Opinion
In the last months all over the world we have been crying, talking, and writing a lot of viruses, given the pandemic that has hit our planet. It is not the first time that mankind has been overwhelmed not only by wars and natural disasters but also by as unexpected as trembling infectious events. On more than one occasion there have been many deaths ascribable to serious viral infections. Nowadays, the deadest virus of all can be Human Immunodeficiency Virus since Global Health Observatory (GHO) data indicated that more than 30 million of persons perished after it was identified in the early 1980s. In 1967 experts identified Marburg virus, its mortality in the first outbreak was 25%, but it was more than 80% in the 1998-2000 outbreak in the Democratic Republic of Congo, as well as in the 2005 outbreak in Angola [1]. Ebola infection caused by Ebola virus (EBOV) is another severe disease with a high case– fatality rate [2]. Rabies is a fatal viral infection of mammals and domestic dogs cause around 95% of all estimated yearly 59,000 rabies deaths among human [3].
Smallpox is caused by the variola virus (VARV) and although a successful vaccination program led to its eradication, about 400 million of fatalities have been estimated in the 20th century [4]. Also, Dengue, rotavirus, and coronaviruses as SARS-CoV, MERS-CoV daily infect and, in the worst cases, kill many people in different parts of the globe [5-7]. In this opinion letter we would like to highlight the dualism of the physiopathology of these simple but powerful infective agents. The notion of duality can be found in many areas but here we would like to highlight this concept in virology since viruses, like two sides of the same coin, can in particular cases exert an oncogenic or an oncolytic action (Figure 1).In several cases viruses, aside from causing infections immediately identified as such, in a much more sneaky way, can be the cause of the onset of a whole series of tumors [8] or, in a much more unexpected and surprising way, they can counteract the onset or spread of some forms of cancer [9,10].
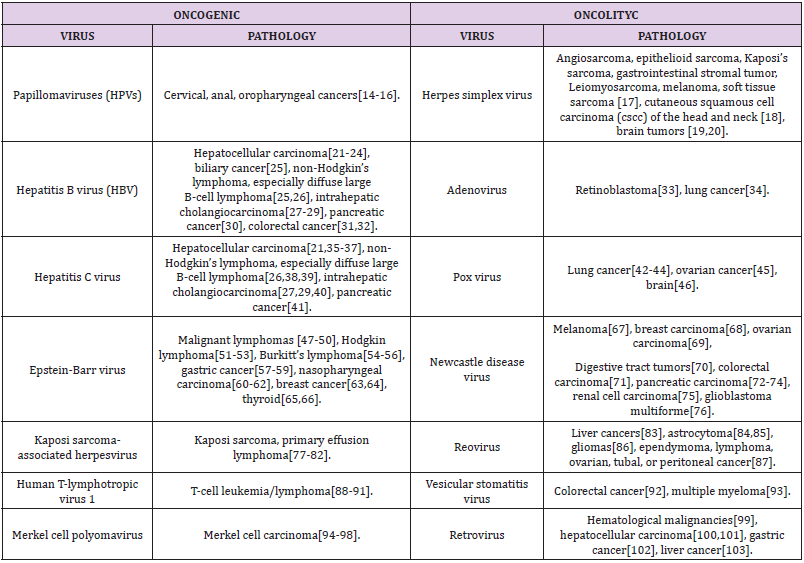
Viruses as human papillomaviruses (HPVs), Epstein–Barr virus (EBV), hepatitis B virus (HBV), hepatitis C virus (HCV), human T-lymphotropic virus 1 (HTLV-1), Kaposi sarcoma-associated herpesvirus (KSHV) and Merkel cell polyomavirus (MCPyV) can manipulate the host cellular signaling and transcriptional systems inducing several different cancer establishment and progression phenomena as summarized in the oncogenic virus section of Table 1.On the other hand, as listed in the oncolytic section of the Table1, oncolytic virotherapy (OVT), recently emerged as promising alternative solution in cancer treatment since, after contagion, oncolityc viruses, by interfering with the protein synthesis of tumor cells, can end cells by virus self-replication. Furthermore, oncolytic viruses can induce effective antitumor immunity reactions by engage and activate cytotoxic T-lymphocytes, mast and natural killer cells promoting the release of tumor antigens and cytokines [11-13]. To conclude, this opinion letter aims to highlight aspects that, although they are still under study, are already showing a great potential in the treatment of several bad prognosis oncological diseases. OVT approach requires more attention than that based on the use of chemotherapy and monoclonal antibodies because viruses remain pathogens that must be handled and engineered with great care. Furthermore, which is certainly no less important, while the side effects of chemotherapy can occur from their first administrations, a complex bioengineering process like the integration of a viral nucleic acid into a host cell, is never fully controllable on a short and long time scale.
