with traditional mastectomy and breast-conserving surgery. There is lower local recurrence rate and comparable distant metastatic rate. Nipple-Sparing Mastectomy and skin-sparing mastectomy definitely reveal much better aesthetic outcomes and quality of life in breast cancer patients.
Limitations
The limitation of our data is that it is retrospective review. Our patient numbers are small and populations are heterogeneous.
References
- Toth BA, Lappert P (1991) Modified skin incisions for mastectomy: the need for plastic surgical input in preoperative planning. Plast Reconstr Surg 87(6): 1048-1053.
- Agha RA, Wellstead G, Sagoo H, Al Omran Y, Barai I, et al. (2016) Nipple sparing versus skin sparing mastectomy: a systematic review protocol. BMJ Open 6(5).
- Hinton CP, Doyle PJ, Blamey RW, Davies CJ, Holliday HW, et al. (1984) Subcutaneous mastectomy for primary operable breast cancer. Br J Surg 71(6): 469-472.
- Kim HJ, Park EH, Lim WS, Jin Young Seo, Beom Suk Koh et al. (2010) Nipple areola skin-sparing mastectomy with immediate transverse rectus abdominis musculocutaneous flap reconstruction is an oncologically safe procedure: a single center study. Ann Surg 251(3): 493-498.
- Moo TA, Pinchinat T, Mays S, Alyssa Landers, Paul Christos, et al. (2016) Oncologic outcomes after nipple-sparing mastectomy. Ann Surg Oncol 23(10): 3221-3225.
- Bailey CR, Ogbuagu O, Baltodano PA, Usamah F Simjee, Michele A Manahan, et al. (2017) Quality-of-life outcomes improve with nipple-sparing mastectomy and breast reconstruction. Plast Reconstr Surg 140(2): 219-226.
- (2017) National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Breast Cancer.
- Zhen Yu Wu, Hee Jeong Kim, Jong Won Lee, Il-Yong Chung, Ji Sun Kim, et al. (2019) Breast Cancer Recurrence in the Nipple-Areola Complex After Nipple-Sparing Mastectomy With Immediate Breast Reconstruction for Invasive Breast Cancer JAMA Surg 154(11): 1030-1037.
- Agarwal S, Agarwal S, Neumayer L, Agarwal JP (2014) Therapeutic nipple-sparing mastectomy: trends based on a national cancer database. Am J Surg 208(1): 93-98.
- Frey JD, Salibian AA, Karp NS, Choi M (2018) Comparing therapeutic versus prophylactic nipple-sparing mastectomy: does indication inform oncologic and reconstructive outcomes? Plast Reconstr Surg 142(2): 306-315.
- Smith BL, Tang R, Rai U, Jennifer K Plichta, Amy S C, et al. (2017) Oncologic safety of nipple-sparing mastectomy in women with breast cancer. J Am Coll Surg 225(3): 361-365.
- Krajewski AC, Boughey JC, Degnim AC, James W, Steven R, et al. (2015) Expanded indications and improved outcomes for nipple-sparing mastectomy over time. Ann Surg Oncol 22(10): 3317-3323.
- Sakurai T, Zhang N, Suzuma T, Teiji Umemura, Goro Yoshimura, et al. (2013) Long-term follow-up of nipple-sparing mastectomy without radiotherapy: a single center study at a Japanese institution. Med Oncol 30(1): 481-483.
- Orzalesi L, Casella D, Santi C, Lorenzo Cecconi, Roberto Murgo, et al. (2016) Nipple sparing mastectomy: surgical and oncological outcomes from a national multicentric registry with 913 patients (1006 cases) over a six year period. Breast 25: 75-81.
- De La Cruz L, Moody AM, Tappy EE, Blankenship SA, Hecht EM (2015) Overall survival, disease-free survival, local recurrence, and nipple-areolar recurrence in the setting of nipple-sparing mastectomy: ameta-analysis and systematic review. Ann Surg Oncol 22(10): 3241-3249.
- Edge S Byrd DR, Compton CC (2010) AJCC Cancer Staging Manual. 7th ed New York Springer.
- Yang SH, Yang KH, Li YP, Y C Zhang, X D He, et al. (2008) Breast conservation therapy for stage I or stage II breast cancer: a meta-analysis of randomized controlled trials. Ann Oncol 19: 1039-1044.
- Kramer R, Osborne CK (2004) Diseases of the Breast. 3rd edition Lippincott Williams and Wilkins Philadelphia
- Shimo A, Tsugawa K, Tsuchiya S, Reiko Yoshie, Kyoko Tsuchiya, et al. (2016) Oncologic outcomes and technical considerations of nipple-sparing mastectomies in breast cancer: experience of 425 cases from a single institution. Breast Cancer 23(6): 851-860.
- Jensen JA, Orringer JS, Giuliano AE (2011) Nipple-sparing mastectomy in 99 patients with a mean follow-up of 5 years. Ann Surg Oncol 18(6): 1665-1670.
- Wang F, Peled AW, Garwood E, Allison Stover Fiscalini, Hani Sbitany, et al. (2014) Total skin-sparing mastectomy and immediate breast reconstruction: an evolution of technique and assessment of outcomes. Ann Surg Oncol 21(10): 3223-3230.
- Petit JY, Veronesi U, Orecchia R, G Curigliano, P C Rey, et al. (2012) Risk factors associated with recurrence after nipple-sparing mastectomy for invasive and intraepithelial neoplasia. Ann Oncol 23(8): 2053-2058.

 Research Article
Research Article