Abstract
Objectives: To explore the character of neurogenic inflammation in the inflammation of oral lichenoid reactions (OLR) and compare the difference between oral lichen planus (OLP) and oral lichenoid mucositis (OLM).
Methods: The patient-based case control study applied twelve antibodies to check 74 biopsy specimens by the immunohistochemistry technique (IHC). Participants included 28 cases of oral lichen planus (OLP), 16 cases of contact stomatitis from dental restorative materials (OLM-dental) cases, 14 cases of mucosal reaction to systemic drug administration (OLM-drug), 15 cases of contact stomatitis from topical chemical exposure (OLM-contact) and one traumatic fibroma (TF) case. Twelve antibodies are anti-mast cell chymase (MCC), anti-calcitonin gene-related peptide (CGRP), antisubstance P (SP), anti-neuropeptide Y (NPY), anti-neurofilament heavy polypeptide (NF-H), anti-vasoactive intestinal peptide (VIP), anti-neurotrophin 3 (NT-3), antiinterferon gamma (INFG), anti-cytokine IL-22 (IL-22), anti-CD3 (CD3), anti-CD8 (CD8) and anti-CD4 (CD4).
Results: Neurogenic inflammation does involve in the inflammation of OLR. The most significant involvement exists in contact stomatitis from dental restorative materials (OLM-dental). In addition, Oral lichen planus (OLP) shows a partial loss of Neurofilament and Neuropeptide Y in the epithelial region. Mucosal reaction to systemic drug administration (OLM-drug) demonstrates a weaker expression of CD8 in epithelial region and INFG in the subepithelial region.
Keywords: Oral Lichenoid Reaction; Oral Lichen Planus; Oral Lichenoid Mucositis; Neurogenic Inflammation; T-Cell Mediated Hypersensitivity Type Iv; Immunohistochemistry Technique
Abbreviations: OLM: Oral Lichenoid Mucositis; OLR: Oral Lichenoid Reactions; OLP: Oral Lichen Planus; IHC: Immunohistochemistry Technique; TF: Traumatic Fibroma; MCC: Mast Cell Chymase; CGRP: Calcitonin Gene-Related Peptide; SP: Substance P; NPY: Neuropeptide Y; NF-H: Neurofilament Heavy Polypeptide; VIP: Vasoactive Intestinal Peptide; NT-3: Neurotrophin 3; INFG: Interferon Gamma (INFG); OBC: Oral Biopsy Service
Introduction
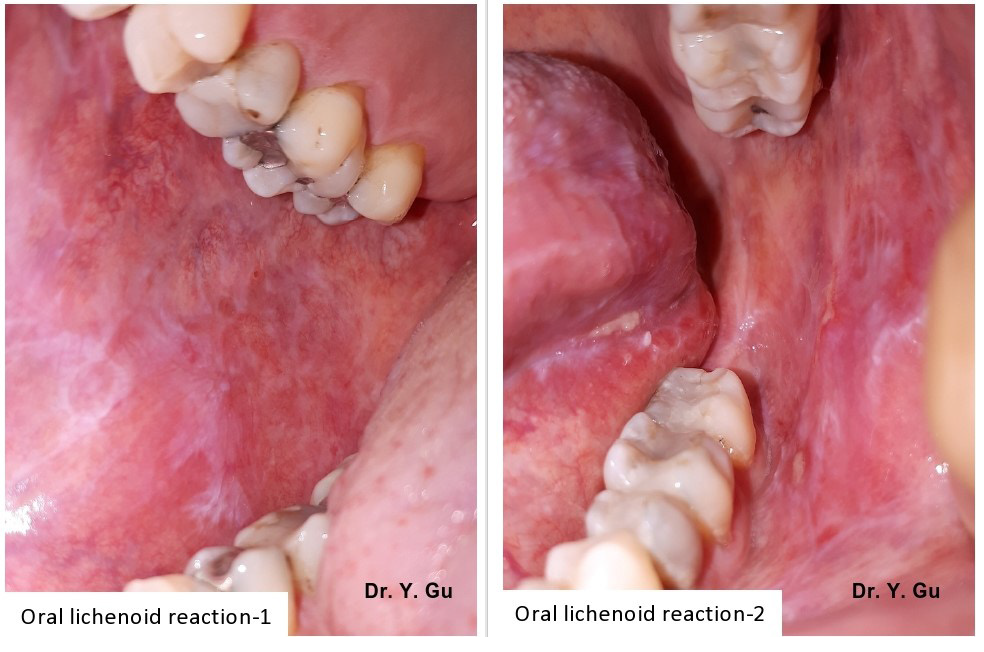
Oral lichenoid reaction (OLR) refers to oral lichen planus (OLP) or oral lichenoid mucositis (OLM). Oral lichenoid mucositis could be one of three conditions, which include lichenoid contact stomatitis from dental restorative materials (OLM-dental), mucosal reaction to systemic drug administration (OLM-drug), and contact stomatitis from topical chemical exposure (OLM-contact). These four oral mucosal conditions share a similar clinical appearance that is white reticular striations on erythematous mucosal base. An incisional biopsy is required to confirm a definitive diagnosis [1] Please see Figure 1. However, it is still a challenge for oral pathologists to differentiate four types of oral lichenoid reactions histopathologically. Oral lichen planus (OLP) shows a band-like lymphohistiocytic infiltration immediately demolishing the basal cell layer of keratinized stratified squamous epithelium. Oral lichenoid mucositis (OLM) displays the similar histopathological features of OLP, but may have other features as well, which include patched lymphohistocytic infiltration and /or other inflammatory cells, such as plasma cells, eosinophils, and mast cells. Nevertheless, there is no gold standard that can be followed [2] please see Figure 2. The pathogenesis of OLP and OLM is categorized in T-cell mediated hypersensitivity type IV. However, the difference in subtypes between OLP and OLM is not clear. Neurogenic inflammation is a pathological process of a mutual interaction between neuropeptides and inflammatory cells, while it is proved in the experimental study level [3,4]. The purpose of this case-control study by using human tissue specimens is to detect different pathways between OLP and OLM. It will benefit for the histopathological diagnosis.