Table 1: List of oncogenic or oncolytic viruses with related pathologies and bibliographic references.
References
- Brauburger K, Hume AJ, Mühlberger E, Olejnik J (2012) Forty-five years of Marburg virus research. Viruses 4: 1878-1927.
- Jacob ST, Crozier I, Fischer WA, Hewlett A, Kraft CS, et al. (2020) Ebola virus disease. Nature Reviews Disease Primers 6: 13.
- Hikufe EH, Freuling CM, Athingo R, Shilongo A, Ndevaetela EE, et al. (2019) Ecology and epidemiology of rabies in humans, domestic animals and wildlife in Namibia, 2011-2017. PLoS Negl Trop Dis 13: e0007355.
- Meyer H, Ehmann R, Smith GL (2020) Smallpox in the Post-Eradication Era. Viruses 12.
- Crawford SE, Ramani S, Tate JE, Parashar UD, Svensson L, et al. (2017) Rotavirus infection. Nat Rev Dis Primers 3: 17083.
- Rodriguez Roche R Gould EA (2013) Understanding the Dengue Viruses and Progress towards Their Control. BioMed Research International 690835.
- De Wit E, van Doremalen N, Falzarano D, Munster VJ (2016) SARS and MERS: recent insights into emerging coronaviruses. Nature Reviews Microbiology 14: 523-534.
- Schiller JT, Lowy DR (2014) Virus infection and human cancer: an overview. Recent Results Cancer Res 193: 1-10.
- Watanabe D, Goshima F (2018) Oncolytic Virotherapy by HSV. Adv Exp Med Biol 1045: 63-84.
- Zou Y, Luo Y, Zhang J, Xia N, Tan G, Huang C (2019) Bibliometric analysis of oncolytic virus research, 2000 to 2018. Medicine (Baltimore) 98: e16817.
- Howells A, Marelli G, Lemoine NR, Wang Y (2017) Oncolytic Viruses-Interaction of Virus and Tumor Cells in the Battle to Eliminate Cancer. Front Oncol 7: 195.
- Hemminki O, dos Santos JM, Hemminki A (2020) Oncolytic viruses for cancer immunotherapy. Journal of Hematology & Oncology 13: 84.
- Russell L, Peng KW, Russell SJ, Diaz RM (2019) Oncolytic Viruses: Priming Time for Cancer Immunotherapy. BioDrugs 33: 485-501.
- Garbuglia AR, Lapa D, Sias C, Capobianchi MR, Del Porto P (2020) The Use of Both Therapeutic and Prophylactic Vaccines in the Therapy of Papillomavirus Disease. Front Immunol 11: 188.
- James CD, Das D, Bristol ML, Morgan IM (2020) Activating the DNA Damage Response and Suppressing Innate Immunity: Human Papillomaviruses Walk the Line. Pathogens 9.
- de la Cour CD SC, Belmonte F, Syrjänen S, Verdoodt F, Kjaer SK (2020) Prevalence of human papillomavirus in oral epithelial dysplasia: Systematic review and meta-analysis. Head & Neck.
- Chen CY, Wang PY, Hutzen B, Sprague L, Swain HM, et al. (2017) Cooperation of Oncolytic Herpes Virotherapy and PD-1 Blockade in Murine Rhabdomyosarcoma Models. Scientific reports 7: 2396-2396.
- Varra V, Smile TD, Geiger JL, Koyfman SA (2020) Recent and Emerging Therapies for Cutaneous Squamous Cell Carcinomas of the Head and Neck. Curr Treat Options Oncol 21: 37.
