Abstract
We have described the characteristic ultrasonographic features using highresolution ultrasonography (US) in palmar fibromatosis or Dupuytren’s disease and plantar fibromatosis or Ledderhose disease. The author suggests that Dupuytren’s disease occurs at the distal metacarpal (MC) region of the fouth digit in the subcutaneous tissue of the hand. It is strongly suggested that gray-scale US provides the association of the lesion and the flexor tendon and that Real-Time Tissue Elastograhy (RTE) images reflect the estimation of elasticity alteration in palmar fibromatosis. Clinically and genetically, it may be important that plantar fibromatosis shoud be considered as a concomitant disease of palmar fibromatosis.
Keywords: Dermatologic Ultrasound; Palmar Fibromatosis, Plantar Fibromatosis, Giant Cell Tumor of the Tendon Sheath, High-Resolution Ultrasonography
Introduction
Dermatologic ultrasound imaging has been rapidly growing in recently years [1,2]. Currently, ultrasonography (US) studies for superficial fibromatoses, palmar fibromatosis, namely Dupuytren’s disease [3] and plantar disease, namely Ledderhose disease [4] which are located in the subcutaneous tissue have been described in ultrasound field. Dupuytren’s disease is a routine for the dermatologist, because patient with subcutaneous nodule may firstly visit a dermatologist. In this review, the author provides an update on the characteristic US appearances in superficial fibromatoses, palmar and plantar diseases.
Dermatological Ultrasound
The international working group, namely DERMUS (Dermatologic Ultrasound) was formed and provided the guidelines [1] and proposed for an assessment training program [2]. Currently, US reports for palmar fibromatosis, namely Dupuytren’s disease [3] and plantar disease, namely Ledderhorse disease [4] which are soft tissue tumors have been described in ultrasound field. We usually perform US studies for dermatologic fields with a high-resolution, broad-band (5MHz-18MHz) linear transducer (Nobulus Hitachi, Ltd.Tokyo, Japan). We have also provided several reports of the dermatologic lesion [5-16].
Normal Skin Elasticity
The author has previously described anatomy in dermatological ultrasound [16]. Klauser et al. [17] suggested that red is used to represent softer tissues, blue represents harder tissues, and yellow or green represent intermediate tissue elasticity on sonoelastography. The dermis consisting of connective tissue, nerves, blood, and lymphatic vessels, gland, mast cells, fibroblasts, histiocytes [18] represents blue color, showing hard nature, namely, low elasticity. While the subcutaneous fat tissue shows red color, consistent with soft nature, suggesting high elasticity, and reflecting contents of adipose cells [18] on RTE. Yang et al. [19] also studied a reference range of normal skin elasticity evaluated by share wave elastography (SWE) in healthy subjects to interpret results in patients with systemic scleroderma. They described that skin elastic modules values estimated by SWE in healthy individuals are affected mainly by the skin site, sex, and age [19]. Additionally, skin elasticity was higher in men than in women at each site and was increased in subjects aged 20 to 50 years than in the other age group at the finger [19].
Plantar Fascia on Real-Time Tissue Elastography
Sonoelastography study has showed a significantly softer of the plantar fascia in older healthy subjects than in the younger subjects [17,20]. It is suggested that plantar fascia is similar to tendon and the age-related alterations of elasticity may occur in plantar fascia [20]. Plantar fasciitis is a common cause of heel pain. Though the etiology of plantar fasciitis is multifactorial, mechanical overload and degeneration have been considered as the main factors. Grayscale US shows the fascial thickening and hypoechoic echotexture of planar fascia in patients with plantar fasciitis. Softer plantar fascias were shown in patients with plantar fasciitis than in healthy subjects [17,20]. Sconfienza et al. [21] also described that Real-Time Sonoelastography revealed elasticity alterations in symptomatic plantar fasciitis in whom gray-scale US features were inconclusive.