Figure 1: The clinical appearance of oral lichenoid reaction is white reticular striations in the erythematous mucosal base with or without ulcerations.
Materials and Methods
The case-control study used twelve antibodies to check biopsy specimens of 28 OLP cases and 45 OLM cases by immunohistochemistry (IHC) technique. OLM cases included 16 OLM-dental, 14 OLM-drug and 15 OLM-contact specimens. In addition, a traumatic fibroma (TF) case, was arranged in the study as a non-specific inflammation control. Participants aged between 28- and 72-year-old (average 43) and with 82% gender ratio (Female by Male) had lesions on buccal mucosa (64%), gingivae (35 %) or tongue (1%). Twelve antibodies are anti-mast cell chymase (MCC), anti-calcitonin gene-related peptide (CGRP), anti-substance P (SP), anti-neuropeptide Y (NPY), anti-neurofilament heavy polypeptide (NF-H), anti-vasoactive intestinal peptide (VIP), anti-neurotrophin 3 (NT-3), anti-interferon gamma (INFG), anti-cytokine IL-22 (IL- 22), anti-CD3 (CD3), anti-CD8 (CD8) and anti-CD4 (CD4). We used a double-blind working protocol to manage the entire study. Specimens in four study groups were numbered alphabetically only by a histologist (AK) herself. The result was unveiled after the scoring and before the data analysis. For avoiding bias, we have positive controls, negative controls, a non-specific inflammation control, the crucial inclusion and exclusion standard in each step.
Inclusion, Exclusion and Preparation
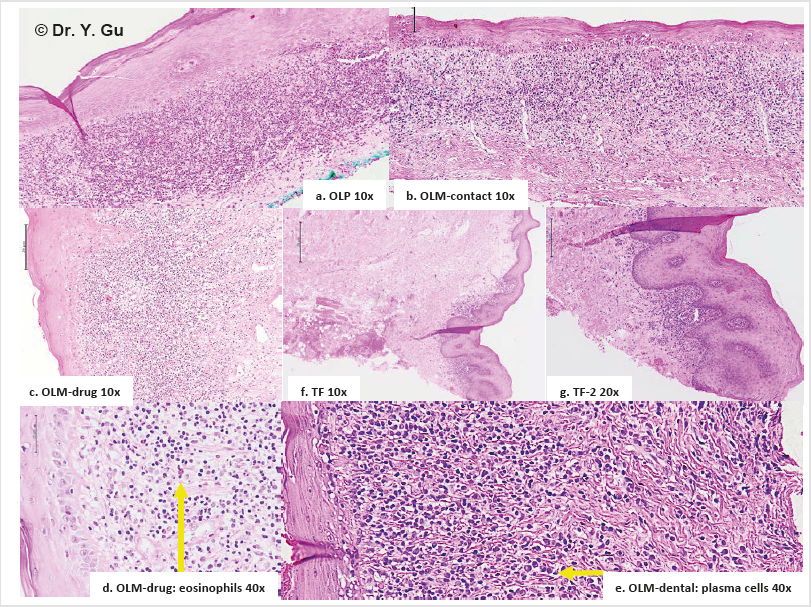
A medical laboratory technologist (AK) searched OLP and/ or OLM as key words in the system of Oral Biopsy Service (OBS) Laboratory of Faculty of Dentistry, Dalhousie University, Halifax, Canada. Totally 296 cases were found between January 1st 2011 and May 31, 2017. An oral pathologist (YG) read all H&E slides. Only 87 OLP and OLM specimens plus one traumatic fibroma specimen were selected. Exclusion is ulcerative type of OLR, bullous type of OLR, OLR combined with fungus infection, inappropriate biopsy specimens and inadequate tissue blocks. The inclusive standard of study groups obeys to relatively specific histopathological features about OLP and OLM described in 4th edition of Neville’s oral and maxillofacial pathology. OLP showed band-like lymphohistiocytic infiltration immediately demolishing the basal cell layer of keratinized stratified squamous epithelium. OLM-dental showed the similar feature as OLP, but has scattered plasma cells. OLM-drug showed the similar feature as OLP, but has scattered eosinophils. OLM-contact showed the similar feature as OLP but has patched distribution please see Figure 2.
