Questions That Officials and Institutions Should Be Clear About
a) Biosafety measures: protection for health personnel,
location of patients with COVID-19
b) Define advanced management strategies
c) Define the medication to be used in the rapid intubation
school
d) Mechanical ventilation management
a. What ventilation mode to use?
b. Use of protective mechanical ventilation parameters
c. When and how to put in a prone position?
d. What neuromuscular relaxant to use?
e) Patient route management for safe transfer to exams.
f) Bioethics and decision-making of ill patients with
prognosis
1) How long do you wait to evaluate response?
g) Route of the corpses and handling of them.
What is the Purpose of this Text?
Provide the scientific community with a concrete and summary review of the subject of COVID-19 in relation to published articles; it does not symbolize a mandatory measure, it is a guide, an aid for this time of health crisis.
Introduction
SARS-COV-2, COVID-19 or Wuhan virus is the last RNA of the seven RNA viruses described whit affection in humans of the family Coronaviridae subfamily Orthocoronavirinae of the order Nidovirals, characterized from the other positively charged RNA viruses (can be translated directly without the need for reverse transcriptase - Baltimore III-mRNA + classification) as having the largest genome, so its cycle and transmission is not entirely clear.
In the Coronaviridae family there have already been two epidemics the first in 2002-2003 with SARS-CoV (Severe Acute Respiratory Syndrome) and the second with the MERS-CoV (Middle East Respiratory Syndrome), whose outbreak was in the 2012, with a mortality of approximately 11% and 35% respectively, highlighting the high mortality that this group of viruses can have in humans [1,2]. Its name is derived from its microscopic shape similar to that of a solar corona and from the Latin corona, it has a wide distribution in the animal kingdom, mainly in mammals (porcines, felines, canines, humans, horses) and avian, considered a zoonotic infection, its clinical manifestations are mainly respiratory (from common flue to Acute respiratory distress syndrome -ARDS) and enteric symptoms (diarrhea), but in some cases neurological and liver involvement has been reported [1,2].
Morphology, Structural Proteins and Access Pathways
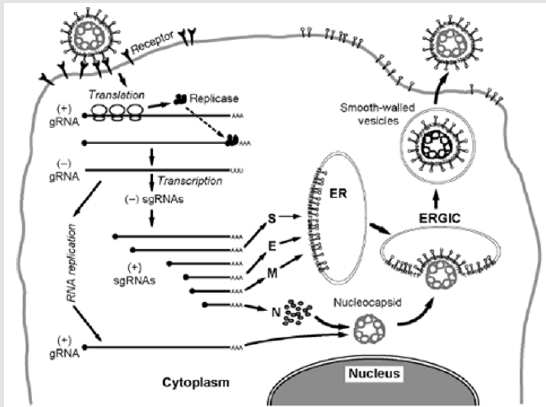
The virus has a spherical shape, with a pleomorphic surface, its envelope is composed of several proteins, the S Protein or Spike proteins (mediate the binding of the virus with the cellular receptor, the M protein or membrane protein shapes the surface of the virus and the E or envelope protein that is part of the virus surface and has a role in the fusion of the virus with the cell to be infected, on average the virus has a diameter of 80-120 nm. Inside the envelope is the nucelocápside protein or N protein, which is responsible for maintaining the binding of the virus genome and protecting it. There are also accessory proteins or that complement the replication and function of the virus such as the HE protein [1] (Figure 1). In Colombia on March 29, was obtained the first SARSCoV2 genome sequence that circulates in Colombia, SARS-CoV2 arrives in the country on February 26, 2020.
Viral Replication, Cycle and Assembly
Where Did it Come From?
On 31 December 2019, an epidemiological alert was issued for an outbreak of pneumonia caused by a new strain of coronavirus that occurred in Wuhan in Hubei Province, China, with possible epicenter the Huanan wholesale seafood market, where wild animals were sold illegally (bats, badgers), which is considered to have originated from bats [2,5,6]. The first confirmed outbreak was 41 patients, isolating the new virus on 7 January calling it the new coronavirus of 2019 or 2019-nCoV, later renamed as severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) and the pathology generated was called Coronavirus Disease 2019, acronym COVID-19 [7,8]. Its spread was rapid to the point that in less than three months of the outbreak in China, the World Health Organization (WHO) declared COVID- 19 as a pandemic on 11 March 2020, being present in more than 110 countries with an estimated attack rate of 1%, accounting for 118.322 infected patients and 4292 deaths [8].
How is it Transmitted and What is Your Incubation Time?
Transmission of the virus is by aerosols or drops, the secretions (saliva, feces, conjunctival) of the infected person or with exposure to an inert area or surface where the virus was [2,9], summarize as follows:
Person-To-Person
COVID-19 is transmitted by airway, fecal-oral and by contact of respiratory, conjunctival secretions, it has not been reported that sweat, seminal fluids or vaginal fluids transmit it.
At the Respiratory Level
The transmission is by inhalation of droplets containing the virus within a contact close to 6 feet or 2 meters away.