Soft-tissue Masses in Hand Region
Soft-tissue masses have the occurrence forms of characteristic locations. The characteristic location and relation to surrounding structure are very important in the accurate diagnosis [22]. In the cystic tumors, ganglion cyst is the most common masses of the hand and wrist regions. It is mutin filled with a fibrous capsule and occurs in association with a joint or tendon sheath [22]. With regard to the solid tumors, palmar fibromatosis, giant cell tumor of the tendon sheath (GCTTS), and peripheral nerve sheath tumor tend to occur in wrist or hand, while plantar fibromatosis, peripheral nerve sheath tumor and Morton neuroma have tendency of occurrence in ankle or foot region [22]. Superficial fibromatoses in adults (palmar and plantar) and children (calcifying aponeurotic fibroma, lipofibromatosis, and inclusion body fibromatosis) are often small slow-growing lesions and their diagnosis have been suggested by location [23]. In addition to these diseases, fibroma of the tendon sheath (FTS) mostly occurs in fingers, hands, and wrists locations [24].
Palmar Fibromatosis (Dupuytren’s Disease)
Clinical Appearances
Dupuytren’s disease is a routine for the dermatologist, because patient with subcutaneous nodule may firstly visit a dermatologist. Kelenjian et al. also suggested that superficial fibromatoses relevant to dermatology include palmar, plantar, and penile fibromatosis, knuckle pads, pachydermodactyly and infantile digital fibromatosis [25]. Palmar fibromatosis was described by the French physician Dupuytren and is referred to as Dupuytren disease [23,26,27]. Dupuytren’s disease or palmar fibromatosis characterized by abnormal proliferation of fibroblasts in the palmar fascia and leads to contractures of the fingers. This entity appearing subcutaneous nodule without pain, commonly occur approximately 20% of the people older than 55 years [3]. The fourth and fifth digits may be most commonly affected by palmar fibromatosis. This disease has other types of fibromatosis such as plantar fibromatosis, Peyrone disease, and knuckle pads [23]. Although the etiology of this disease is completely unknown, this entity has the relationship with trauma, microvascular injury, immunologic factors and genetic factors. In addition, palmar fibromatosis is associated with diabetes mellitus, epilepsy, alcoholism, and keloids [23]. Patel et al. [28] suggested that patients with the palmoplantar phenotype of psoriasis were more likely to develop Dupuytren’s disease. With regard to the treatment, Eberlein et al. described that therapy of Dupuytren’s disease in early stage is necessary and radiation therapy shows an effective and safe threatment [29].
US and MRI Appearances
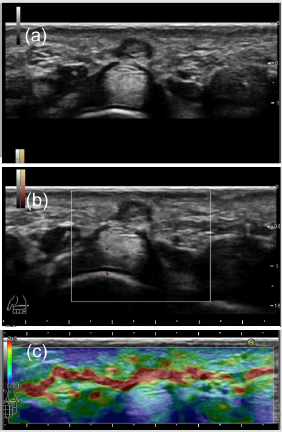
There have been a few reports on US study [23, 30]. Hypoechoic nodules in the palmar subcutaneous tissues, superficial to the flexor tendons [23] or mixed echogenicity [30] were shown on gray-scale US. With respect to color Doppler US findings, the nodule shows hypervascular lesion [23]. MRI appearance show multiple nodular or cordlike, superficial soft-tissue masses arising from the proximal palmar aponeurosis [23]. The corresponding histologic analysis demonstrated that the lesions of low signal intensity with all pulse sequences contained hypocellularity and abundant dense collagen. While, the lesion of intermediate signal intensity corresponds to the more cellular or mixed, with less abundant collagen on both T1- and T2-weighted images. Recently, Morris et al. [3] noted that the lesions were located directly superficial to the flexor tendons with an epicenter at the distal metacarpal, most commonly the fourth digit region. They described that the lesions show hypoechoic nodule and non-compressible condition on gray-scale US. With respect to color Doppler US features, vascular flow was not detected in most cases [3]. Figure 1 shows palmar fibromatosis pathologically diagnosed at te distal metacarpal (MC) region of the fouth digit in the right hand in a 60-year-old man. The small hypoechoic nodule with partially hypoechoic layer was depicted at the level of the distal metacarpal (MC) region on gray-scale US. The size of this nodule is 3 mm in depth and 4 mm in width on short axis view [Figure 1a]. There are no blood flow signals in the nodule on power Doppler US [Figure 1b]. As far as I know, bibliographically, there have been no report of palmar fibromatosis on RTE fearures. Palmar fibromatosis shows blue color with a green color, showing moderately hard nature, moderately low elasticity compared to the surrounding tissue on RTE [Figure 1c]. Flexor tendon represents blue color, suggesting hard nature. Subcutaneous nodule showed low signal intensity on both T1 and T2- weighted images in this case (image not shown).