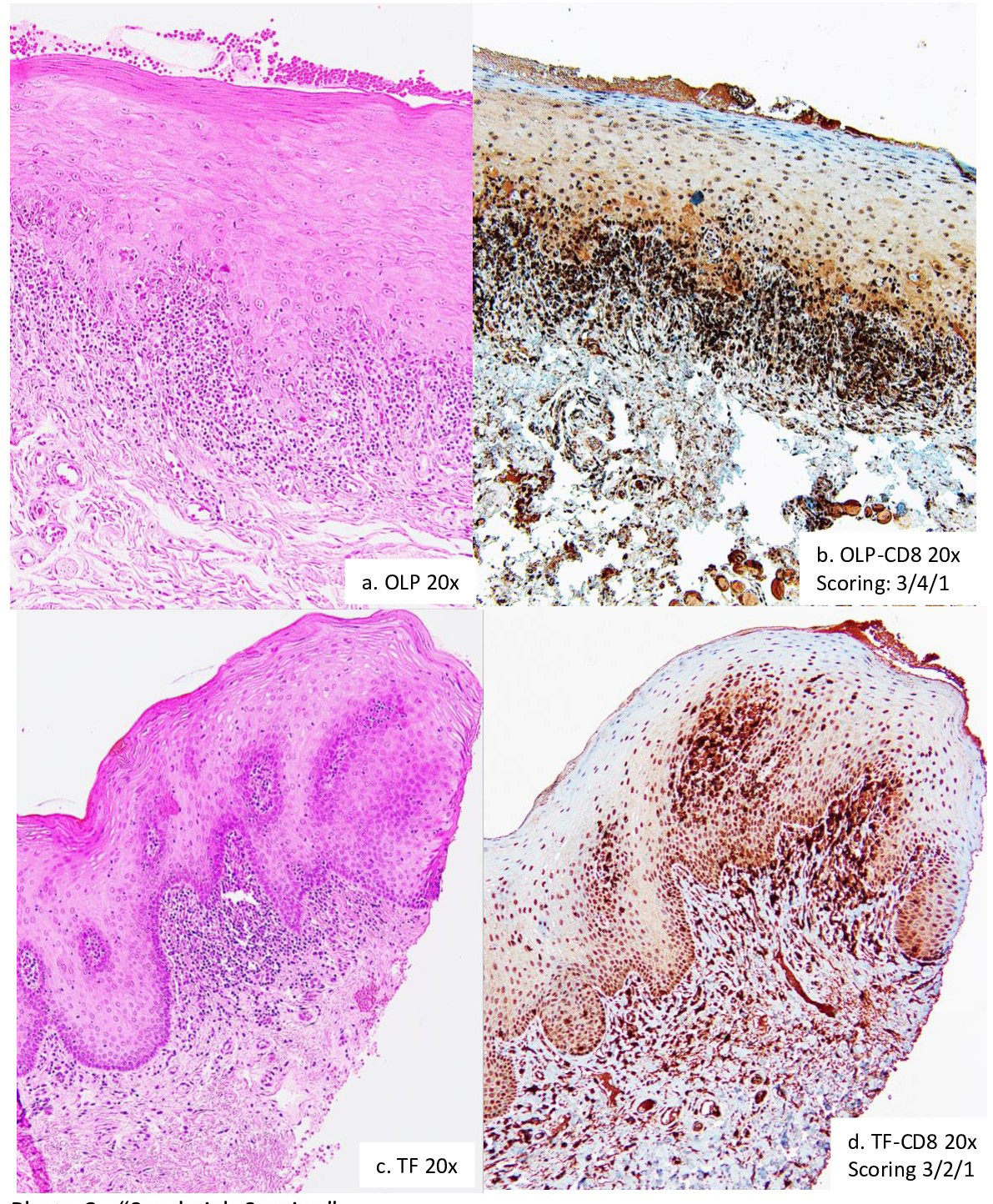
Figure 2: Histopathological features of oral lichenoid reactions. a. OLP (oral lichen planus): Band-like lymphohistiocytic infiltration immediately demolishing the basal cell layer of keratinized stratified squamous epithelium. b. TF (traumatic fibroma): Dense fibrous connective tissue mass covered by keratinized stratified squamous epithelium with focally mild chronic inflammation. c. c/d. OLM-drug (mucosal reaction to systemic drug administration): The similar feature as OLP, but has scattered eosinophils. d. e. OLM-dental (contact stomatitis from dental restorative materials): The similar feature as OLP, but has scattered plasma cells. e. f. OLM-contact (contact stomatitis from topical chemical exposure): The similar feature as OLP, but has patched distribution.