Contact with Surfaces, Contaminated or Inert Objects [10].
a) The droplets of 5 micrometers, maintain stability on the following surfaces or vehicles.
b) In the air in aerosol mode 3h.
c) Plastic, steel metal up to 72h.
d) Surfaces of copper (4h) and cardboard (8h).
It is therefore that COVID-19 has high propagation efficiency, reporting that by 1 infected patient it has already transmitted it to 2.2 patients [2], so preventive isolation means can decrease rapid growth in incidence. Its incubation period has been obtained in relation to SARS and MERS episodes with an average incubation time of 2-14 days after exposure to the vector [6,11].
How is it Diagnosed?
The most relevant thing at the moment is the epidemiological nexus whether it is to travel in the last 14 days to areas where this virus or contact with infected people, then the presence or not of symptoms should be evaluated (see below), it is essential not to panic since the first error is to consult emergency department for mild symptoms, in case of alarm signs (see below) consult these services so as not to generate saturation in the services and promote transmission of the virus. Likewise, each institution must have areas specifically designated for patients with suspected COVID-19, transfer routes, fitting beds in the intensive care unit (ICU) for management, institutional route guides, boarding and creating teams of experts to lead decision-making in difficult situations and the whole group handles the same language and follow the same technical-scientific and ethical guideline.
Symptoms of COVID-19 Infection
The clinical spectrum of COVID-19 range from asymptomatic
cases, mild rhinopharyngitis that account for approximately 80% of cases, to generating serious manifestations represented by 20%
mainly pneumonia that will require hospitalization with some type
of intervention, of which 5-8% will require intensive care in which
Acute respiratory distress syndrome (ARDS) will be their most
serious complication in a 46- 60% of cases. But not everything is
bleak as the resolution in mild cases is very high up to 80% [12,13],
so being at home if you have mild symptoms is the best way to help
the health team mitigate the pandemic. Generally, symptoms may
appear within 2 to 14 days of exposure to the virus and the three
most common symptoms are:
1) T fever 38oC with axillary thermometer.
2) Dry cough.
3) Shortness of breath or difficulty breathing.
4) Others: myalgia, fatigue, chills, gastrointestinal (diarrhea
and nausea).
If the person has ANY of the following symptoms or warning
signs, he or she should have prompt medical attention or consult
the emergency department.
1) Difficulty breathing.
2) Persistent chest pain or pressure.
3) Impairment in consciousness or inability to awaken.
4) Bluish lips or face.
5) Hemoptysis (cough with blood).
What Symptoms Does the Medical Literature Report?
From the population of Wuhan described by Huang et al [2], Wang et al [14] and Zhu et al [11], Guan et al [15] and Zhou et al [16] described that the symptoms and characteristics of the patients were:
Demographics
a) Similar rate in men and women (54% men).
b) Average age between 42-68 years.
c) Transmission to health personnel was 29% and hospital patients were 12.3%, implying that the 41% of cases are processed at the nosocomial level.
The Most Common comorbidities Were
a) Diabetes 10-20%.
b) High blood pressure 15-30%.
c) Cardiovascular disease 15%.
d) Cancer 5.6%.
e) More than 50% of patients had no comorbidities [2,11,14].
(Table 1)
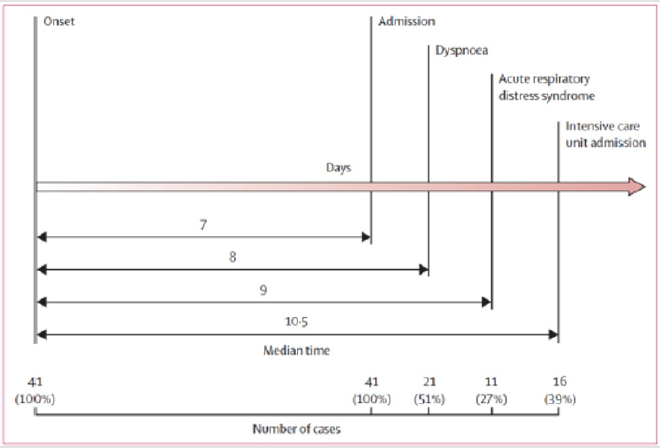
If we make a timeline like the one shown in Figure 3 regarding the development of symptoms since the onset of the disease it is described that admission to the hospital occurred on average at 7 days (1-8 days), dyspnea at day 8 (0-0 9 (0-14 days), need for mechanical ventilation 9-10 days (7-17 days) and admission to ICU on day 10-11 ( 8-17 days) , gives us a picture to be able to evaluate the clinical evolution of these patients indicating that the course is stationary between 7-10 and after that time you can see the progression or improvement of the picture, it should be noted that the severity can be present from the beginning of the symptoms. Regarding hospitalization time in non-complicated cases were on average 10-12 days and in ICU 8-10 days [2,11,14,15,16] (Figure 3).
In the blood chemistry, The Most Common Abnormalities Were
a) Lymphopenia (<1000 x10-9) 63-70%.
b) Thrombocytopenia <150,000 36-40%.
c) Prolonged activated partial thromboplastin time (PTT)
test 58%.
d) Elevated lactate dehydrogenase (LDH) >240 U/L: 40-
74%.
e) C-reactive protein (CRP) elevated > 10mg/L 40-60%.
f) D-dimer > 500 mcg/L 3-2%.
g) Transaminase elevation > 40 U/L 20-40%.
h) Creatine kinase fraction MB (CK-MB) > 180 U/L 15%.
