Synopsis
In vitro fertilization (IVF) is a process that is exposed to high oxygen concentration (20%) resulting in the production of reactive oxygen species (ROS). N-acetyl-L-cysteine (NAC) is a potent free radical scavenger. However, it is reported that NAC supplementation during IVF has no or detrimental effect on oocyte development. The present study was designed to evaluate the effects of different concentrations of NAC supplemented at different stages of in vitro culture of bovine oocytes on fertilization rate and polyspermy. In Experiment 1, NAC was added at a fixed concentration (1.25 mM) to IVM medium (Group NAC IVM+/IVF-), to the fertilization medium (Group NAC IVM-/IVF+) and in both mediums (Group NAC IVM+/IVF+). In experiment 2, NAC was added only to the IVF medium at higher concentrations (5mM and 10mM). The control group were processed without the addition of NAC in mediums.In the present work, NAC (1.25mM) negatively influenced the rate of fertilized oocytes only in a NAC IVM+/IVF+ Group, while in Groups NAC IVM+/IVF- and IVM-/IVF+ a significant greater rate of normal fertilized oocytes has been recorded.In conclusion from the results of the present study we can state that addition of low doses of NAC (1.25 mM) to the IVM or IVF medium has positive effects on the quality of the fertilized oocytes by reducing polyspermic rates, while doses greater than 5 mM in IVF medium demonstrated a detrimental effect on the fertilization rate.
Keywords: Semen; In VitroFertilization; Polyspermy; N-Acetyl-L-Cysteine; Bovine
Abbreviations: IVF: In vitroFertilization; ROS: Reactive Oxygen Species; NAC: N-Acetyl-L-Cysteine; H2O2: Hydrogen Peroxide; BSA: Bovine Serum Albumin; TCM: Tissue Culture Medium; COCs: Cumulus-Intact Oocyte Complexes
Introduction
In vitro fertilization of oocytes in general is a process that is exposed to high oxygen concentration (20%) resulting in the production of reactive oxygen species (ROS) such as hydrogen peroxide (H2O2), superoxide anion (O2) and the hydroxyl radicals (OH) [1,2]. ROS are known to be involved in a wide range of physiological reproductive functions including sperm capacitation [3], oocyte maturation, steroidogenesis and corpus luteum function [4-6]. The negative effect of ROS on in vitro fertilized oocytes has been demonstrated in various animal species, due to induction of mitochondrial dysfunction[7], causing damage to nuclear material [8] as well as inhibiting oocyte-sperm fusion [9]. Oxidative stress occurs when oxidants outnumber the antioxidants[10]; therefore, a balance between production of oxidants and their scavengers is an aspect of in vitro fertilization that can affect its outcome [11]. Moreover, both enzymatic and non-enzymatic antioxidants are involved in contrasting oxidative damage caused by ROS. Glutathione (GSH), a tripeptidethiol, is the main non-enzymatic antioxidant produced by gametes and helps in providing a reduced environment in IVF [12].
Glutathione synthesis is stimulated by the presence of other low molecular weight thiol compounds such as cysteine, cystine, cysteamine and β mercaptoethanol, and reduced levels of GSH often lead to decreased numbers of competent oocytes during IVF [12,13].N-acetyl-L-cysteine (NAC) is a potent free radical scavenger as a result of its nucleophilic actions on ROS and can be considered as a supplement to overcome GSH depletion and free radical formations during oxidative stress [14]. The molecular structure of NAC facilitates crossing of the plasma membrane where it is deacetylated; in this condition, thiol groups are available for glutathione synthesis within the gametes [13,15,16]. However, some research on bovine oocytes indicates that NAC supplementation during oocyte maturation has either no or a detrimental effect on oocyte development [12,17]. Recently, Rocha Frigoni, et al. [18] evaluated the effect of antioxidants during IVM both on oocytes quality and acquisition of developmental competence; they concluded that antioxidants improved oocytes quality but not increased embryo production rate.
The effect of supplementing NAC appears to be time and dose dependent; when supplemented in IVF systems during early stage of development, it reduces embryonic survival [17,19], while an increase of foetal survival caused by reduction of oxidative stress was reported after oral administration in rat [20]. As regards the dose of NAC used in the IVF system there is a wide variability, ranging from 0.1-0.6 to 1,2 mM[17,21].Fertilization of the oocyte depends on the successful penetration by the sperm cell through the zona pellucida. Polyspermy is a process that involves penetration of the oocyte by more than one spermatic cell at fertilization [22].Higher in vitro incidence of polyspermy compared to those in vivo has been reported [23,24]. Some studies attribute polyspermy to a lack of exposure of the freshly collected oocytes to the heparin oviduct-specific glycoproteins complex that would help mask the fusion sites[22]. Other studies support the fact that reactions triggered by the release of the cortical granules contribute in blocking polyspermy [25-27].The present study was designed to evaluate the effects of different concentrations of NAC supplemented at different stages of in vitro culture of bovine oocytes on fertilization and polyspermy rate.
