Abstract
The current study was conducted to determine the effects of pistachio by-product (PBP) polyphenols, on oxidative stability and α-linolenic acid retention in extruded linseed during long term storage. In a completely randomized design, linseed was mixed with PBP and alfalfa hay in 80:10:10 ratios and extruded with (ELINT) or without (ELIN) tert-butylhydroquinone as a synthetic antioxidant. All quality parameters, were similar between treatments (P<0.05). The peroxide value (PV) of treatments after the extrusion process was higher in ELIN treatment (P>0.05). After extrusion, oleic, linoleic, and linolenic fatty acids were higher in ELINT treatment, and the ELIN group had higher palmitic and stearic acid content (P>0.05). Fatty acid composition of treatments changed during storage period (P>0.05), and by increasing the storage period, the α-linolenic acid content of treatment decreased (P>0.05). Moreover, as the storage period increased, the PV of treatments also increased (P>0.05). The α-linolenic acid retention of treatments was different between treatments during the storage (P>0.05). As storage increased, the level of α-linolenic acid retention decreased, and this decrease in the ELIN treatment was higher than the ELINT treatment (P>0.05). In conclusion, phenolic contents of PBP in a 10% extruded linseed, could not be useful as a natural antioxidant for the stability of extruded linseed during the extrusion process and storage period.
Keywords: Antioxidant; Extruded Linseed; Peroxide; Pistachio By-Product; Tannin
Introduction
Pistachio by-products are produced during the dehulling of the pistachio nuts and contain 64.5% soft external hull, 25% twigs, 10% leaves, and 0.5% kernel and bony shells [1,2]. Like pistachio nut and pistachio skin [3], pistachio hulls [4,5] and leaves [6] are good sources of natural phenolic and antioxidants. Phenolic contents of PBP include 7.5%-13.7% phenol and 3.5%-10% (% of dry matter) tannins [7-9] by the addition of phenolic compounds to corn grain, before the extrusion process showed corn oil stability for oxidation during extrusion, significantly increased. Moreover, [10] resulted that vegetative simple phenolic compounds can use as a natural antioxidants during the extrusion process of oat grains. Nowadays, there is an excellent interest in finding new and safe antioxidants from natural sources [11]. There are serious concerns about synthetic antioxidants, given their carcinogenic potential and link to liver damage in humans [12] .However, to date, there is not enough information about the antioxidant activity of pistachio by-product poly-phenols as use during the extrusion process of oilseeds.
In another side, in the last decade, due to increasing consumer information about a healthier animal products, there is more interested in consuming a dairy products with less deleterious saturated FA and trans-FA [13]. The simplest way to modify milk fat composition is to supplement dairy cow diets with unsaturated lipids [14], such as linseed. Among oilseeds, linseed (Linum usitatissimum) has the highest proportion of linolenic acid, averaging 18% of total seed weight and constituting 53% of total FA [15]. Altering the physical structure of oilseeds through heat treatment, like to extrusion processing technology, may help to protect the dietary FA of oilseeds from ruminal biohydrogenation [16,17]. However, there are some inherent problems in the extrusion process of an oilseed-based feed ingredient. In fact, due to the extrusion process of linseed, the rapid release of intracellular oil may lead to considerable oil loss [18,19]. Besides, the potential production of oxidized compounds and reduced shelf life due to the high ALA content of linseed is also of concern [20]. So, using an appropriate binder (absorbent) material for the production of extruded linseed can be an excellent preventative resolution to reduce oil losses [21,22] and oil oxidation during extrusion. Hence, high natural antioxidants of pistachio hull can make this suitable potential oil binder a good substitute for synthetic antioxidants [23] for the stability of linseed oil during the extrusion process and storage period. Also, alfalfa hay can be an absorbent with functional fat absorption capacity for producing extruded linseed [24]. Therefore, the objectives of this study were to produce an extruded linseed mixed with PBP and alfalfa hay product, and the evaluation of these products for quality parameters, oxidative stability, and FA profile changes during long term storage.