It should be noted that, although not all of the following
laboratories were statistically significant, having elevated D-dimer,
lymphopenia, elevated LDH, CK-MB and elevated liver function
was seen more in ICU patients, with an average admission SOFA
of 5 points , indicating that these laboratories might indicate
that patients would be more likely to have a more severe disease,
emphasizing that lymphopenia could be considered a marker of
this disease from baseline[2,11,14].
Procalcitonin (PCT) and COVID-19
The Huang and Wang studies (2.8) did not report PCT elevation, suggesting that if it rises in these patients it may be a sign of bacterial infection, which was seen at 25% (PCT %-0.5 ng/ml) in the Huang series and 75% in Wang’s respectively, indicating that the PCT may have the identification of bacterial infection in these cases.
Radiologically
Lung abnormalities can be identified in 86% of patients
from the first day of admission to the hospital, where extensive
pulmonary involvement is evident in chest x-ray in more than two
quadrants, the most common patterns on pulmonary tomography
finds were [2,14,11,15,17].
a) Bilateral ground glass patchy opacities in peripheral or
subpleural areas 95-98%
b) Bilateral compromise of ground glass, without affecting
central space 70-98%
c) Multilobar consolidation in subpleural area 70-80%
d) Other findings: Crazy paving pattern (thickening of
interlobulillary septum with opacity with ground glass),
subpleural reticular interstitial pattern, halo signs nodules,
diffuse consolidation or bat sign.
e) Findings to think in other etiologies instead of COVID-19:
pleural effusion, lymphadenopathies, cavitation’s, three in bud.
(Figure 4).
POCUS and COVID-19
Little have been reported from the POCUS (Point of care ultrasound) experience, in which pulmonary ultrasound with microconvex, convex or sectoral probe have been used to identify different patterns, in which pattern B, interstitial pattern, consolidation pattern has been described, it is important that, if this tool is available and no CT scanner is available, ultrasound is used.
Place of Destination and Complications
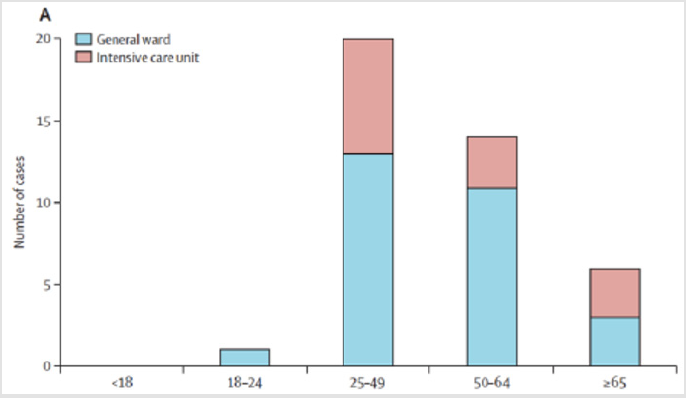
Among Huang and Wang’s studies, the average number of patients who were hospitalized were mostly in general rooms 65-70% and 25-30% in the ICU with age variation as outlined in Figure 4. Regarding its complications the most common were the development of ARDS, shock, heart injury and secondary infection, attributed by cytokine release syndrome (cytokine storm) varying in the groups of ICU vs No ICU (general room) (Figure 5) (Table 2).
Cardiovascular involvement and COVID-19
Coronaviruses have tropism and extensive cardiovascular disease in case of SARS and MERS with spectra of myocardial injury and acute or chronic heart failure, the various manifestations that have been reported in the Cases of Wuhan of COVID-19 reflect this same pattern [2,11,14,15], a pattern manifested with development of arrhythmias, acute heart injury manifested with elevated troponin and CPK-Mb, even reporting that some patients consulted in the emergency room not for respiratory symptoms but by cardiovascular symptoms given by palpitations and chest pain [3] showing SARS-CoV2 may have clinical outcomes such as acute viral myocarditis , shock and acute heart failure [3,18]. It is important to consider the role of cardiology societies in managing patients with cardiovascular disease and COVID-19, in which Driggin and colleagues make an impeccable summary [6].
Cardiovascular Spectrum and COVID-19 [6,16]
a) Myocardial injury: elevation of cardiac biomarkers
(troponin, CPK-MB).
b) Acute heart injury: elevation of biomarkers >p99th +
electrocardiographic and echocardiographic changes.
c) Arrhythmias: do not specify what type, but is considered
in relation to metabolic, hypoxic and neurohumoral stress.
d) Acute heart failure: present in an average of 23% and
more than half of patients who died in studies in China [16].
e) Risk of development of post-infection acute coronary
syndrome.
f) Others: Fulminant viral myocarditis.