Materials and Methods
Ethics Statement
This study used ovaries obtained regularly from local bovine slaughterhouse, so no animal experiments were performed for the purpose of this study.
Reagents and Media
All reagents used were supplied by Sigma-Aldrich (Milan, Italy) unless otherwise specified.Medium for in vitro maturation (IVM) was tissue culture medium 199 (TCM199) (Gibco BRL, Grand Island, NY, USA) supplemented with 10% fetal calf serum (FCS; Gibco BRL), 0.2 mM sodium pyruvate, 25 mM sodium bicarbonate, 50 µg ⁄ ml amikacin, 0.5µg⁄ml FSH (Pluset; HertapeCalier, Juatuba, MG, Brazil), 100 IU⁄ml HCG (Profasi; Serono, Sao Paulo, Brazil) and 1.0µg⁄ml estradiol-17-β. IVF medium consisted of Tyrode’s albumin lactate pyruvate TALP [28], supplemented with 0.2 mM Na-pyruvate, 6mg⁄ml fatty acid-free bovine serum albumin (BSA), 25 mM sodium bicarbonate, 13 mM Na-lactate, 50 mg⁄ml amikacin, 4µl⁄ml PHE solution (2 mM penicillamine, 1 mM hypotaurine, and 250 mM epinephrine) and 10mg⁄ml heparin.
Oocyte Collection
Bovine ovaries were obtained from regularly slaughtered beef cows, which were declared clinically healthy based on pre- and postmortem Veterinary Inspection and transported to the laboratory in sterile NaCl solution (9gr/L) supplemented with antibiotics (100mg/L streptomycin) at 23/250C within 2 hours after slaughter. The ovaries were collected regardless of stage of the oestrous cycle of donors. Cumulus-intact oocyte complexes (COCs) were obtained by slicing ovaries and evaluated morphologically at a magnification 400× and classified as denuded, degenerated or healthy as reported by Vassena, et al. [29]. Only healthy COCs, with homogeneous oocyte cytoplasm and >3 layers of compact granulosa cells, were eligible for the experiment.
In Vitro Maturation
Pools of 30-35 oocytes each were incubated in four-well dishes, in 0.5 ml of IVM medium at 38.5°C under an atmosphere of 5% CO2 and 20% O2 in humidified environment for 24 hours. After maturation, COCs were then decumulated in 2ml medium H-SOF supplemented with 20μl hyaluronidase through the mechanical action of the pipette leaving only the cellular layers of the corona radiata; complexes were then incubated until the time of fertilization
In Vitro Fertilization
Frozen-thawed semen from Marchigiana bull with proven fertility was used. In order to obtain an adequate concentration of motile sperm for IVF, two doses were thawed, and semen was suspended on a Percoll gradient. In a 15 ml falcon 2 ml of 90% Percoll was deposited followed by 2 ml of 45% Percoll and finally the sperm suspension was layered on. The tube was then centrifuged at 300 xg for 40 minutes. Then pellet was collected and subsequently deposited in 2 ml of TALP medium and centrifuged twice at 300 xg for 10 minutes. Subsequently, sperm concentration was assessed through a haemocytometer; then the semen was resuspended in TALP-IVF medium in order to reach a final concentration of 2x106 sperm cells/ml and left to incubate at 380C 5% CO2 in humidified environment for 60 min. In each IVF well, 20 matured oocytes were co-incubated with 100µl of washed semen in an atmosphere of 5% CO2, 20% O2 at 38.5°C in humidified environment.After 18 hours of incubation, zygotes were stripped from cumulus cells, then mounted on slides and fixed for 48 hours with 25% acetic acid at room temperature. After fixation, slides were stained with 1% orcein in 45% acetic acid. Zygotes were then examined using a phase-contrast microscope at 400X magnification and considered as non-fertilized if one pronucleus was observed, normally fertilized when two pronuclei were evident, and fertilized but polyspermic if more than two pronuclei were identified.