The medical laboratory technologist (AK) sectioned specimens (4um thickness, 3-5mm length and 2-3mm wideness) from paraffin tissue blocks, which were stored in the pathology archive of the OBS. She mounted 5 - 8 specimens on one glass slide. Therefore, 88 specimens were arranged in 14 slides. We named it “Minor Array Plate”. Totally 252 slides (14 x 18) were prepared for IHC study. The process was conducted in the OBS laboratory. A chief medical laboratory technologist (PC) conducted the IHC manual staining procedure (Capillary Gap Technology) in the IHC laboratory of Faculty of Medicine, Dalhousie University, Halifax, Canada. The oral pathologist (YG) read all IHC slides. Eventually 28 OLP and 45 OLM specimens plus one traumatic fibroma specimen were selected. Exclusion is uneven staining, unusual weak or absent staining, background or artifactual staining and inadequate amount of a specimen.
Antibodies and Optimal Dilution
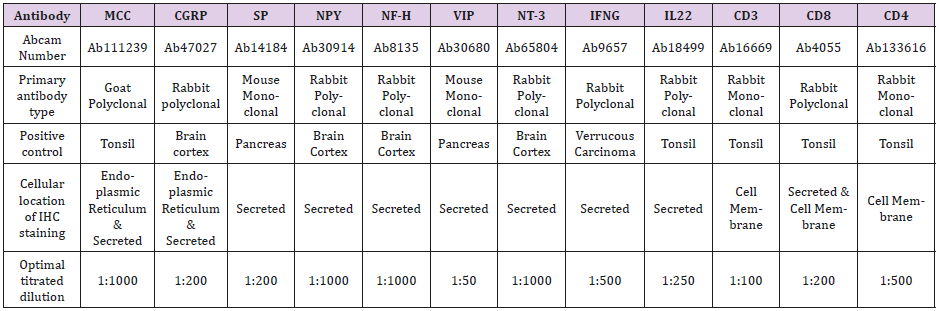
Twelve primary antibodies and the antibody dilution buffer were purchased from Abcam (Toronto, ON, Canada M5W 0E9). Second antibody (mouse IgG) and reagents were bought from Inter Medico (Markham, ON, Canada L3R 6E9). All antibodies are able of reacting to human tissue. We toned the optimal antibiotic dilution in positive control specimens that were recommended by Abcam and published reference. Negative controls were obtained by replacing the primary antibody with mouse IgG. Please see details in Table 1.
Note: All primary antibodies react to human tissue. Positive controls are recommended by Abcam and published reference. Type of retrieval solution is Decloaker Citrate buffer solution (PH6.0). Second antibody is mouse probe. Detection system is MACH4 polymer and HRP polymer with DAB chromogen, but Dako envision polymer is applied for anti-mast cell chymase. Antibody dilution buffer was purchased in Abcam. MCC: anti-mast cell chymase; CGRP: anti-calcitonin gene-related peptide; SP: anti-substance P; NPY: antineuropeptide Y; NF-H: anti-neurofilament heavy polypeptide; VIP: anti-vasoactive intestinal peptide; NT-3: anti-neurotrophin 3; INFG: anti-interferon gamma; IL-22: anti-cytokine IL-22; CD3: anti-CD3; CD8: anti-CD8; CD4: anti-CD4.
Immunohistochemistry Procedure
Antigen retrieval was processed in the Biocare Autoclave by PH 6.0 citrate buffer solution (DeCloaker) immediately after deparaffinization. The enzymatic digestion was conducted by Proteinase K. The procedure of protein block and endogenous enzyme block was achieved by Peroxidizer, Biocare’s background Sniper, automation buffers (Triton-X-100 and Tween 20). The primary and secondary antibody were washed by automation buffer (1% BAS and 0.025% Triton X-100) after enough incubation time respectively. The detection system was MACH 4 probe plus HRP-Polymer with DAB (diaminobenzidine) chromogen. The counterstain was Hematoxylin.