Used Therapies Reported in Literature
The experience that is had in the use of therapies is based on the report of the different series and therefore find levels of AI evidence, it will be very difficult, and we will guide ourselves with the recommendations of Class C i.e. expert concept and evidence level II-III, which will be case reports, case series and some small cohorts From the previous what was reported has been:
a) Oxygen support.
i. Nasal cannula 66% or
ii. NIMV (Non-invasive mechanical ventilation) or HFCN
(high-flow nasal cannula) 41%.
iii. IMV (Invasive Mechanical Ventilation)47% or
iv. ECMO 2-5%.
b) Antibiotics: 100%.
i. Reported moxifloxacin, azithromycin, ceftriaxone.
c) Antivirals 89-93%.
a. Oseltamivir 75mg (80-93%).
b. Lopinavir /ritonavir.
d) Systemic steroids 22-45% (methylprednisolone).
e) Vasopressors 22%.
f) Immunomodulators
i. Immunoglobulin 13-24%, tocilizumab, chloroquine,
hydroxychloroquine.
g) Renal replacement therapy (RRT) 7%.
Prognosis
At the moment unlike their relatives SARS and MERS who had a mortality of 11% and 35% respectively [2], COVID-19 mortality has been variable in WHO statistics, with 4.3% in Wuhan, China up to 8% in Italy with an average of 4.5% [12], of course these data must be constantly being monitored, but it is not a negligible mortality to be assessed in relation to population size, mentioning that the main patients at risk of mortality were those with cardiovascular comorbidity or have advanced age. An example of this is Colombia with an estimated population of approximately 50,000,000 habitants with a serious estimate of infection [17]. In Wuhan case studies, mortality in ICU averaged 38% and non-ICU 15% studies [2,9,11].
Case Definition (Colombian Guides- National Institute of Health)
Following guidelines similar to those of WHO, the National Institute of Health with the guidance ‘GUIDELINES FOR THE DETECTION AND MANAGEMENT OF COVID-19 CASES BY HEALTH SERVICE PROVIDERS IN COLOMBIA VERSION 05, Ministry of Health and Social Protection Bogotá, March 19, 2020, classified in 5 cases the suspicion of patients with COVID-19 being these:
a) Case 1 Patient with unusual Severe Acute Respiratory
Infection (uSARI).
b) Case 2 Symptomatic non-serious patient who does NOT
require hospitalization.
c) Case 3 Severe Acute Respiratory Infection (SARI)
intensified surveillance for COVID-19 sampling.
d) Case 4 Probable death by COVID-19.
e) Case 5 Asymptomatic patient.
It is also Essential to Define Close Contact
a) Contact between two people in space less than 2m/6ft with a confirmed or probable case for 15 minutes
b) Having direct contact with secretions of a probable or confirmed case
Cases are Defined as Follows
Case Criteria 1
Probable Case: Patient with cough and fever, with severe acute respiratory infection box - SARI who develops an unusual or unexpected clinical course, especially a sudden deterioration despite proper treatment, requiring hospitalization (does not require travel history) (unexpected SARI).
Case 2 Criteria
Probable Case: Person with at least 2 of the following symptoms: fever greater than or equal to 38oC, cough, shortness of breath, odynophagia, fatigue and also has one of the following (NO HOSPITALIZATION REQUIRED MANAGEMENT): a. Travel history or that you have lived in China or countries and areas with viral circulation confirmed of the new coronavirus (SARS-CoV-2) in the 14 days prior to the onset of symptoms, or b. An occupation as a health worker or other staff working in an environment that cares for IRAG patients with unknown etiology, or c. A history of close contact in the last 14 days with a probable case or confirmed of severe acute respiratory infection by the new coronavirus.
Case 3 Criteria: Surveillance of severe acute respiratory infection (SARI) in hospitalized patient
Person with acute respiratory infection (ARI) with a history of fever and cough no more than 10 days of evolution, requiring inhospital management, samples should be taken for COVID- 19.
Case Criterion 4: Probable death by COVID-19
All deaths from severe acute respiratory infection with clinical picture of unknown etiology.
Case Criterion 5: Asymptomatic case
Narrow contact of confirmed case COVID-19 that has not developed symptoms in the first 7 days after the last unprotected exposure.
Case classification
Several revisions are there to classify severity, this review will take the classification of the Handbook of COVID-19 Prevention and Treatment, made by Chinese authors who handled COVID-19 infection [13], which offer simple criteria and the severity classification data have been taken based on them [15], however, there are other guidelines such as those of WHO [4].
Mild Cases 80%
Clinical symptoms are mild, and no manifestations of pneumonia can be found in imaging.
Moderate Cases
Patients have symptoms such as fever and respiratory tract symptoms and may have manifestations of pneumonia in the images.
Severe Cases 14%
a) Adults who meet any of the following criteria
1) Respiratory frequency >= 30 breaths/min
2) Oxygen Saturations <=93% at sea, in Bogota <90%*
3) Ratio PaO2 /FiO2 <= 300 mm Hg
4) Patients who more than 50% of lung injuries progressed
within 24-48h.
Critical Cases 5%
a) Meets any of the following criteria (Derive to Intensive
Care Unit/COVID-19)
1) Appearance of Respiratory Failure Requiring Mechanical
Ventilation
2) Presence of shock
3) Another organic failure that requires monitoring and
treatment in the ICU
Critical cases are divided into early, middle and late stages
according to the oxygenation rate and state of the respiratory
system.