Figure 1: Palmar fibromatosis pathologically diagnosed at the fouth digit of the right hand in a 60-year-old man.
(a): The hypoechoic nodule with partially hypoechoic layer was depicted at the level of the distal metacarpal region in the
subcutaneous layer on gray-scale US (short axis view).
(b): There are no blood flow signals on power Doppler US.
(c): Palmar fibromatosis shows blue color with a green color, suggesting moderately hard nature, moderately low elasticity on
Real-Time Tissue Elastography.
Pathological Appearances
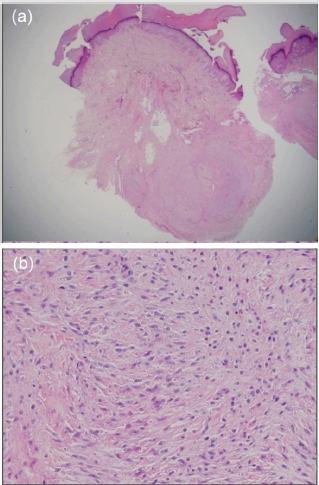
Palmar fibromatosis is a very small nodule and it shows a uniform fibroblastic-myofibroblastic proliferation of spindleshaped cells with variably prominent vascularity. The degree of cellularity depends on the age of the lesion. Younger lesions appear as hypercellularity, whereas older ones reveal much less cellularity with more prominent collagen. Moderate mitotic activity without malignancy is shown [25]. Figure 2 represents palmar fibromatosis pathologically diagnosed at the distal metacarpal (MC) region of the fouth digit in the right hand in a 60-year-old man. Histopathologically, the lesion is characterized by a uniform proliferation of spindle-shaped cells with collagen contents in the subcutaneous fatty tissue [Figure 2a]. A uniform proliferation of spindle-shaped cells with collagen contents in the subcutaneous fatty tissue was also observed in high magnification view [Figure 2b]. Genetically, Liu et al. described that differentially expressed genes (DEGs) including NTF3, FBN2, NR4A1, FGF22, BDNF, PRKCE, ZNF516, SLCO2A1, C10orf10 and KLHDC7A, may play important roles for disease development and serve as candidate molecular targets for the therapy in Dupuytren’s contracture [31].
Figure 2: Palmar fibromatosis pathologically diagnosed at the fouth digit of the right hand in a 60-year-old man. (a): Histopathological finding of the lesion is characterized by a uniform proliferation of spindle-shaped cells with collagen components in the subcutaneous fatty tissue (hematoxylin and eosin, original magnification X10). (b): A uniform proliferation of spindle-shaped cells with collagen components in the subcutaneous fatty tissue was observed (hematoxylin and eosin, original magnification X200).
Plantar Fibromatosis (Ledderhose Disease) as a Concomitant Disease of Palmar Fibromatosis
Clinical Appearances
It has been reported that concomitant palmar fibromatosis occurs in 10%-65% of patient with plantar disease. Plantar fibromatosis is a benign fibroproliferative disorder of the plantar fascia described by Georg Ledderhose [4]. This entity appears as a palapable mass in the midfoot or forefoot region. Plantar fibromatosis was shown as a firm subcutaneous nodule. Similar to palmar fibromatosis, plantar fibromatosis is also associated with diabetes mellitus, epilepsy, keloid and alcoholism. Pathologically, the features of plantar fibromatosis is identical to that of palmar fibromatosis. Family studies have revealed that there is a genetic component with manifestation of palmar and plantar fibromatosis. Zaragoza et al. described the novel co-occurrence of palmar and plantar fibromatosis and LMNA-related cardiac disease in a family with a previously described nonsense mutation, LMNA c.736C>T (p. Gln246Stop) [32]. A novel, heterozygous missense variant (c.230T>C, p. Val77Ala) in the Asteroid Homolog 1 (ASTE1) gene has been revealed as a potential risk factor in fibrotic disease [32]. They suggest that ASTE1 may contribute to the increased risk for palmar/plantar fibromatosis in patient with Lamin A/C haploinsufficiency [32].