Materials and Methods
Products
Linseed was purchased from a local market (Mashhad, Iran). Pistachio by-product and alfalfa hay were used as absorbent materials for producing extruded linseed. Also, tertbutylhydroquinone (TBHQ) was added as a synthetic phenolic antioxidant to linseed in one treatment mixed with propylene glycol in a 1:3 ratio (TBHQ: propylene glycol). Therefore, the experimental treatments were extruded linseed without TBHQ (ELIN); and extruded linseed with 1000 mg TBHQ/kg linseed (ELINT). Before extrusion processing, at first PBP and alfalfa hay were ground to pass through a 2-mm screen. For treatment with antioxidant, linseed was mixed with TBHQ, and in the following step, a mix of linseed: PBP: alfalfa hay (by mass) (80:10:10 ratio) was prepared. All treatments were triplicated.
Extrusion Process
Extrusion was performed in a double screw extruder (DS56- III, Jinan Saixin Machinery Co., Shandong, China), consisting of three independent zones of controlled temperature in the barrel. The temperature profiles in the first and second zones were kept constant at 70 and 80 ºC, respectively, and the die head temperature was about 110 ºC. The extruded materials were cut with a die face cutter as they left the extrusion die. After stable conditions were established, about 700 g of extruded product was collected and dried in an air oven at 40 ºC for 24 h. Extruded material was stored at 4 ºC in plastic bags for further analysis.
Analysis of Quality Parameters
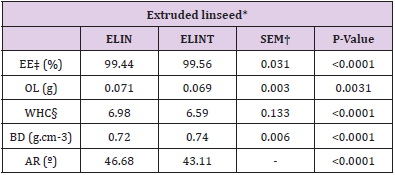
Extrusion effectiveness (EE) is the ability of the extruder to grind whole seeds, measured by comparing the number of whole grains in a sample before extrusion and the number of entire grains remaining after extrusion. In this study, EE was determined using the technique of Eggie (2010). Oil retention (OR) was measured using the method of Eggie (2010). Water holding capacity (WHC) was determined using a modified version of the AACC 56-30.01 method (AACC International, 1999) as amended by Eggie (2010). Bulk density (BD) was determined using a modified version of ASAE Standard S269.4 DEC 01(ASAE, 2007), as amended by Eggie (2010). The angle of repose (AR) was measured using the established method for the Carr Angle of Repose (Carr, 1965) and described by ASTM D6393-99 (ASTM, 2006). According to Carr (1965), low angles of 30° to 40° indicate a material with relatively easy-flowing characteristics, while high angles of 50° to 60° represent severe flow conditions [25].
Phenolic Content of Pistachio By-Product
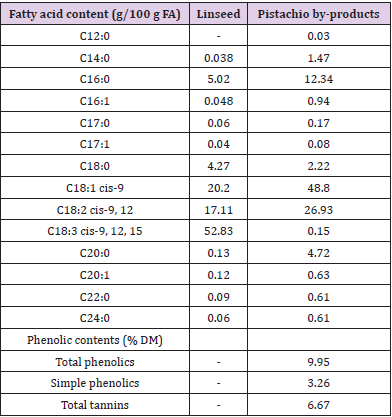
For tannin assay, PBP was dried in an oven at 40 ºC to constant weight to minimize changes in tannin content and activity [26]. Dried samples were ground to pass a 2 mm and then 0.5 mm sieve. Samples (200 mg) were extracted in four replicates in 70%, (v/v) aqueous acetone overnight at 4 °C. The extracts were centrifuged at 3000 g at 4 °C for 15 min, and the supernatant was obtained and used in the subsequent assay. The concentration of total phenolic (TP) compounds was determined using the Folin-Ciocalteu procedure [27] and the regression equation of tannic acid (Merck GmbH, Darmstadt, Germany) standard. Total tannins (TT) were estimated indirectly after being absorbed into insoluble polyvinylpolypyrrolidone (PVPP). The concentration of TT was calculated by subtracting the TP remaining after the PVPP treatment in the assay mixture (Table 1).
Table 1: Fatty acid composition of linseed and pistachio byproducts and phenolic contents of pistachio by-product.