Early Stage
1) PaO2 /FiO2 150-100 mm Hg; Pulmonary distensibility > at 30 ml / cm without organ insufficiency apart from the lungs.
2) It has a great possibility of recovery through active antivirals, cytokine anti-storm and supportive treatment.
Middle Stage
PaO2 /FiO2 100-60 mm Hg; Pulmonary distensibility 30-15 ml / cm can be complicated by another type of mild to moderate dysfunction of other organs.
Late Stage
1) PaO2 /FiO2 less than 60 mm Hg; Pulmonary distensibility less than 15 ml / cm, diffuse consolidation of both lungs requiring the use of ECMO; Or failure of other vital organs.
2) The risk of mortality increases significantly.
Addressing and Diagnostic Tools
The first step is the triage system in which the physician trained to identify these patients, does so and provide them with a conventional eye cap, gloves and refer it to the designated isolation area for those patients where drop and contact protection must be present. If the patient is stable and without an alarm sign, he or she may be referred home and contacts his health insurance or National health institute (INS) for sampling, if the patient with alarm signs will be hospitalized, the samples for COVID- 19 established by nasal swab or pharyngeal, basic blood chemistry studies will be taken: blood count, clotting times, liver function, renal function, chest x-ray, electrolytes, LDH. In our medium for the diagnosis of COVID-19 is counted with the RT- PCR (real time- polymerase chain reaction) method, this sample is obtained from respiratory swab (nasal or pharyngeal) with an average process time of 3-4 days, is taken in the clinic where the case is and is derived to the INS laboratory. It is important to mention that this method is not 100% sensitive, it has a sensitivity of 61-71% implying a margin of false negatives of 29-30%, recently published as the use of tomography could supplement and rectify false negatives by the RTPCR method, indicating that 93% of patients may have a suspicious chest tomography before the result of the chest-PCR [19,20]. In case the patient arrives with indication of orotracheal intubation (see below) it should be referred to the intensive care unit or isolation unit designated in order to perform the procedure and mitigate the biological risk.
What Protective Measures to Take?
In general, the most important protective measures to be taken
are:
1) Be quarantined during the pandemic: leave alone if
necessary, if infected DO NOT leave, designate someone for
food.
2) Proper hand washing as it reduces transmission of the
virus by 50%.
3) If you are infected with COVID-19, remain in strict
quarantine.
Protective Measures at Home
a) The insulation of the carrier case applies to grooming,
clothing, places that are exposed.
b) Hand washing with soap water for at least 60 seconds or
with alcohol-concentration gel 60%.
c) Hand washing should be performed.
1) After blowing your nose, coughing or sneezing o After
using the bathroom
2) Before eating or preparing food.
3) After contact with animals or pets.
4) Before and after providing routine care to another person
who needs assistance (for example, a child)
d) Use of chlorinated solutions to clean surfaces
1) 1 gallon of water (3.8 liters of water) + 5 tablespoons of chlorine or 1/3 cup is mixed so that a mixture with the
eradication capacity of the virus remains.
2) This mixture can be packaged in a disperser or atomizer
to be applied to surfaces.
e) Avoid contact of the hands with mucous membranes
(eyes, nose, mouth).
f) Home care INS guidelines or
g) Home isolation INS guidelines or
https://www.minsalud.gov.co/Ministerio/Institucional/ Procesos%20y%20procedimientos/GIPS06.pdf.Protection Measures Away from Home
a) Ideally not to leave unless strictly necessary, local or
national lines should be followed when the quarantine is
established.
b) Bottom line: Better to stay home and not go out.
Protective Measures in Hospitals
a) Recommendation of personal protective elements (PPE) according to the level of exposure or https://www.minsalud. gov.co/sites/rid/Lists/BibliotecaDigital/RIDE/VS/tabla-eppconsenso. pdf
b) Handwashing with soap and water in 5 moments or using
alcohol of 60% concentration.
c) Patients with probable, suspected or confirmed cases
should be isolated using aerosol and contact protection measures
1) The sick must wear a mask.
2) Have a default route for the patient to minimize patient
exposure with the medium.
Personal Health Biosecurity Equipment
a) High-efficiency masks FFP2 (In the case of procedures
with risk of aerosolization)
b) Eye protection with goggles
c) Long-sleeved clean bat.
d) Use double glove if in contact with coVID-19 patients.
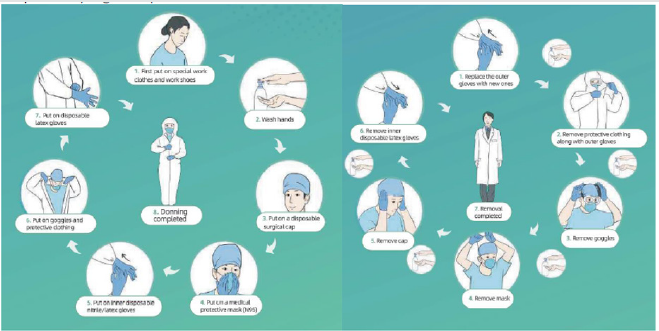
The following illustrations from reference 12 illustrate how-to
put-on PPE and how to remove them, remember to be patient, and
follow the steps (Figure 6).