US and MRI Apearances
Plantar lesions appear similar to palmar type on gray-scale US. The well-defined or fusiform lesion with hypoechoic or mixed echogenic were depicted in the subcutaneous layer superficial to the plantar aponeurosis [23]. Color Doppler US demonstrate hypervascularity lesion in most cases [23]. Cohen et al. [4] described a novel appearance termed the “comb sign” on grayscale US. They also mentioned that plantar fibromatosis shows mixed echogenicity and posterior echo enhancement on grayscale US. Approximately half cases show internal vascularity in the nodule on color Doppler US. With regard to MRI features, the lesion demonstrates predominantly low to intermediate signal intensity on T1-weighted and T2-weighted images. It is indicated that the termed “fascial tail sign “along the plantar aponeurosis are common and seen on enhanced MRI image [23,26].
Giant Cell Tumor of the Tendon Sheath
As similar to palmar fibromatosis, giant cell tumors of the tendon sheath (GCTTS) commonly occurs in the volar aspect of the finger. Tthereby, it is important to distinguish palmar fibromatosis from GCTTS. GCTTS is a type of benign soft tissue tumor that was first described by Chassaignac [33, 34]. The World Health Organization distinguishes between two types, namely localized and diffuse forms of of GCTTS originating from the tendon and the synovium [34, 35]. GCTTS is also termed tenosynovial giant cell tumor. Clinically, the localized type occurs most frequently in the hand, especially in the volar aspect of the first three fingers [36]. The author suggests that clinical manifestation of palmar fibromatosis appears at the fouth or fifth digits, while GCTTS occurs at the first three digits. With dynamic information, GCTTS do not move with flexion and extension of the digits. Because they arise from the tendon sheath and not the tendon [22]. Histologic examination of GCTTS classically shows a lobulated, circumscribed, cellular tumor [37]. GCTTS consists of three cell types, namely, multinucleated osteoclastlike giant cells, round or polygonal mononuclear cells and foam cells or histiocytes [38]. Wan et al. [39] reviewed imaging of GCTTS and suggested that US showed a soft mass related to the tendon sheath on gray-cale US. They represent hypervascular lesion on color or power Doppler US. With regard to MRI imaging, the lesion typically shows low to intermediate signal on T1- and T2-weighted spin-echo sequencies due to the hemosiderin [39]. Middleton et al. [40] described that this entity shows solid homogeneous hypoechoic tumors associated with the flexor tendons of the fingers on gray-scale US. The lesions represent internal vascularity on color and power Doppler US. While, Wang et al. [41] described that this disease appears as hypoechoc nodule on gray-scale US. Substantial flow could be seen in most lesions [41]. Ansari et al. mentioned that a well-defined hypoechoic mass was depicted on gray-scale US [42]. It is suggested that GCTTS typically exhibits a low signal intensity on T1 and T2 weighted images due to the presence of hemosiderin [34]. On MRI appearances, GCTTS appears as a focal mass, often adjacent to or surrounding a tendon. They usually show intermediate or low signal intensity on T1 and T2 weighted images and are well enhanced with contrast medium administration [43-45].
In Summary
It is putative that palmar fibromatosis shows hypoechoic nodule which is located in the subcutaneous tissue on gray-scale US. It is plausible that plantar fibromatosis shows mixed echogenicity on gray-scale US and tends to represents vascular flow on color Doppler US. It is suggested that GCTTS show hypoechoic nodule with vascularity on US study and represent low signal intensity on T1 and T2 weighted MR images due to the hemosiderin.
Conclusion
a) The author suggests that Dupuytren’s disease occurs at the
distal metacarpal region of the fouth digit in the subcutaneous
tissue of the hand.
b) It is strongly suggested that gray-scale US provides the
association of the lesion and the flexor tendon and that Real-
Time Tissue Elastograhy images reflect the elasticity alteration
in palmar fibromatosis.
c) Clinically and genetically, it may be important that
Ledderhose disease shoud be considered as a concomitant
disease of Dupuytren’s disease.
Acknowledgement
The author appreciates Dr. Minoru Oishi and Dr. Ken Hayashi for their kind support.
Conflict of Interest
There are no conflicts of interest.
Financial Support and Sponsorship
None
References
- Wortsman X, Alfageme F, Roustan G, Arias-Santiago S, Martorell A, et al. (2016) Guidelines for performing dermatologic ultrasound examinations by the DERMUS group. J Ultrasound Med 35: 577-580.
- Wortsman X, Alfageme F, Roustan G, Arias-Santiago S, Martorell A, et al. (2016) Proposal for an assessment training program in dermatologic ultrasound by the DERMUS group. J Ultrasound Med 35: 2305-2309.