Lipid Peroxidation and Fatty Acid Profile
Extruded materials were stored in plastic bags and kept in ambient temperature for 90 days. The stored samples were analyzed for peroxide value (PV) and FA profile on days 0, 10, 20, 30, 60, and 90 after extrusion. Each time, oil was extracted from samples. Also, the FA composition of PBP we analyzed. For oil extraction, dried samples were ground to powder in a grinder (Model 1093; sieve size, 1mm). The powders were extracted with n-hexane (1:4 wt/vol) by agitation in a dark place at ambient temperature for 48 h. The solvent was evaporated in vacuo at 40 °C to dryness. Peroxide value was assessed by colorimetric determination of iron-thiocyanate according to Shantha and Decker. The fatty acid profile was determined by gas chromatography. Fatty acid methyl esters (FAME) were prepared, according to Wijngaarden. A fused silica capillary column (WCOT Fused Silica Capillary, DANI, Model 1000, Rome, Italy) 120 m in length, 0.32 mm internal diameter, and 0.2 μM film thicknesses on an HP 6890 GC equipped with flame ionization detector was used to quantify FAMEs. The initial column temperature was set at 180 °C for 20 min, which was increased to 225 °C by increments of 5 °C/min, then to 250 °C by 10 °C/min and held for 12 min. Hydrogen was used as carrier gas with a flow of 1.7 ml/min for the first 10 min. Then, the flow was decreased to 1.3 ml/min, and maintained until the end of the analysis. The detector temperature was set at 300 °C. Identification of FA was performed by comparison with the retention times of FAMEs standards (Sigma-Aldrich, Catalog #18919). Separations of all FAME were obtained with a single chromatographic run.
α-Linolenic Acid Retention
The retention of ALA in extruded linseed product was calculated according to expression is given below (Imran et al., 2013): α-linolenic acid retention, % = (the content of ALA after extrusion ÷ the content of ALA before extrusion) × 100
Statistical Analysis
Data on quality parameters, PV, and FA changes were analyzed in a completely randomized design, with two treatments and four replicates using the general linear model procedure of SAS (2003). The statistical model was:
Where yijk is the observation, μ is the overall mean; Ti is the effect of treatments, and; eij is the residual error.
Data of PV and FA changes of treatments during the storage period were analyzed as a repeated measures approach using ANOVA with mixed linear models of SAS (2003). The Duncan’s t-test compared the means at a 0.05 probability level.
Results and Discussion
There were no differences between treatments for quality parameters (Table 2), and all factors were the same in all treatments (P<0.05). Peroxide value evaluation of treatments after the extrusion process (Table 3) demonstrated that ELINT treatment had lower PV than the ELIN group (P<0.05). A PV of less than five cannot be rancid for cattle and probably handled fat should not exceed a PV of 10 [28,29] showed that pistachio hulls, which at present are often considered as agricultural waste, contain an antioxidant that may usefully be extracted and added to foods. However, phenolic contents of PBP in a 10% extruded linseed mixture (by mass) could not be useful as a natural antioxidant for the stability of linseed during an extrusion process. [30] confirmed that the high-temperature extrusion process does not deactivate phenolic antioxidant compounds. So, disability of this content of phenolic prepared from PBP as a natural antioxidant can be for two possible reasons;
Table 2: Quality parameters of extruded linseed with or without antioxidants.
*ELIN, extruded linseed without antioxidant; ELINT, extruded linseed with TBHQ antioxidant.
†SEM, standard error of the mean;
‡E, Extrusion effectiveness; OL, Oil lost; WHC, Water holding capacity; BD, Bulk density and AR, Angle of repose.
§Measured in g H2O per gram dry matter.
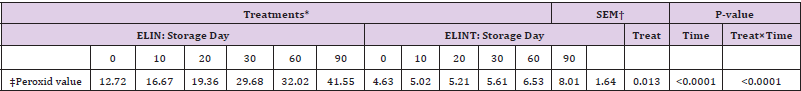
Table 3: Peroxide value of extruded linseed with or without antioxidant after the extrusion process.