Handling and Disposition of Corpses
a) The Ministry of Health already has guidelines for how to gag, to manage the patient’s corpse with COVID-19 on the following link:
b) https://www.minsalud.gov.co/sites/rid/Lists/ BibliotecaDigital/RIDE/VS/ED/VSP/orientaciones-handlingcadaveres- covid-19.pdf
c) The most relevant here the person who performs it must ALWAYS maintain the high-efficiency head covering and the hospital where they work must already have the route of these patients established.
Special Population
Mothers and COVID-19 (19)
a) Few studies have described this relationship at the time
only a series of 9 maternal cases have been reported in the
third trimesters, in which the clinical course was similar to
that described in the general population in relation to the
clinic concluding from this report:
1) No fetus-placental transmission of COVID-19 was
demonstrated, not found in breast milk.
2) It is not established if there is vertical transmission since
all deliveries were by caesarean section.
3) None of the maternal ones died or severe SDRA
development.
4) There were no postpartum complications of the newborn
or alterations in APGAR.
b) WHO recommends the support measure as the leading
therapy in this group of patients [17]?
Treatment
There is currently no specific treatment or proven vaccine for any coronaviruses or that are endorsed by regulatory bodies such as FDA, EMA or INVIMA in our country, however it is emphasized that if we expect to find treatments with AI level of evidence (randomized studies, double-blind, meta-analysis) will be very difficult to adjudicate the effectiveness of a treatment in these times, that is why we must go to gray literature or what at a level of recommendation will be C (small observational studies, expert opinion), with Class II-III evidence (series of cases, cases and controls or small cohorts), the most important thing is to follow the guidelines of ethics and the principle of non-maleficence. Available pharmacological treatments have been demonstrated in in vitro studies, few in vivo, through small series of cases and controls, describing as objectives: reducing the amount of circulating RNA (RNAemia), impacting clinical outcomes and impact on mortality, that at the moment none of the pharmacological therapies to be mentioned below to complicate all of these objectives, the Atlanta CDC on its page (https://www.cdc.gov/coronavirus/2019-ncov/ hcp/therapeutic-options.html) mentions some pharmacological therapies and may serve as a reference with WHO to consider the use of certain therapies [4,21]. The main current management is supportive and biosecurity therapy (contact and droplets, oxygen therapy, symptomatic management), below will describe what has been used and reported with more clinical involvement, but many therapies have been described experimentally [6] The approach may be made if the patient has mild-moderate symptoms or has signs of severe acute respiratory infection (IRAG), the latter outlines WHO guidelines in its COVID-19 severe respiratory infection management guide [21].
Isolation and Biosecurity Measures
a) It is the first tool to be implemented with isolation of the
patient with droplets and contact, in a unit where only those
cases are, to mitigate hospital transmission.
b) Implement special routes for these patients.
c) Safe sample handling.
Oxygen Therapy
a) Oxygen substitution is the first most commonly used pharmacological measure in descriptive studies, with indication of nasal cannula when oxygen saturation (SatO2) <=90% or a PaO2/FiO2 <= 300 with a SatO2 goal ,90% in non-pregnant women and >=92-95% [21].
b) if PaO2/FiO2 between 200-300 mmHg the use of HFNC with flow of 50-60 L/min can be considered.
IOT indications [13]
a) PaO2/FiO2 <150 mmHg in patients under 60 years and over 60years PaO2/FiO2 < 200mmHg
b) Respiratory frequency < of 30/minute
c) Glasgow comma scale =< 8
d) CO2 greater than 50 mm Hg
e) Respiratory clinical course impairment whit signs of multiple organ failure following therapy with another oxygenation medium.
Invasive Mechanical Ventilation
In this scenario the patient must managed by an expert in this subject, since it is possibly found in a case of ARDS (acute respiratory distress syndrome) but the following parameters can initially be adjusted by no non-expert personal
a) Ventilation Mode: Volume-Controlled Assist-Control
Mode (A/C).
b) Current or tidal volume (VC/VT) 6-8 cc/kg by ideal weight.
a. Adjust according to peak pressure and pulmonary
distensibility
b. Predicted Weight:
c. Male: 50+0.91(height-154).
d. Woman: 45.5+9.91(height-152.4).
c) SotO2 Goals: 86-90% or
d) Initial PEEP 8-12 cm H2O, pitiable according to the
method that has the most knowledge (distensibility, driving
pressure, ultrasound, etc.).
a. Objective of the PEEP: maintains alveolar recruitment and
facilitates the exchange Pulmonary
e) Respiratory rate 12-25, higher generates air entrapment
f) Ratio I: E 1:2-3
g) FiO2: initially high, but carry 60-50% to mitigate adverse
oxygen events
h) Peak pressure and plateau press < 30 cm H2O
i) Driving pressure < 15 cm H2o
Other Therapies
1) Pronation and ECMO: topic to be determined at each
institution
2) Pronation: consider early pronation (PaO2/FiO2 ratio 140- 150 mmHg), 18h of prone position rest 6h.