- Morris G, Jacobson JA, KalumeBrigido M, Gaetke-Udager K, et al. (2019) Ultrasound features of palmar fibromatosis or Dupuytren contracture. J Ultrasound Med 38: 387-392.
- Cohen BE, Murthy NS, Mckenzie GA (2018) Ultrasonography of plantar fibromatosis: updated case series, review of the literature, and a novel descriptive appearance termed the “comb sign”. J Ultrasound Med 37: 2725-2731.
- Fujioka K, Fujioka A, Eto H, Suzuki K, Sanuki E, et al. (2006) Nodular fascitis in the thigh followed up using ultrasonography. J Med Ultrason 33: 49-53.
- Fujioka K, Fujioka A,Oishi M, Eto H, Tajima S, et al. (2017) Ultrasonography findings of intradermal nodular fasciitis; a rare case report and review of the literature. ClinExpDermatol 42: 335-336.
- Fujioka K, Fujioka A, Tajima S, Oishi M, Hayashi K, et al. (2018) Characteristic power Doppler sonographic imaging of nodular fasciitis from dermatological perspective: another case and review of three cases. J Clin Case Rep 8: 1165.
- Fujioka K, Fujioka A, Oishi M, Hayashi K, Nakayama T (2018) High-resolution ultrasound imaging for angioleiomyoma: a painful and vascularized superficial tumor. Biomed J Sci& Tec Res 9(5).
- Fujioka K (2018) Presentations of clinical, ultrasonographic and pathological features of nodular fasciitis from an established cytogenetic viewpoint: review of the case series. J Carcinog Mutagen 9: 326.
- Fujioka K (2018) Usefulness of comprehensive high-resolution ultrasound imaging in dermatologic field: epidermal cyst. Biomed J Sci& Tec Res 12(1).
- Fujioka K (2019) Painful cutaneous and subcutaneous tumors accompanied with vascularized appearance using high-resolution ultrasound in dermatology: the acronym “ENGLAND” or “LEND AN EGG”. Biomed J Sci& Tec Res 12(4).
- Fujioka K (2019) Real- time Tissue elastography in epidermal cyst: along with enhanced MRI and elasticity features of peripheral rim. Biomed J Sci& Tec Res 16(5).
- Fujioka K (2019) A comparison between superficial and deep-seated lipomas on high-resolution ultrasonography: with RTE and MRI appearances. Biomed J Sci& Tec Res 19(2).
- Fujioka K (2019) Characteristic appearances of nodular fasciitis on high-resolution ultrasonography: with vasculature status from a lesion-seated perspective. Biomed J Sci& Tec Res 20(4).
- Fujioka K, Fujioka A, Oishi M, Masahiro Okada (2019) A new application in dermatological ultrasound. Biomed J Sci& Tec Res 22(5).
- Fujioka K, Fujioka A, Masahiro Okada (2019) Utility of high-resolution ultrasonography by using acoustic coupler in dermatology. Biomed J Sci& Tec Res 23(5).
- Klauser AS, Miyamoto H, BellimannWeiler R, Feuchtner GM, et al. (2014) Sonoelastography: musculoskeletal applications. Radiology 272: 622-633.
- Mandava A, Ravuri PR, Konathan R(2013) High-resolution ultrasound imaging of cutaneous lesions. Indian J Radiol Imaging 23:269-277.
- Yang Y, Wang L, Yan F, Xiang X, Tang Y, et al. (2018) Determination of normal skin elasticity by using real-time share wave elastography. J Ultrasound Med37 811): 2507-2516.
- Wu CH, Chang KV, Mio S, Chen WS, Wang TG (2011) Sonoelastography of the plantar fascia. Radiology 259: 502-507.
- Sconfienza LM, Slvestri E, Oriandi D, Fabbro E, Ferrero G, et al. (2013) Real-time Sonoeastography of the plantar fascia: comparison between patients with plantar fasciitis and healthy control subjects. Radiology 267: 195-200.
- Carra BJ, Bui-Mansfield LT, O’Brien SD, Chen DC(2014) Sonographic of musculoskeletal soft-tissue masses: techniques, pearls, and pitfalls. AJR 202: 1281-1290.
- Murphey MD, Ruble CM, Tyszko SM, Zbojiniewicz AM, Potter BK, et al. (2009) From the archives of the AFIP: musculoskeletal fibromatoses-radiologic-pathologic correlation. RadioGraphics 29: 2143-2173.