Quantification of the Data
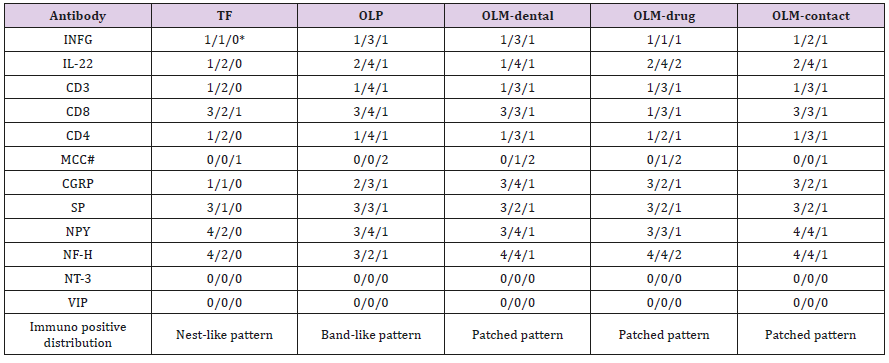
IHC slides were analyzed under an optical microscope (Olympus BX51 microscope; Olympus Optical Co., Tokyo, Japan) connected to a digital color camera/Q-Color 5 (Olympus). Images were obtained with 10x, 20x and 40x objectives UPLanFI (resolution: 2.75mm), at a size of 2560 x 1920 pixels (resolution: 1mm = 3000 pixels), under standard conditions. Pictures were taken from the whole slide to perform image analysis. The proportion of immune-positive cells is used to account scores if the staining located in the nucleus, on the cellular membrane or within the endoplasmic reticulum. The combinative semiquantitative scoring is applied for the secreted staining. The scoring outcome will be the combination of two types of accounting methods if antibodies have two staining locations. The scoring principle is score 1: < 10% and/or mild stain; score 2: 10-50% and/or mild-moderate stain; score 3: 50-90% and/or moderate-strong stain; score 4: >90% and/or strong stain. The scoring procedure was conducted by two researchers (EL and TB) and an oral pathologist (YG) respectively for 18 times. The scoring protocol is to score the staining outcome to epithelial region, subepithelial region and submucosal region separately. Each case was marked by three scores. We called it “Sandwich Scoring”. Please see details in Figure 3. The consistency of the immunopositive staining is different between study groups as well. Oral lichenoid mucositis (OLM) demonstrated a patched staining pattern. Oral lichen planus (OLP) addressed a band-like staining pattern. Traumatic fibroma showed a nest-like staining pattern. Those were recorded in the scoring data as well please see details in table 2.
Note 1: *Scoring location: epithelial region/subepithelial region/submucosal region Note 2: #MCC didn’t showed the difference between groups in the immunopositive distribution. Note 3: MCC: anti-mast cell chymase; CGRP: anti-calcitonin gene-related peptide; SP: anti-substance P; NPY: anti-neuropeptide Y; NF-H: anti-neurofilament heavy polypeptide; VIP: anti-vasoactive intestinal peptide; NT-3: anti-neurotrophin 3; INFG: antiinterferon gamma; IL-22: anti-cytokine IL-22; CD3: anti-CD3; CD8: anti-CD8; CD4: anti-CD4; TF: traumatic fibroma; OLP: oral lichen planus; OLM-dental: lichenoid contact stomatitis from dental restorative materials; OLM-drug: allergic mucosal reaction to systemic drug administration; OLM-contact: contact stomatitis from topical chemical exposure (OLM-contact).
Figure 3: “Sandwich Scoring”. I. Scoring principle: Score 1: < 10% and/or mild stain; Score 2: 10-50% and/or mild-moderate stain; Score 3: 50-90% and/ or moderate-strong stain; Score 4: >90% and/or strong stain. II. Scoring protocol: Epithelial region; Subepithelial region; Submucosal region. a. OLP (oral lichen planus). b. OLP CD8 IHC scoring: Epithelial region 3; Subepithelial region 4; Submucosal region 1. c. TF (traumatic fibroma). d. TF CD8 IHC scoring: Epithelial region 3; Subepithelial region 2; Submucosal region 1.
Analysis of the Data
The study size are 28 cases in OLP group, 16 cases in OLMdental group, 14 cases in OLM-drug group and 15 cases in OLMcontact group. The outcome of immunopositive scores in each group was obtained by the mean value of all cases. It is surprised that the standard deviation for the mean value in each group is one. It means specimens in each study group showed the same specific immunopositive pattern. Therefore, we did the comparison and contrast based on their patterns, rather than on their individual data. We call it “Pattern Comparison”. The comparing baseline is represented by the non-specific inflammation control. There is a remarkable difference if the mean score in a study group is higher than the score in the same region in the non-specific inflammation control please see details in table 2.