*ELIN, extruded linseed without antioxidant; ELINT, extruded linseed with TBHQ antioxidant.
†SEM, standard error of the mean;
a, b: Within a row, means value with typical letter are not different (P<0.05).
a) the quantity of phenolic which prepared was low to act as an antioxidant in the extrusion process
b) the phenolic compounds of PBP did not release during extrusion process, so could not affect as antioxidant. Because most study which confirmed the antioxidant activity of plant phenolic, extracted the phenolic contents from unusual plants and used them as an aqueous solution containing phenolic compounds, but in our research, we used PBP as a feedstuff include phenolic.
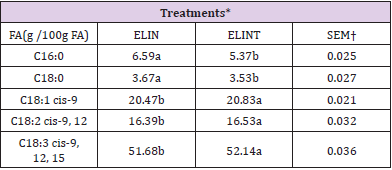
Camire and Dougherty (1998) demonstrated that the addition of phenolic compounds increased lipid stability in extruded corn. Moreover, [31] indicated that simple phenolic compounds could act as antioxidants for extruded oat cereals. The fatty acid composition of extruded linseed (Table 4) differed in treatments with or without TBHQ (P<0.05). The content of C16:0 and C18:0 for ELIN treatment were higher than for ELINT treatment (P<0.05), but ELINT treatment had higher content of C18:1-c9, C18:2-c9, c12 and C18:3-c9, c12, c18 than the ELIN group (P<0.05). (Table 5) It suggests that oxidation of the unsaturated FAs during the extrusion process in ELIN was higher than ELINT. Eggie (2010) used a synthetic antioxidant for extrusion process of linseed mixture with different absorbents (alfalfa, soy hulls, and corn gluten). It demonstrated that there was no significant change in the proportion of individual FA content in any products of extruded linseed, between before and after extrusion analysis. Peroxide value measurement of different treatments during the storage period indicated (Table 6) that treatment without TBHQ had higher PV than ELINT treatment (P<0.05). [32-35] Also, the time of storage had a significant effect on PV (P<0.05), and there was a significant interaction between treatment and time of storage for PV (P<0.05). As storage increased, the level of PV increased, and this increase was higher for ELIN treatment than for ELINT treatment. Also, the FA composition of extruded linseed between treatments (Table 7) during the storage period was different (P<0.05). Also, there was a significant effect for time of storage on the FA proportion (P<0.05). Furthermore, the interaction between treatment and time of storage on the FA proportion was significant (P<0.05). As storage increased, the percentage of unsaturated FA decreased (P<0.05), and this reducing was higher in ELIN treatment compared to ELINT (P<0.05).
Table 4: Fatty acid composition of extruded linseed with or without antioxidant after the extrusion process.
*ELIN, extruded linseed without antioxidant; ELINT, extruded linseed with TBHQ antioxidant.
†SEM, standard error of the mean;
a, b: Within a row, means value with typical letter are not different (P<0.05).
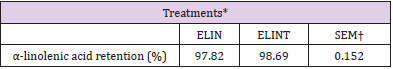
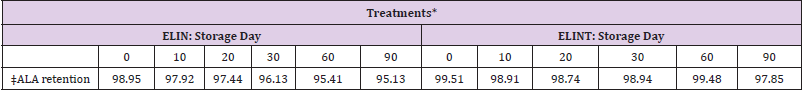
Table 5: α-linolenic acid retention of extruded linseed with or without antioxidant after the extrusion process.
*ELIN, extruded linseed without antioxidant; ELINT, extruded linseed with TBHQ antioxidant.
†SEM, standard error of the mean;
Table 6: Peroxide value of extruded linseed with or without antioxidant during the storage period.
*ELIN, extruded linseed without antioxidant; ELINT, extruded linseed with TBHQ antioxidant. †SEM, standard error of the mean.
‡meq O2/kg oil.
Table 7: Fatty acid composition of extruded linseed with or without antioxidant during the storage period.
*ELIN, extruded linseed without antioxidant; ELINT, extruded linseed with TBHQ antioxidant.