- Ma X, Xu J, Wang X, Wang C, ZhangYJ(2013) Toe deformity after pregnancy due to fibroma of tendon sheath: a case report and review of the literature. OrthopSurg 5: 225-228.
- Kelenjian S, Mattjie RA, Franz R, Biedermann T, Brockow K(2019) Clinical features and management of superficial fibromatoses. J DtschDermatolGes 17: 393-397.
- KransdorfMj, Murphy MD (2006) Benign fibrous and fibrohistocystic tumors. In: imaging of soft tissue tumors. Philadelphia, Pa: Lippincott Williams & Wilkins189-256.
- Weis SW (2008)(Edn,), Soft tissue tumors. St Louis, Mo: Mosby Elsevier, USA.
- Patel M, Freeman NR, Dhaliwal S, Wright N, daoud Y, et al. (2014) The prevalence of Dupuytren contractures in patients with psoriasis. ClinExpDermatol39: 894-899.
- Eberlein B, Biedermann T(2016) To remember: radiotherapy-a successful treatment for early Dupuytren’s disease. J EurAcadDermatolVenereol 30: 1694-1699.
- Molenkamp S, Van straalen RJM, Werker PMN, Broekstra DC(2019) Imaging for Dutuytren disease: a systematic review of the literature BMC Musculoskeletal Disorders 20: 224.
- Liu H, Yin W, Liu B, Liu Y, Guo B, et al. (2015) Screening of cabdidate genes in fibroblasts derived from patients with Dupuytren’s contracture using bioinformatics analysis. RheumatolInt 35: 1343-1350.
- Zaragoza MV, Nguyen CHH, widyastuti HP, McCarthy LA, Grosberg A (2017) Dupuytren’s and Ledderhose disease in a family with LMNA-related cardiomyopathy and a novel variant in the ASTE1 gene. Cells 6(4):Pii: E40
- Sharon W, WJ (2001) Enzinger and Weiss’s soft tissue tumors, 4th(Edn,),St Louis, Mosby pp: 1037-
- Wang C, Song R, Kuang PD, Wang LH, Zhang MM(2017) Giant cell tumor of the tendon sheath: magnetic resonance imaging findings in 38 patients. Oncology letters 13: 4459-4462.
- Fletcher CDM BJ. Hogendoorn P, Mertens F (2013) WHO Classification of tumors of soft tissue and bone. WHO
- Llauger J, Palmer J, Roson N, et al. (1999) Pigmented vilonodularssynovitis and giant cell tumors of the tendon sheath: radiologic and pathologic features. AJR Am J Roentgenol 172: 1087-1091.
- Farah RS, Holahan HM, Swick BL(2014) Giant cell tumor of the tendon sheath. Cutis 93: 181-182.
- Ushijima M,Hashimoto H, Tsuneyyoshi M, Enjoji M (1986) Giant cell tumor of the tendon sheath (nodular tenosynovitis9: a study of 207 cases to compare the large joint group with the common digit group. Cancer 57: 875-884.
- Wan JM, Magarelli N, Peh WC, GuglielmiG,Shek TW(2010) Imaging of giant cell tumour of the tendon sheath. (2010) Radiol Med 115: 141-151.
- Middieton(2004) Sonography of giant cell tumors. AJR :183.
- Wang Y, Tang J, Luoy(2007) The value of sonography in diagnosing giant cell tumors of the tendon sheath. J Ultrasound Med26: 1333-1340.
- Ansari SMM, Tambe SA, Nayak CS (2019) Giant cell tumor of tendon sheath in children. Indian J DermatolVenereolLeprol 85: 496-498.
- Lee SM, Ha DH, Kang SH, Kim SW, Chol YR (2019) Desmoplastic fibroblastoma mimicking tenosynovial giant cell tumor encasing a tendon of the foot. Skeletal Radiology48:637-641.
- Murphey MD, Rhee JH, Lewis RB, Fanburg-Smith JC, Flemming DF, et al. (2008). Pigmented villonodular synovitis: radiologic-pathologic correlation. RadioGraphics 28: 1493-1518.
- Plotkin B, Sampath SC, Sampath SC, Motamedi K(2016) MR imaging and US of the wrist tendons. Radiographics 36: 1688-1700.

 Review Article
Review Article