Result
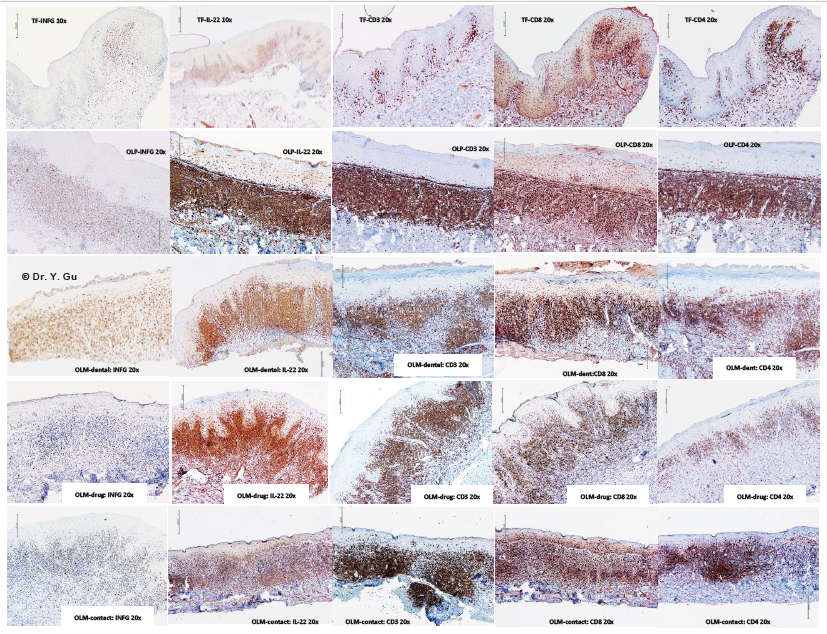
Figure 4: Immunohistochemistry (IHC) outcome of oral lichenoid reactions regarding to antibodies of INFG, IL-22, CD3, CD8 and CD4. Four study groups: oral lichen planus (OLP); mucosal reaction to systemic drug administration (OLM-drug); contact stomatitis from dental restorative materials (OLM-dental); contact stomatitis from topical chemical exposure (OLM-contact). Non-specific inflammation control: traumatic fibroma (TF). Five IHC antibodies: anti-interferon gamma (INFG), anti-cytokine IL-22 (IL-22), anti-CD3 (CD3), anti-CD8 (CD8), and anti-CD4 (CD4). 2. OLP and neurogenic inflammation -Yang Gu Figure 4 Figure 4. Immunohistochemistry (IHC) outcome of oral lichenoid reactions regarding to antibodies of INFG, IL-22, CD3, CD8 and CD4 Four study groups: oral lichen planus (OLP); mucosal reaction to systemic drug administration (OLM-drug); contact stomatitis from dent
Common molecular signals of T-cell mediated hypersensitivity type IV are CD3, CD8, CD4, IL-22 and Interferon gamma (INFG). We received all immunopositive outcomes in four study groups (Please see Table 2 and Figure 4). Traumatic fibroma (TF) was used as a baseline control for the comparison. There is a significantly stronger expression of all signals in the subepithelial region of four study groups, while OLP group is the strongest one. The significantly weaker expression of CD8 in the epithelial region and INFG plus CD4 in the subepithelial region was identified in the OLM-drug group. OLM-dental group presented with weaker expression of IL-22 in the epithelial region. The intensity of T-cell mediated hypersensitivity type IV is arranged in a descending order of OLP, OLM-contact, OLM-dental and OLM-drug. Available molecular signals of neurogenic inflammation are Mast cell chymase (MCC), Calcitonin gene-related peptide (CGRP), Substance P (SP), Neuropeptide Y (NPY), Neurofilament heavy polypeptide (NF-H), Vasoactive intestinal peptide (VIP) and Neurotrophin 3 (NT-3) [4]. We received five immunopositive outcomes in four study groups (Please see Table 2 and Figure 5). VIP and NT-3 showed negative results in all groups. Traumatic fibroma (TF) was used as a baseline control for the comparison. There is a significantly stronger expression of CGRP, SP, NPY and NF-H in the subepithelial region in four study groups, while the stronger expression of MCC in the subepithelial region was only found in OLM-dental and OLM-drug groups. The remarkable increase of CGRP in epithelial, subepithelial and submucosal regions was noticed in four study groups, but the strongest one is OLM-dental group. The significant decrease of NPY in epithelial region was identified in OLP, OLM-dental and OLMdrug groups. The only notable decrease of NF-H staining in the epithelial region was in the OLP group. The intensity of neurogenic inflammation is organized in a descending order of OLM-dental group, OLM-contact group, OLM-drug group and OLP group.
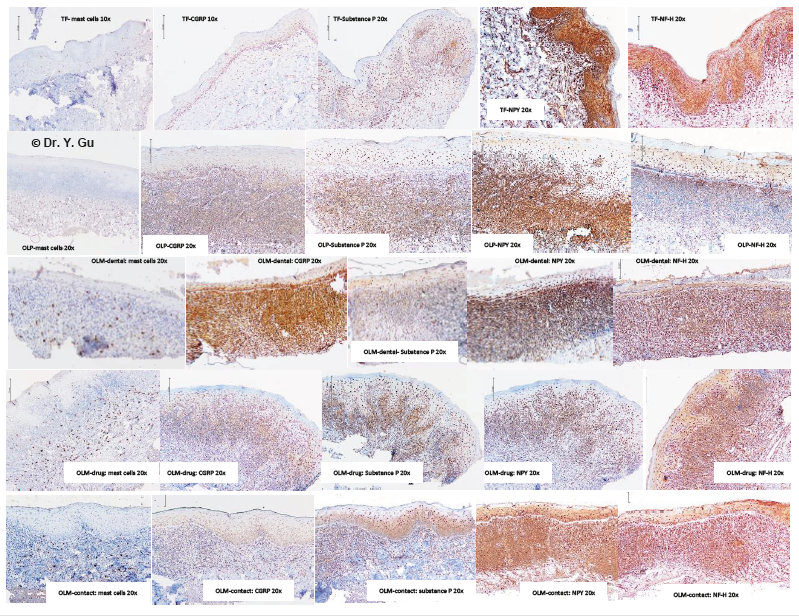
Figure 5: Immunohistochemistry (IHC) outcome of oral lichenoid reactions regarding to antibodies of MCC, CGRP, SP, NPY and NF-H. Four study groups: oral lichen planus (OLP); mucosal reaction to systemic drug administration (OLM-drug); contact stomatitis from dental restorative materials (OLM-dental); contact stomatitis from topical chemical exposure (OLM-contact). Non-specific inflammation control: traumatic fibroma (TF). Five IHC antibodies: anti-mast cell chymase (MCC), anti-calcitonin gene-related peptide (CGRP), anti-substance P (SP), anti-neuropeptide Y (NPY), anti-neurofilament heavy polypeptide (NF-H).