†SEM, standard error of the mean.
The α-linolenic acid is associated with vegetable oil instability [36]. Eggie (2010) reported that the proportions of FAs of extruded linseed with different absorbents (alfalfa, soy hulls, and corn gluten) were affected by the storage period, and it is logical that the concentration of FAs is affected. It is essential to realize that antioxidants are not capable of preventing oxidation, just delaying it [37-40]. (Table 8) demonstrated that milled linseed was stored up to 4 months at ambient temperatures without noticeable changes in quality. Furthermore, [41-44] reported ground linseed was stable for 280 d when stored at room temperature and in a 12-h light/dark cycle. It seems that extrusion temperature is a more important factor for the stability of unsaturated fatty acids during the extrusion process [45-47]. indicated that extrusion of the semolina-linseed mixture in 45 ºC for use in human food was not affected on the stability of triacylglycerol and ALA in the final product.
Table 8: α-linolenic acid retention of extruded linseed with or without antioxidants during the storage period.
*ELIN, extruded linseed without antioxidant; ELINT, extruded linseed with TBHQ antioxidant.
†SEM, standard error of the mean.
‡α-linolenic acid retention (%).
Conclusion
Phenolic compounds of pistachio by-product in 10% extruded linseed were not useful as a natural antioxidant for the stability of extruded linseed during the extrusion process and storage period. The peroxide value of ELIN treatment was higher than the ELINT group after the extrusion process and during the storage period. During the storage period, as storage increased, the level of PV increased. However, α-Linolenic acid retention in extruded linseed was not different between treatments after the extrusion process. Still, it decreased as increasing of storage period, and this decrease in the ELIN treatment was higher than the ELINT group.
Acknowledgments
We gratefully acknowledge Mrs. M. Akbarpour for assisting in the laboratory analyses, Mr. G. A. GolMovahed for his skilled technical assistance in the extrusion process, and S. Skies for editing this article.
References
- (1999) AACC International 56-30.01 Water hydration capacity of protein materials. In: A. International (Ed.), Approved Methods of Analysis (11th Ed.). St. Paul, MN: AACC International.
- Akraim F, Nicot MC, Juaneda P, Enjalbert F (2007) Conjugated linolenic acid (CLnA), conjugated linoleic acid (CLA) and other biohydrogenation intermediates in plasma and milk fat of cows fed raw or extruded linseed. Animal 1(6): 835-843.
- ASAE (2007) ASAE S 269.3 DEC 1991 -Cubes, Pellets, and Crumbles-Definitions and Methods for Determining Density, Durability, and Moisture Content. ASABE Standards. St. Louis, MI: ASABE.
- ASTM (2006) ASTM-D6393: Standard Test Method for Bulk Solids Characterization by Carr Indices. West Conshohocken, PA, USA.
- Bagheripour E, Rouzbehan Y, Alipour D (2008) Effects of ensiling, air-drying and addition of polyethylene glycol on in vitro gas production of pistachio by-product. Animal Feed Science and Technology 146(3-4): 327-336.
- Behgar M, Valizadeh R, Mirzaee M, Naserian AA, Nasiri MR (2009) Correlation between the physical and chemical properties of some forages and non-forage fiber source. Journal of Animal Veterinary Advances 8: 2280-2285.
- Bohluli A, Naserian AA, Valizadeh R, Eftekarshahroodi F (2007) The chemical composition and in vitro digestibility of pistachio by-product. In: BSAS Proceedings 224.
- Camire ME, Dougherty MP (1998) Added phenolic compounds enhance lipid stability in extruded corn. Journal of Food Science 63: 516-518.
- Camire ME, Dougherty MP, Osborn H, Davis-Dentici K, Briggs J, et al. (1999) Simple phenolic compounds as antioxidants for extruded oat cereals. Abstract 50A-38. Institute of Food Technologists: Chicago, IL.
- Carr RL (1965) Evaluating flow properties of solids. Chem Engin. 72(1): 163-168.