- Totsch SK, Schlappi C, Kang KD, Ishizuka AS, Lynn GM, et al. (2019) Oncolytic herpes simplex virus immunotherapy for brain tumors: current pitfalls and emerging strategies to overcome therapeutic resistance. Oncogene 38: 6159-6171.
- Hua L, Wakimoto H (2019) Oncolytic herpes simplex virus therapy for malignant glioma: current approaches to successful clinical application. Expert Opin Biol Ther 19: 845-854.
- Perz JF, Armstrong GL, Farrington LA, Hutin YJ, Bell BP (2006) The contributions of hepatitis B virus and hepatitis C virus infections to cirrhosis and primary liver cancer worldwide. J Hepatol 45: 529-538.
- Petruzziello A (2018) Epidemiology of Hepatitis B Virus (HBV) and Hepatitis C Virus (HCV) Related Hepatocellular Carcinoma. Open Virol J 12: 26-32.
- Xie Y (2017) Hepatitis B Virus-Associated Hepatocellular Carcinoma. In: Infectious Agents Associated Cancers: Epidemiology and Molecular Biology, Cai Q, Yuan Z, Lan K (Eds.), Springer Singapore: Singapore, pp. 11-21.
- Levrero M, Zucman Rossi J (2016) Mechanisms of HBV-induced hepatocellular carcinoma. Journal of Hepatology 64: S84-S101.
- An J, Kim JW, Shim JH, Han S, Yu CS, et al. (2018) Chronic hepatitis B infection and non-hepatocellular cancers: A hospital registry-based, case-control study. PloS one 13: e0193232.
- Taborelli M, Polesel J, Montella M, Libra M, Tedeschi R, et al. Hepatitis B and C viruses and risk of non-Hodgkin lymphoma: a case-control study in Italy. Infect Agent Cancer 11: 27.
- Seo JW, Kwan BS, Cheon YK, Lee TY Shim CS, et al. Prognostic impact of hepatitis B or C on intrahepatic cholangiocarcinoma. Korean J Intern Med 35: 566-573.
- Jeong S, Tong Y, Sha M, Gu J, Xia Q (2017) Hepatitis B virus-associated intrahepatic cholangiocarcinoma: a malignancy of distinctive characteristics between hepatocellular carcinoma and intrahepatic cholangiocarcinoma. Oncotarget 8: 17292-17300.
- Wang Z, Sheng YY, Dong QZ, Qin LX (2016) Hepatitis B virus and hepatitis C virus play different prognostic roles in intrahepatic cholangiocarcinoma: A meta-analysis. World J Gastroenterol 22: 3038-3051.
- Desai R, Patel U, Sharma S, Singh S, Doshi S, et al. (2018) Association Between Hepatitis B Infection and Pancreatic Cancer: A Population-Based Analysis in the United States. Pancreas 47: 849-855.
- Su FH, Le TN, Muo CH, Te SA, Sung FC, et al. Chronic Hepatitis B Virus Infection Associated with Increased Colorectal Cancer Risk in Taiwanese Population. Viruses 12.
- Jung YS, Kim NH, Park JH, Park DI, Sohn CI (2019) Correlation between Hepatitis B Virus Infection and Colorectal Neoplasia. J Clin Med 8: 2085.
- Pascual Pasto G, Bazan Peregrino M, Olaciregui NG, Restrepo Perdomo CA, Mato Berciano A, et al. (2019) Therapeutic targeting of the RB1 pathway in retinoblastoma with the oncolytic adenovirus VCN-01. Science translational medicine 11.
- Lei J, Li QH, Yang JL, Liu F, Wang L, et al. (2015) The antitumor effects of oncolytic adenovirus H101 against lung cancer. Int J Oncol 47: 555-562.
- Axley P, Ahmed Z, Ravi S, Singal AK (2018) Hepatitis C Virus and Hepatocellular Carcinoma: A Narrative Review. J Clin Transl Hepatol 6(1): 79-84.
- Baumert TF, Jühling F, Ono A, Hoshida Y (2017) Hepatitis C-related hepatocellular carcinoma in the era of new generation antivirals. BMC Med 15: 52.