Discussion
Interferon gamma (INFG) is encoded by INFG gene and classified by ontology as cytokine activity [www.genecards.org]. INFG is produced by inflammatory cells and mucosal epithelium. There was no difference between traumatic fibroma and four study groups in the epithelial regions. In addition, IFNG promotes the Th1 differentiation first, and then Th1 produces INFG consequently [5]. The weakness of INFG expression in the subepithelial region of OLM-drug group implies Th1 less likely involves in the OLMdrug inflammation. CD8, a transmembrane glycoprotein binding to major histocompatibility complex (MHC) I, mainly expresses on cytotoxic T cells. CD4, a membrane glycoprotein binding to major histocompatibility complex (MHC) II, mainly expresses on T helper cells and macrophages. CD3 is a protein complex of T cell co-receptor. There is no significant difference between four study groups in CD3 staining. The significant weakness of CD8 expression in the epithelial region and CD4 expression in the subepithelial region was found in the of OLM-drug group. It may indicate CD8 T cells probably don’t play a dominant role in the OLM-drug inflammation. IL-22 is encoded by IL-22 gene and classified by ontology as cytokine activity [www.genecards.org]. Th17 cells and ILC3 mainly produce IL-22, while the IL-22 receptor expresses on epithelial and stromal cells. The receptor-ligand interaction leads to activation of the transcription factor STAT3 in the target cell [6]. There is a significant increase of IL-22 in the subepithelial region of four study groups, while the stronger expression of IL-22 in the epithelial region of OLP, OLM-drug and OLM-contact groups was identical. It may implicit Th17 and ILC3 involved in their inflammatory process.
Mast cells are sensitive to stimuli of neuropeptides (CGRP and Substance P), damage associated molecular pattern (DAMP) and pathogen-associated molecular pattern (PAMP). Those stimuli bind G protein-couple receptor (GPCR) or pattern recognition receptor (PRR) [7]. The degranulation of mast cells triggers innate immunity and aggravates adaptive immunity. OLM-dental group and OLM-drug group showed a relatively stronger expression in the subepithelial and submucosal regions. The severity of DAMP and PAMP in OLP and OLM shall be on the similar level. Therefore, we may assume neuropeptides involved in the inflammatory process of OLM-dental group and OLM-drug group significantly. CGRP is encoded by CALCA gene and works as a vasodilator binding Calcitonin Receptor Like receptor (CRLR) on the vascular smooth muscle cells [www.genecards.org]. It serves as a nociceptive signal to link Transient Receptor Potential Vanilloid 1 (TRPV1) receptor [8]. Peripheral neurons produce CGRP as per stimulation of TNF alpha [9]. Mast cells release TNF-alpha to stimulate neurons for production of CGRP, while CGRP links the CRLR (GPCG family) on mast cells for the further mast-cell degranulation. The higher expression of CGRP in the epithelial, subepithelial and submucosal regions was identified in four groups. The highest expression of CGRP was found in the OLM-dental group. Substance P (SP) is encoded by Tachykinin gene and effects as a vasodilator stimulating endothelium to release Nitric Oxide [www.genecards.org]. It works as a nociceptive signal to link Neurokinin-1 receptor (NK- 1R) [10]. Peripheral neurons produce SP as per stimulation of pro-inflammatory cytokines [11]. Mast cells release TNF-alpha to stimulate neurons for production of Substance P, while Substance P links the NK-1R (GPCG family) on mast cells for the further mastcell degranulation. The relatively higher expression of Substance P in the subepithelial region of the OLP group explains the clinical feature of the erythematous mucosa base.