- Chen ZY, Ratnayake WMN, Cunnane SC (1994) Oxidative stability of linseed lipids during baking. Journal of American Oil Chemistry Society 71(6): 629-632.
- Chilliard Y, Glasser F, Ferlay A, Bernard L, Rouel J, et al. (2007) Diet, rumen biohydrogenation and nutritional quality of cow and goat milk fat. European Journal of Lipid Science and Technology 109(8): 828-855.
- Chouinard PY, Lévesque J, Girard V, Brisson GJ (1997) Dietary soybeans extruded at different temperatures: Milk composition and in situ fatty acid reactions. Journal of Dairy Science 80(11): 2913-2924.
- Coppen PP (1994) The use of antioxidants. in: Rancidity in Foods. 3rd JC Allen and R. J. Hamilton, eds. Chapman and Hall. London, UK: 84-103.
- Eggie K (2010) Development of an extruded lin-based feed ingredient. M. Sc. thesis. McGill University, Montreal.
- Ferlay A, Doreau M, Martin C, Chilliard Y (2013) Effect of incremental amounts of extruded linseed on milk fatty acid composition in dairy cows receiving hay or corn silage. Journal of Dairy Science 96(10): 6577-6595.
- Folch J, Lees M, Sloane Stanley GH (1957) A simple method for the isolation and purification of total lipids from animal tissues. Journal of Biology Chemistry 226(1): 497-509.
- Fontagné S, Lataillade E, Brèque J, Kaushik S (2008) Lipid peroxidative stress and antioxidant defence status during ontogeny of rainbow trout (Oncorhynchus mykiss). British Journal of Nutrition 100: 102-111.
- Frank AM, Robert EL, Clifford AH (2002) Processing and cooking effects on lipid content and stability of α-linolenic acid in spaghetti containing ground linseed. Journal of Agricultural Food Chemistry 50(6): 1668-1671.
- Ganesan V, Rosentrater KA, Muthukumarappan K (2005) Flowability and Handling Characteristics of Bulk Solids and Powders: A Review. In: ASAE Proceedings.
- Goli AH, Barzegar M, Sahari MA (2005) Antioxidant activity and total phenolic compounds of pistachio (Pistachia vera) hull extracts. Food Chemistry 92(3): 521-525.
- Gonthier C, Mustafa AF, Berthiaume R, Petit HV, Martineau R, et al. (2004) Effects of feeding micronized and extruded linseed on ruminal fermentation and nutrient utilization by dairy cows. Journal of Dairy Science 87(6): 1854-1863.
- Gülçin İ, Elias R, Gepdireme, A, Boyer L, Köksal E, et al. (2007) A comparative study on the antioxidant activity of fringe tree (Chionanthus virginicus L.) extracts. African Journal of Biotechnology 6(4): 410-418.
- Hammond EG (1984) Stability of Soybean Oil to Oxidation. In Proceedings of the World Soybean Research Conference III; Shibles, R, Ed; Westview Press: Boulder CO 251-258.
- Hosseinzadeh H, Tabassi S AS, Moghadam NM, Rashedinia M, Mehri S, et al. (2012) Antioxidant activity of pistacia vera fruits, leaves and gum extracts. Iranian Journal of Pharmaceutical Research 11 (3): 879-887.
- Imran M, Anjum FM, Arshad MU (2013) Influence of extrusion processing on fatty acids retention in full-fat linseed (Linum usitatissimum L.) meal. Journal of Food Processing Technology 4 (9): 2-5.
- Juaneda P, Rocquelin G (1985) Rapid and convenient separation of phospholipids and nonphosphorus lipids from rathe artusing silica cartridges. Lipids 20: 40-41.
- Kennelly JJ (1996) The fatty acid composition of milk fat as influenced by feeding oilseeds. Animal Feed Science and Technology 60(3-4): 137-152.