Experimental Design
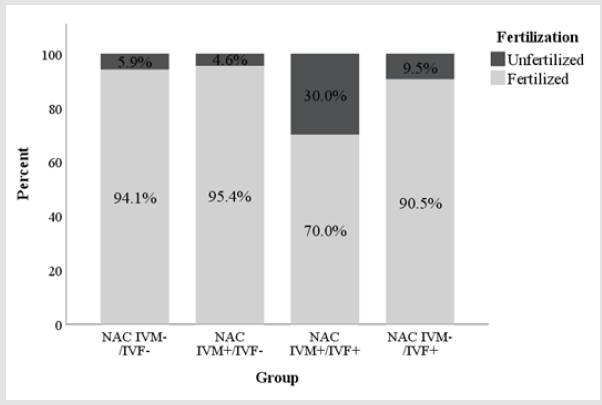
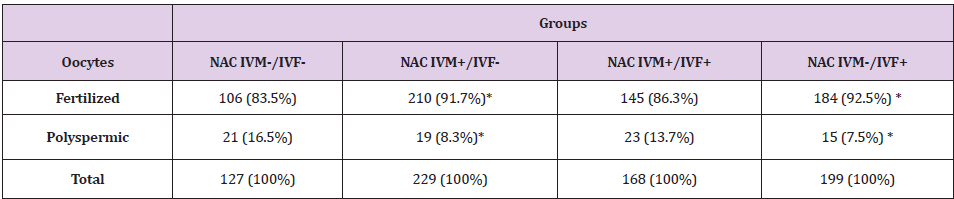
In the experiment 1, NAC was added at a concentration of 1.25
mM as follows:
i) Oocytes matured in IVM medium supplemented with
NAC and fertilized without supplementation of NAC in IVF medium
(Group NAC IVM+/IVF-).
ii) Oocytes matured and fertilized with the supplementation
of NAC in both mediums (Group NAC IVM+/IVF+).
iii) Oocytes supplemented with NAC only in the fertilization
medium (Group NAC IVM-/IVF+).
iv) Oocytes matured and fertilized without supplementation
of NAC on medium (Group NAC IVM-/IVF-).
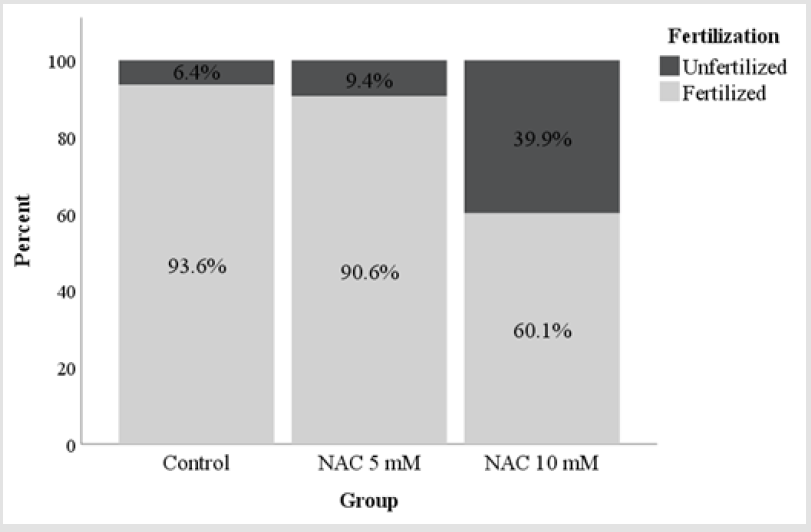
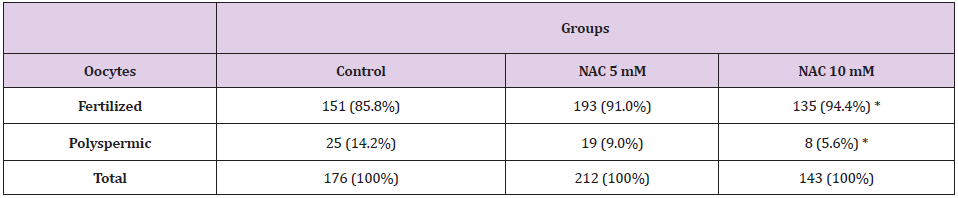
In experiment 2, in order to evaluate the effects of a higher
concentration, NAC was added at 5mM and 10mM to the IVF
medium. The control group was processed without the addition of
NAC in IVF medium.
Statistical Analysis
Statistical data in all experiments were analyzed using Chisquare test or Fisher’s exact test and Bonferroni-adjusted Z-test. Statistical analysis was performed using SPSS statistics version 23 (IBM, SPSS Inc, Chicago, IL, USA) and P < 0.05 was the accepted level of statistical significance.In both experiments, all the content within one well of a plate was defined as experimental unit while replicates were defined as the total number of wells analysed. Data were pooled from at list three replications.

 Review Article
Review Article