Neuropeptide Y (NPY) is encoded by NPY gene and classified by ontology as GRCR activity and hormone activity [www.genecards. org]. It serves as a vasoconstrictor via peripheral sympathetic nerve fibers [12]. The hormone activity performs in the central nerve system level. The lower expression in the epithelial region of OLP, OLM-dental and OLM-drug groups might imply a degeneration probability of peripheral sympathetic nerve fibers. Neurofilament is encoded by NEFL gene and involved in maintenance of neuronal caliber, which forms the neuron cytoskeleton [www.genecards. org]. Neurofilament is type IV intermediate filament. Type I and II are epithelial keratin. Type III is desmin and vimentin. We used neurofilament heavily polypeptide (NF-H, 180-200 KDa) polyclonal antibody (Ab8135) to avoid the cross-reaction with type I, II and III intermediate filaments. Sensory nerve fibers within epithelium is not doubtable. The density of NF-H in the epithelial region of OLP group showed a significant decrease. It may draw a picture that a peripheral nerve fiber degeneration occurs in OLP group. Neurotrophin-3 (NT-3) is encoded by NT-3 gene and is classified in the family of Nerve Growth Factor (NGF). Vasoactive intestinal peptide (VIP) is a peptide hormone at intestine. They didn’t express in oral mucosal tissue.
Conclusion
1. Neurogenic inflammation does play a role in the inflammatory process of oral lichenoid reaction (OLR), but different types of OLR have different levels of involvement.
2. Lichenoid contact stomatitis from dental restorative materials (OLM-dental) significantly involves in neurogenic inflammation. The stronger expression of CGRP in epithelial, subepithelial and submucosal regions is special.
3. Oral lichen planus (OLP) most likely involve in cytolysistype of T-cell mediated hypersensitivity type IV. The partial loss of Neurofilament and Neuropeptide Y in the epithelium is unique.
4. Mucosal reaction to systemic drug administration (OLMdrug) most likely involve in delayed-type of T-cell mediated hypersensitivity type IV. The weaker expression of CD8 in epithelial region and INFG in the subepithelial region is distinctive.
Conflicts of Interest
All authors declare they have no conflict of interest.
Ethical Approval
All procedures performed in this study were in accordance with the principles of the Tri-Council Policy Statement Ethical Conduct for Research Involving Humans. This study was approved by Health Sciences Research Ethics Board of Dalhousie University with REB number 2016-4020.
Funding
This study was funded by Faculty of Dentistry Research Grant of Dalhousie University (No. 36650 and No. 34899).
References
- Yi Shing Lisa Cheng, Alan Gould, Zoya Kurago, John Fantasia, Susan Muller (2016) Diagnosis of oral lichen planus: a position paper of the American Academy of Oral and Maxillofacial Pathology. Journal of oral surgery, oral medicine, oral pathology and oral radiology 122(3): 332-354.
- Brad W Neville, Douglas D Damm, Carl M Allen, Angela C Chi (2016) Oral and Maxillofacial pathology (4th). Elsevier Inc. St. Louis, Chapter 9: 317-326 and Chapter 16: 729-734.
- Dimitris N Xanthos, Jurgen Sandkuler (2014) Neurogenic neuroinflammation: inflammatory CNS reactions in response to neuronal activity. Nature Reviews-Neuroscience 15(1): 43-53.
- Isaac M Chiu, Christian A von Hehn, Clifford J Woolf (2013) Neurogenic inflammation: the peripheral nervous system’s role in host defense and immunopathology. Nature Neuroscience 15(8): 1063-1067.
- Vinay Kumar, Abul K Abbas, Nelson Fausto, Jon C Aster (2010) Robbins and Cotran Pathologic Basis of Disease (8th). Saunders Elsevier Inc. Philadelphia, Chapter 6: 205-208.
- Lauren A Zenewicz (2018) IL-22: There is a gap in our knowledge. ImmunoHorizons 2(6): 198-207.
- George H Caughey (2007) Mast cell tryptases and chymases in inflammation and host defense. Immunol Review 217: 141-154.
- FA Russell, R King, SJ Smillie, X Kodji, SD Brain (2014) Calcitonin gene-related peptide: physiology and pathology. Physiology Review 94(4): 1099-1142.
- Schäfers M, Svensson CI, Sommer C, Sorkin LS (2003) Tumor necrosis factor-alpha induces mechanical allodynia after spinal nerve ligation by activation of p38 MAPK in primary sensory neurons. Journal of Neuroscience 23(7): 2517-2521.
- Yang Gu (2018) Interpretation of orofacial pain symptoms in the basis of new findings in central and peripheral sensitizations, Biomedical Journal of Scientific & Technical Research 3(3): 1-4.
- Terence M O’Connor, Joseph O’Connell, Darren I O’Berien, Triona Goode, Charles P Bredin, et al. (2004) The role of substance P in inflammatory disease. Journal of cellular physiology 210: 167-180.
- Aitak Farzi, Florian Reichmann, Peter Holzer (2015) The homeostatic role of neuropeptide Y in immune function and its impact on mood and behavior. Acta Physiologica(Oxf) 213(3): 603-627.

 Research Article
Research Article