- Ninio L, Nissani A, Meirson T, Domovitz T, Genna A, et al. (2019) Hepatitis C Virus Enhances the Invasiveness of Hepatocellular Carcinoma via EGFR-Mediated Invadopodia Formation and Activation. Cells 8(11): 1395.
- Khaled H, Abu Taleb F, Haggag R (2017) Hepatitis C virus and non-Hodgkin's lymphomas: A minireview. J Adv Res 8(2): 131-137.
- Pozzato G, Mazzaro C, Dal Maso L, Mauro E, Zorat F, et al. (2016) Hepatitis C virus and non-Hodgkin's lymphomas: Meta-analysis of epidemiology data and therapy options. World J Hepatol 8: 107-116.
- Navas MC, Glaser S, Dhruv H, Celinski S, Alpini G, et al. (2019) Hepatitis C Virus Infection and Cholangiocarcinoma: An Insight into Epidemiologic Evidence and Hypothetical Mechanisms of Oncogenesis. Am J Pathol 189(6): 1122-1132.
- Arafa A, Eshak ES, Abdel Rahman TA, Anwar MM (2020) Hepatitis C virus infection and risk of pancreatic cancer: A meta-analysis. Cancer Epidemiol 65: 101691.
- Sun A, Durocher Allen LD, Ellis PM, Ung YC, Goffin JR, et al. (2019) Initial management of small-cell lung cancer (limited- and extensive-stage) and the role of thoracic radiotherapy and first-line chemotherapy: A systematic review. Curr Oncol 26: e372-e384.
- Kellish P, Shabashvili D, Rahman MM, Nawab A, Guijarro MV, et al. (2019) Oncolytic virotherapy for small-cell lung cancer induces immune infiltration and prolongs survival. J Clin Invest 129: 2279-2292.
- Rahman MM, McFadden G (2020) Oncolytic Virotherapy with Myxoma Virus. J Clin Med 9(1): 171.
- Nounamo B, Liem J, Cannon M, Liu J (2017) Myxoma Virus Optimizes Cisplatin for the Treatment of Ovarian Cancer In Vitro and in a Syngeneic Murine Dissemination Model. Mol Ther Oncolytics 6: 90-99.
- Pisklakova A, McKenzie B, Zemp F, Lun X, Kenchappa RS, et al. (2016) M011L-deficient oncolytic myxoma virus induces apoptosis in brain tumor-initiating cells and enhances survival in a novel immunocompetent mouse model of glioblastoma. Neuro Oncol 18: 1088-1098.
- Crombie JL, LaCasce AS (2019) Epstein Barr Virus Associated B-Cell Lymphomas and Iatrogenic Lymphoproliferative Disorders. Front Oncol 9: 109.
- Peng RJ, Han BW, Cai QQ, Zuo XY, Xia T, et al. (2019) Genomic and transcriptomic landscapes of Epstein-Barr virus in extranodal natural killer T-cell lymphoma. Leukemia 33: 1451-1462.
- Anastasiadou E, Stroopinsky D, Alimperti S, Jiao AL, Pyzer AR, et al. (2019) Epstein−Barr virus-encoded EBNA2 alters immune checkpoint PD-L1 expression by downregulating miR-34a in B-cell lymphomas. Leukemia 33: 132-147.
- Shannon Lowe C, Rickinson AB, Bell AI (2017) Epstein-Barr virus-associated lymphomas. Philos Trans R Soc Lond B Biol Sci 372(1732): 20160271.
- Murray PG, Young LS (2019) An etiological role for the Epstein-Barr virus in the pathogenesis of classical Hodgkin lymphoma. Blood 134(7): 591-596.
- Vrzalikova K, Sunmonu T, Reynolds G, Murray P (2018) Contribution of Epstein⁻Barr Virus Latent Proteins to the Pathogenesis of Classical Hodgkin Lymphoma. Pathogens 7(3): 59.
- Carbone A, Gloghini A (2018) Epstein Barr Virus-Associated Hodgkin Lymphoma. Cancers (Basel) 10(6): 163.