Other Therapies
1) Pronation and ECMO: topic to be determined at each
institution
2) Pronation: consider early pronation (PaO2/FiO2 ratio 140- 150 mmHg), 18h of prone position rest 6h.
Antibiotics
a) It is vitally important that having COVID-19 is not an
indication of antibiotic therapy especially in mild to moderate
cases. The indication to be evaluated in severe cases where
bacterial overinfection is suspected in which the therapy to be
chosen will be in relation to the local guides of each hospital or
having as a line the national guidelines of community-acquired
pneumonia [22], remembering that before considering the
initiation of antimicrobial therapy, take blood cultures, Gram
samples and cultures of secretion/sputum, procalcitonin, so
to be able to perform an effective management of antibiotics
and remove it appropriately since ideally they will be removed in less than 48-72 if there is no documentation of bacterial
infection.
b) In case of macrolide association associate management
with azithromycin 500mg day 1, then 250mg day for 5 days,
with strict QT surveillance recommendation.
c) In the case of the patient with unexpected SARI following
the national lines should be initiated with oseltamivir 75 mg
every 12h for 5 days, in relation to influenza virus or H1N1,
whit the subsequent rapid molecular testing such as PCR or
the one available at your institution to identify such coinfection
and thereby determine the continuity of the antiviral.
Antivirals and SARS Cov-2
a) As previously mentioned, no antiviral is approved by FDA,
EMA or INVIMA for the management of SARS-Cov-2, but due to
the rapid progression of the disease SARS and MERS data have
been taken to indicate antiviral therapy in SARS-Cov-2.
b) The main objective of antiviral therapy is to reduce viral
load and thereby mitigate severe clinical outcomes of the
virus, currently data from remdesivir, lopinavir/ritonavir,
Darunavir/cobicistat.
c) Remdesivir: is a nucleoside analogue with in vitro and in
vivo activity against COVID-19, which is incorporated into viral
RNA generating an early termination of coVID-19.
1) Posology: 200mg IV D1 then 100mg day IV for 5-10 days.
2) Currently several experts consider it if available as a first
line, their evidence in combination with other therapies such
as antimalarials have shown an impact on outcomes described
above [23].
3) Interactions: Relevant since it has CYP3A4 system.
4) Adverse events: hypotension during infusion, not in
patients with CHILD C or TFG < 30 mL/min/1.73 m2.
5) Ongoing studies: NCT04252664, NCT04257656.
Ritonavir/lopinavir Tab 200mg/50 mg or Sol Oral (80mg/20mg)/ml
a) Posology: 400 mg/100 mg twice daily (two capsules or 5
mL VO or per SNG for up to 14 days
b) Recently the NEJM public a study of control cases where
the mortality rate at 28 days was numerically lower with
intervention vs standard management (19.2% vs 25) but had
no statistical significance [24].
c) Interactions: inhibits the CYP3A4 system
d) Adverse events: QT prolongation being watching you,
Gastrointestinal (nausea and diarrhea), hypertriglyceridemia
Immunomodulatory Therapy
Hydroxychloroquine and Chloroquine
a) Alters the pH of the endosome it requires for the fusion
of the virus with the cells, and they also describe mitigating
cytokine release syndrome.
b) Hydroxychloroquine [25,26].
1) Posology: Loading dosage 400mg every 12h day 1, then
200 mg BID for 5 days
a) Ongoing studies: ChiCTR2000031174, NCT04261517
c) Chloroquine Phosphate Tab 250 mg (150 mg chloroquine
base)
1) Posology: 500mg every 12h for 5-10 days
2) In the FAZHU hospital experience the scheme used was
500mg BID for 2 days, then 500 mg day for 5 days [13].
3) Ongoing studies: ChiCTR2000031204,
ChiCTR2000030718
d) Interactions: Relevant as both inhibits inhibits CYP2D6
e) Adverse events: watch QT, documented macular toxicity
and atrial blockage are long but surveillance in both molecules
of QT.
Glucocorticoids
a) Its use is controversial and not proven to be clear benefit,
the main argument is the delay of the purification of RNAI, in
addition the presence of H1N1 infection should be ruled out
already if increased mortality is administered [27].
b) Indications follow in relation to refractory shock, presence
of ASTHMA or COPD
c) Posology: 1-2mg/kg/day per 5 days
d) Steroid studies and COVID-19 underway:
ChiCTR2000029386
Conclusion
Knowledge of respiratory disease caused by SARS-CoV-2 is constantly evolving, so it is necessary to stay up to date on the results of developing clinical trials and real-world experience to match therapy for the greatest benefit of patients. In the same way it is necessary to consider the extrapulmonary manifestations (cardiovascular, digestive, neurological, among others) to recognize complications early. Regarding treatment, it should be guided by institutional schemes and based on the body of evidence available pending further global clinical data. Finally, it is important to emphasize the vital importance of population prevention measures in order to reduce transmission chains, prevent the collapse of health systems and thus alleviate the morbidity resulting from this infection.
Competing Interests
The author declare that they have no competing interests.