- Makkar HPS (Ed.) (2000) Quantification of Tannins in Tree Foliage. A Laboratory Manual for the FAO/IAEA Coordinated Research Project on Use of Nuclear and Related Techniques to Develop Simple Tannin Assays for Predicting and Improving the Safety and Efficiency of Feeding Ruminants on Tanniniferous Tree Foliage. Joint FAO/IAEA, FAO/IAEA of Nuclear Techniques in Food and Agriculture. Animal Production and Health Sub-program. FAO/IAEA Working Document. IAEA, Vienna, Austria.
- Malcolmson LJ, Przybylski R, Daun JK (2000) Storage stability of milled linseed. Journal of American Oil Chemistry Society 77: 235-238.
- Mustafa AF, Chouinard PY, Christensen DA (2003) Effects of feeding micronized linseed on yield and composition of milk from Holstein cows. Journal of the Science of Food and Agriculture 83(9): 920-926.
- Mustafa A, Mckinnon JJ, Christensen DA, He T (2002) Effects of micronization of linseed on nutrient disappearance in the gastrointestinal tract of steers. Animal Feed Science and Technology 95(3-4): 123-132.
- Oniszczuk T, Oniszczuk A, Gondek E, Guz L, Puk K, et al. (2019) Active polyphenolic compounds, nutrient contents and antioxidant capacity of extruded fish feed containing purple coneflower (Echinacea purpurea (L.) Moench.). Saudi Journal of Biological Sciences. 26(1): 24-30.
- Palmquist DL, Jenkins TC (1980) Fat in Lactation Rations-Review. J Dairy Sci 63(1): 1-14.
- Rahimi A, Naserian AA, Saremi B, Shahdadi AR (2012) Effect of deactivation of tannins of pistachio hull by Polyethylene Glycol on gas production in vitro. In: BSAS Proceedings 224.
- Reddy V, Urooj A, Kumar A (2005) Evaluation of antioxidant activity of some plant extracts and their application in biscuits. Food Chemistry 90(1-2): 317-321.
- Riaz MN (2005) Extrusion processing of oilseed meals for food: In Shahidi. F. (editor), Bailey’s Industrial Oil and Fat Products. John Wiley & Sons, New York 533.
- Shahidi F, Wanasundra PK (1992) Phenolic antioxidants. Critical Rev. Food Science & Nutrition 32: 67-103.
- Shantha NC, Decker EA (1994) Rapid, sensitive, iron-based spectrophotometric methods for determination of peroxide values of food lipids. Journal of AOAC International 77(2): 421-424.
- Shakeri P, Riasi A, Alikhani M, Fazaeli H, Ghorbani GR (2012) Effects of feeding pistachio by-products silage on growth performance, serum metabolites and urine characteristics in Holstein male calves. Journal of Animal Physiology and Animal Nutrition 97(6): 1022-1029.
- Statistical Analysis System (SAS) (2003) User Guide: Statistics. Version 9.2. SAS institute Inc, Cary, NC, USA.
- Sterk A, Hovenier R, Vlaeminck B, van Vuuren AM, Hendriks WH, et al. (2010) Effects of chemically or technologically treated linseed products and docosahexaenoic acid addition to linseed oil on biohydrogenation of C18:3n-3 in vitro. Journal of Dairy Science 93(11): 5286-5299.
- Tomaino A, Martorana M, Arcoraci T, Monteleone D, Giovinazzo C, et al. (2010) Antioxidant activity and phenolic profile of pistachio (Pistacia vera L.,variety Bronte) seeds and skins. Biochemistry 92(9): 1115-1122.
- Vahmani P, Naserian AA, Valizadea R, Nasiri-Moghadam H (2006) Nutritive value of pistachio by-products and their effects on Holstein cows in mid lactation. Journal of Agricultural Science and Technology 20: 201-210.
- Wichi HP (1988) Enhanced tumor development by butylated hydroxyanisole (BHA) from the prospective of effect on fore stomach and oesophageal squamous epithelium. Food and Chemical Toxicology 26: 717-723.
- Wijngaarden D (1967) Modified rapid preparation of fatty acid esters from lipids for gas chromatographic analysis. Analytical Chemistry 39(7): 848-849.
- Zinn RA (1995) Fat quality and feeding value of fat for feedlot cattle.

 Research Article
Research Article