- Oduor CI, Movassagh M, Kaymaz Y, Chelimo K, Otieno J, et al. (2017) Human and Epstein-Barr Virus miRNA Profiling as Predictive Biomarkers for Endemic Burkitt Lymphoma. Front Microbiol 8: 501.
- Hernandez Vargas H, Gruffat H, Cros MP, Diederichs A, Sirand C, et al. (2017) Viral driven epigenetic events alter the expression of cancer-related genes in Epstein-Barr-virus naturally infected Burkitt lymphoma cell lines. Scientific reports 7: 5852.
- Rowe M, Fitzsimmons L, Bell AI (2014) Epstein-Barr virus and Burkitt lymphoma. Chin J Cancer 33(12): 609-619.
- Naseem M, Barzi A, Brezden Masley C, Puccini A, Berger MD, et al. (2018) Outlooks on Epstein-Barr virus associated gastric cancer. Cancer Treat Rev 66: 15-22.
- Nishikawa J, Iizasa H, Yoshiyama H, Shimokuri K, Kobayashi Y, et al. (2018) Clinical Importance of Epstein⁻Barr Virus-Associated Gastric Cancer. Cancers (Basel) 10(6): 167.
- Saito R, Abe H, Kunita A, Yamashita H, Seto Y, et al. (2017) Overexpression and gene amplification of PD-L1 in cancer cells and PD-L1(+) immune cells in Epstein-Barr virus-associated gastric cancer: The prognostic implications. Mod Pathol 30(3): 427-439.
- Xu M, Yao Y, Chen H, Zhang S, Cao SM, et al. (2019) Genome sequencing analysis identifies Epstein-Barr virus subtypes associated with high risk of nasopharyngeal carcinoma. Nat Genet 51: 1131-1136.
- Chan KCA, Woo JKS, King A, Zee BCY, Lam WKJ, et al. (2017) Analysis of Plasma Epstein-Barr Virus DNA to Screen for Nasopharyngeal Cancer. N Engl J Med 377: 513-522.
- Tsao SW, Tsang CM, Lo KW (2017) Epstein-Barr virus infection and nasopharyngeal carcinoma. Philos Trans R Soc Lond B Biol Sci 372 (1732): 20160270.
- Lawson JS, Salmons B, Glenn WK (2018) Oncogenic Viruses and Breast Cancer: Mouse Mammary Tumor Virus (MMTV), Bovine Leukemia Virus (BLV), Human Papilloma Virus (HPV), and Epstein-Barr Virus (EBV). Front Oncol 8: 1.
- Glaser SL, Canchola AJ, Keegan TH, Clarke CA, Longacre TA, et al. (2017) Variation in risk and outcomes of Epstein-Barr virus-associated breast cancer by epidemiologic characteristics and virus detection strategies: An exploratory study. Cancer Causes Control 28(4): 273-287.
- Moghoofei M, Mostafaei S, Nesaei A, Etemadi A, Sadri Nahand J, et al. (2019) Epstein–Barr virus and thyroid cancer: The role of viral expressed proteins. Journal of Cellular Physiology 234: 3790-3799.
- Almeida JFM, Proenca Modena JL, Bufalo NE, Peres KC, de Souza Teixeira E, et al. (2020) Epstein–Barr virus induces morphological and molecular changes in thyroid neoplastic cells. Endocrine.
- Schirrmacher V, van Gool S, Stuecker W (2019) Breaking Therapy Resistance: An Update on Oncolytic Newcastle Disease Virus for Improvements of Cancer Therapy. Biomedicines 7.
- Raihan J, Ahmad U, Yong YK, Eshak Z, Othman F, et al. (2019) Regression of solid breast tumours in mice by Newcastle disease virus is associated with production of apoptosis related-cytokines. BMC Cancer 19: 315.
- Matuszewska K, Santry LA, van Vloten JP, AuYeung AWK, Major PP, et al. (2019) Combining Vascular Normalization with an Oncolytic Virus Enhances Immunotherapy in a Preclinical Model of Advanced-Stage Ovarian Cancer. Clinical cancer research : an official journal of the American Association for Cancer Research 25: 1624-1638.
- Liang W, Wang H, Sun TM, Yao WQ, Chen LL, et al. (2003) Application of autologous tumor cell vaccine and NDV vaccine in treatment of tumors of digestive tract. World J Gastroenterol 9: 495-498.