Acknowledgement
To the effort made by the COVID-19 unit in the Hospital Universitario Mayor, Mederi, Bogota Colombia.
References
- Masters PS (2006) The Molecular Biology of Coronaviruses. Vol. 65, Advances in Virus Research. Academic Press pp. 193-292.
- Huang, Yeming Wang, Xingwang Li, Lili Ren, Jianping Zhao, et al. (2020) Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 395: 497-506.
- Zheng Y, Yi-Tong Ma, Jin-Ying Zhang, Xiang Xie (2020) COVID-19 and the cardiovascular system. Nat Rev Cardiol.
- Ensayosclinicosencurso COVID-19.Estudios Chinos.
- Emergenciaen china por nuevo coronavirus.
- Elissa Driggin, Mahesh V Madhavan, BehnoodBikdeli, Taylor Chuich, Justin Laracy, et al. (2020)Cardiovascular Considerations for Patients, Health Care Workers, and Health Systems During the Coronavirus Disease 2019 (COVID-19) Pandemic, Journal of the American College. of Cardiology.
- (2020) Coronaviridae Study Group of the International Committee on Taxonomy of Viruses. The species Severe acute respiratory syndrome-related coronavirus: classifying 2019-nCoV and naming it SARS-CoV-2. Nat Microbiol.
- (2020) Declaración de OMS: COVID-19 es una pandemia WHO Situation Report.
- Reporte No 7 de situación de COVID 19 por OMS.
- Van Doremalen N, Bushmaker T, Morris DH, Holbrook MG, Gamble A, et al. (2020) Aerosol and Surface Stability of SARS-CoV-2 as Compared with SARS-CoV-1. N Engl J Med.
- Zhu, Dingyu Zhang, Wenling Wang, Xingwang Li, Bo Yang, et al. (2020) A Novel Coronavirus from Patients with Pneumonia in China, 2019. N Engl J Med 382: 727-733.
- (2019) Casos de OMS de COVID-19, consultado 20 de marzo.
- Handbookof COVI0-19 Prevention and Treatment.
- Wang, Bo Hu, Chang Hu, Fangfang Zhu, Xing Liu, et al. (2020) Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus–Infected Pneumonia in Wuhan, China. JAMA 323(11): 1061-1069.
- Guan, Zheng-yi Ni, Yu Hu, Wen-hua Liang, Chun-quanOu, et al. (2020) Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med.
- Fei Zhou, Ting Yu, Ronghui Du, GuohuiFan, Ying Liu, et al. (2020) Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study . Lancet 395(10229):1054-1062.
- Clinical characteristics and intrauterine vertical transmission potential of COVID-19 infection in nine pregnant women: a retrospective review of medical records.
- Tian-Yuan Xiong, Simon Redwood, Bernard Prendergast, Mao Chen (2020) Coronaviruses and the cardiovascular system: acute and long-termimplications. Eur Heart J.
- DANE poblacioncolombiana.
- Ai T, Yang Z, Hou H, Zhan C, Chen C, et al. (2020) Correlation of Chest CT and RT-PCR Testing in Coronavirus Disease 2019 (COVID-19) in China: A Report of 1014 Cases. Radiology: 200642.
- (2020) World Health Organization. (2020). Clinical management of severe acute respiratory infection when novel coronavirus (2019-nCoV) infection is suspected: interim guidance, 28 January 2020. World Health Organization.
- (2013) Recomendaciones para el diagnóstico, tratamiento y prevención de la neumoníaadquiridaen la comunidadenadultosinmunocompetentes. Infectio 17(Supl 1): 1-38.
- Manli Wang, Ruiyuan Cao, Leike Zhang, Xinglou Yang, Jia Liu, et al. (2020) Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro.Cell Res 30(3): 269-271.
- A Trial of Lopinavir–Ritonavir in Adults.
- Philippe Gautret, Jean-Christophe Lagier, Philippe Parola , Van Thuan Hoang , Line Meddeb, et al. (2020) Hydroxychloroquine and azithromycin as a treatment of COVID‐19: results of an open‐label non‐randomized clinical trial. International Journal of Antimicrobial Agents.
- Yao X, Ye F, Zhang M, Cui C, Huang B, Niu P, et al.(2020) In Vitro Antiviral Activity and Projection of Optimized Dosing Design of Hydroxychloroquine for the Treatment of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). Clin Infect Dis.
- Han K, Ma H, An X, Jing Chen, Zhiyong Lian, et al. (2011) Early use of glucocorticoids was a risk factor for critical disease and death from pH1N1 infection. Clin Infect Dis 53: 326-333.
Videos informativos e informaciónadicional
https://www.youtube.com/watch?v=9gJV2NcVcps&feature=push-u-sub&attr_tag=cZfT0Q-QhXJ0207T%3A6
https://www.youtube.com/watch?v=bKJVYkAAogI&feature=push-u-sub&attr_tag=SVczckSAAr0PvrmL%3A6
https://www.youtube.com/watch?v=LAwfRkBtFAo&feature=push-u-sub&attr_tag=fnNFj4hV3G4SDzLE%3A6

 Review Article
Review Article