- Schulze T, Kemmner W, Weitz J, Wernecke KD, Schirrmacher V, et al. (2009) Efficiency of adjuvant active specific immunization with Newcastle disease virus modified tumor cells in colorectal cancer patients following resection of liver metastases: results of a prospective randomized trial. Cancer Immunol Immunother58: 61-69.
- Buijs PR, van Eijck CH, Hofland LJ, Fouchier RA, van den Hoogen BG (2014) Different responses of human pancreatic adenocarcinoma cell lines to oncolytic Newcastle disease virus infection. Cancer Gene Ther 21: 24-30.
- Buijs P, van Nieuwkoop S, Vaes V, Fouchier R, van Eijck C, et al. (2015) Recombinant Immunomodulating Lentogenic or Mesogenic Oncolytic Newcastle Disease Virus for Treatment of Pancreatic Adenocarcinoma. Viruses 7: 2980-2998.
- Schwaiger T, Knittler MR, Grund C, Roemer Oberdoerfer A, Kapp JF, et al. (2017) Newcastle disease virus mediates pancreatic tumor rejection via NK cell activation and prevents cancer relapse by prompting adaptive immunity. International Journal of Cancer 141: 2505-2516.
- Ch'ng WC, Stanbridge EJ, Yusoff K, Shafee N (2013) The oncolytic activity of Newcastle disease virus in clear cell renal carcinoma cells in normoxic and hypoxic conditions: the interplay between von Hippel-Lindau and interferon-β signaling. J Interferon Cytokine Res 33: 346-354.
- Ali Saeed R, Alabsi AM, Ideris A, Omar AR, Yusoff K, et al. (2019) Evaluation of Ultra-Microscopic Changes and Proliferation of Apoptotic Glioblastoma Multiforme Cells Induced by Velogenic Strain of Newcastle Disease Virus AF2240. Asian Pac J Cancer Prev 20: 757-765.
- Li T, Ju E, Gao SJ (2019) Kaposi sarcoma–associated herpesvirus miRNAs suppress CASTOR1-mediated mTORC1 inhibition to promote tumorigenesis. J Clin Invest 129: 3310-3323.
- Chen J, Del Valle L, Lin HY, Plaisance Bonstaff K, Forrest JC, et al. (2019) Expression of PD-1 and PD-Ls in Kaposi's sarcoma and regulation by oncogenic herpesvirus lytic reactivation. Virology 536: 16-19.
- Hussein HAM, Okafor IB, Walker LR, Abdel Raouf UM, Akula SM (201) Cellular and viral oncogenes: the key to unlocking unknowns of Kaposi's sarcoma-associated herpesvirus pathogenesis. Arch Virol 163: 2633-2643.
- Goncalves PH, Ziegelbauer J, Uldrick TS, Yarchoan R (2017) Kaposi sarcoma herpesvirus-associated cancers and related diseases. Curr Opin HIV AIDS 12: 47-56.
- Wong JP Damania B (2017) Modulation of oncogenic signaling networks by Kaposi's sarcoma-associated herpesvirus. Biol Chem 398: 911-918.
- Dittmer DP, Damania B (2016) Kaposi sarcoma-associated herpesvirus: immunobiology oncogenesis and therapy. J Clin Invest 126: 3165-3175.
- Samson A, Bentham MJ, Scott K, Nuovo G, Bloy A, et al. (2018) Oncolytic reovirus as a combined antiviral and anti-tumour agent for the treatment of liver cancer. Gut 67: 562-573.
- Forsyth P, Roldán G, George D, Wallace C, Palmer CA, et al. (2008) A Phase I Trial of Intratumoral Administration of Reovirus in Patients With Histologically Confirmed Recurrent Malignant Gliomas. Molecular Therapy 16: 627-632.
- Samson A, Scott KJ, Taggart D, West EJ, Wilson E, et al. (2018) Intravenous delivery of oncolytic reovirus to brain tumor patients immunologically primes for subsequent checkpoint blockade. Science translational medicine 10.
- Kicielinski KP, Chiocca EA, Yu JS, Gill GM, Coffey M, et al. (2014) Phase 1 clinical trial of intratumoral reovirus infusion for the treatment of recurrent malignant gliomas in adults. Mol Ther 22: 1056-1062.
- Cohn DE, Sill MW, Walker JL, O Malley D, Nagel CI, et al. (2017) Randomized phase IIB evaluation of weekly paclitaxel versus weekly paclitaxel with oncolytic reovirus (Reolysin®) in recurrent ovarian tubal or peritoneal cancer: An NRG Oncology/Gynecologic Oncology Group study. Gynecol Oncol 146: 477-483.
- Adkins BD, Ramos JC, Bliss Moreau M, Gru AA (2020) Updates in lymph node and skin pathology of adult T-cell leukemia/lymphoma biomarkers and beyond. Seminars in Diagnostic Pathology 37: 1-10.
- Kagdi HH, Taylor GP (2018) Transcriptome and Somatic Mutation Associated with Non-Malignant Human T Lymphotropic Virus Type 1 Infection and Adult T-Cell Leukemia/Lymphoma. Blood 132: 2843-2843.
- James T, Fivenson D, Cotton J (2018) Human T-lymphotropic virus 1 associated with adult T-cell leukemia/lymphoma. Cutis 102: E4-e7.
- Esau D (2017) Viral Causes of Lymphoma: The History of Epstein-Barr Virus and Human T-Lymphotropic Virus 1. Virology (Auckl) 8: 1178122x17731772.
- Stewart JH, Ahmed M, Northrup SA, Willingham M, Lyles DS (2011) Vesicular stomatitis virus as a treatment for colorectal cancer. Cancer Gene Therapy 18: 837-849.
- Calton CM, Kelly KR, Anwer F, Carew JS, Nawrocki ST (2018) Oncolytic Viruses for Multiple Myeloma Therapy. Cancers (Basel) 10: 198.
- DeCaprio JA (2017) Merkel cell polyomavirus and Merkel cell carcinoma. Philos Trans R Soc Lond B Biol Sci 372: 20160276.
- Verhaegen ME, Mangelberger D, Harms PW, Eberl M, Wilbert DM, et al. (2017) Merkel Cell Polyomavirus Small T Antigen Initiates Merkel Cell Carcinoma-like Tumor Development in Mice. Cancer Res 77: 3151-3157.
- Liu W, MacDonald M, You J (2016) Merkel cell polyomavirus infection and Merkel cell carcinoma. Curr Opin Virol 20: 20-27.
- Starrett GJ, Marcelus C, Cantalupo PG, Katz JP, Cheng J, et al. (201) Merkel Cell Polyomavirus Exhibits Dominant Control of the Tumor Genome and Transcriptome in Virus-Associated Merkel Cell Carcinoma. mBio 8.
- Liu W, Yang R, Payne AS, Schowalter RM, Spurgeon ME, et al. (2016) Identifying the Target Cells and Mechanisms of Merkel Cell Polyomavirus Infection. Cell Host Microbe 19: 775-787.
- Schambach A, Morgan M (2016) Retroviral Vectors for Cancer Gene Therapy. Recent Results Cancer Res 209: 17-35.
- Lu YC, Chen YJ, Yu YR, Lai YH, Cheng JC, et al. (2012) Replicating retroviral vectors for oncolytic virotherapy of experimental hepatocellular carcinoma. Oncology reports 28: 21-26.
- Zhang YW, Niu J, Lu X, Yang YX , Zhao HW, et al. (2013) Multi-target lentivirus specific to hepatocellular carcinoma: in vitro and in vivo studies. J Hepatol 58: 502-508.
- Li R, Yang HQ, Xi HL, Feng S, Qin RH (2017) Inhibition of CDH17 gene expression via RNA interference reduces proliferation and apoptosis of human MKN28 gastric cancer cells. Int J Oncol 50: 15-22.
- Ranzani M, Cesana D, Bartholomae CC, Sanvito F, Pala M, et al. (2013) Lentiviral vector-based insertional mutagenesis identifies genes associated with liver cancer. Nat Methods 10: 155-161.

 Opinion
Opinion