Abstract
Additive Manufacturing (AM) is one of the most significant in bio medical application to manufacture the implant for bone repairing applications. The implant has a cellular structure inside which is used in biomedical implant, that is used to allow our bone ingrowth. The implant material should be biodegradable, non-toxic, have similar biomechanical properties and have a porous. Due to this reason it is very significant important to design and produce implant with high accuracy. In this review, design cellular structure and AM technology of fabrication biomedical implant using Selective Laser Melting (SLM) process is detail reviewed. As per According to necessities of medical orthopedic applications, the choice of high quality AM technology printing technique and Ti6Al4V metal powder were surveyed. Aside from this biomedical -AM printing application, the biocompatible Ti6Al4V alloy will be broadly used in implant bone application, in addition to that used for scaffold in tissue engineering.
Keywords: EBM: Electron Beam Melting, SLM: Selective Laser Melting, AM: Additive Manufacturing
Introduction
In biomedical engineering, the medical orthopedic is a speedily developing multidisciplinary research area to rebuild implants and scaffolds[1] by using a biocompatible and biodegradable implant and scaffolding structure. As described early, it is very important to manufacture implants with high accuracy and precision. The implant has been manufactured using various AM methods such as SLM, Electron Beam Melting (EBM), In these methods, the smallest printable size is 50-200μm which is too small to be used for some biomedical scaffold or certain producing big in size on biomedical implant application [2].
Design of Cellular Structures
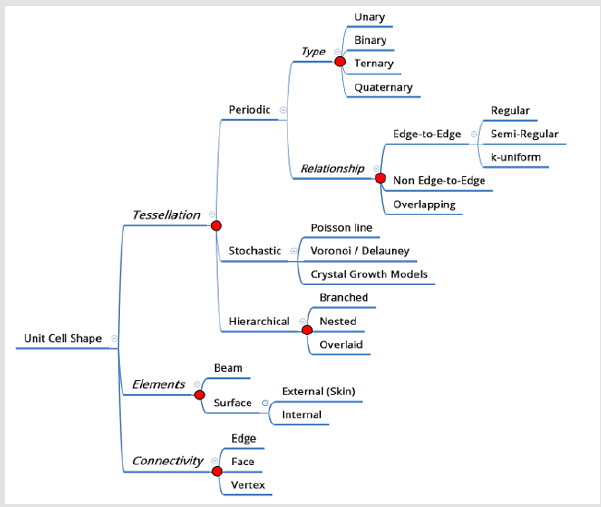
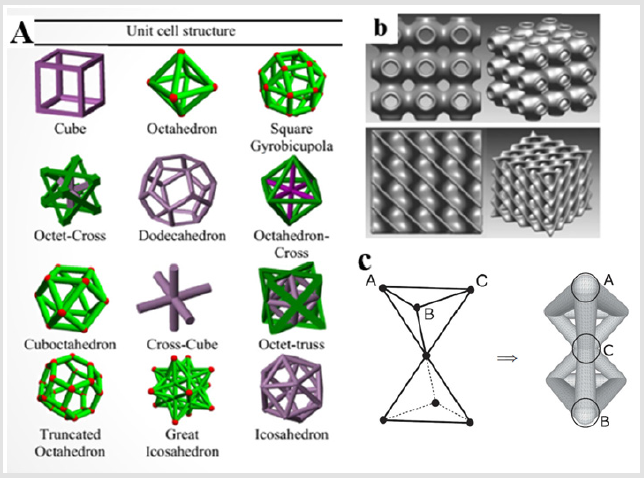
In biomedical orthopedics application, lattice structures are used as cellular implant that attempt to function of bone and mimic the structure [3]. The cellular structures can be used either asan implant or scaffold. In either case, the cellular implant ought to be designed to such an extent that its mechanical performance similar that of human bone, though considering different issues that optimized bone ingrowth. For instance, the mass transport and cell migration in our bone tissue can be controlled through well-designed ofpermeabilitycellular structure structures [4,5]. During the most recent two decades, a number ofesearchershave been investigated for designing cellular implant for scaffold which considered the properties of cellular implant such as biocompatibility, mechanical properties, bio-functionality and biodegradability of the cellular implant of scaffold [6-9].In this review, we focused on the design tools for cellular structure aimed for biomedical orthopedics application (Figure 1) the flow chat of design process. Generally, there are 3 types of methods used by engineers to design a suitable unit cell for a bio-medical application see in (Figure 2) examples design unit cells.
Figure 2: Methods of design of cellular structure
A. Manually generated structures,
B. Mathematical model ,
C. Tessellation method [8,9].
SLM Fabrication of Cellular Structures
Based on ASTM definition of AM is “technique of joining
materials to make parts by the fashion layer upon layer from STL
format rather than subtractive fabrication process.” As a new
advanced technology since the late 1980s, therefore, due to the
improvement of Additive Manufacturing innovation in biomedical
applications the researchers are become more and more inserting
mainly with fast growth of supercomputer technology. The
development of AM technology was dependent on the computerized
model by the guide of Computer Aided Design (CAD), which can
solidify the powder particles layer by layer, then 3D component
can be produced. The powder particles (Ti6AL4V) is usually used
for cellular biomedical implant. Therefore, Additive Manufacturing
technology, for example Selective Laser Melting (SLM) are generally
used to fabricate Ti-6Al-4V alloys cellular implants for orthopedic
application. The SLM technology the most suitable process for
manufacture periodic cellular implants based on the STL file [1].
Recently, numerous studied have been used SLM process
to produce cellular implant. Santorinaios, et al. investigated the
fabrication of open-cell structures with a humble geometry with
cross bracing and vertical struts. Brooks et al. designed and fabricated
periodic cellular structures with different struts diameter and unit
cell sizes using SLM process. McKown, et al.fabricated with cellular
metallic implant based on lattice topologies using SLM method.
The literature survey has discovered to a certain extent that SLM
process of Ti6AL4V can be suitable for the manufacture of custom
orthopaedic implants. The in-vivo tests results revealed that the
implant made by SLM were able to allow bone growth and that SLM
as an advanced fabrication technology is a promising additional to
conventional implant surface topographies. On the other hand, the
review has shown that cellular Ti6AL4V implant made by SLM are
biocompatible, in addition to that pore size and the porosity can
influence the biomedical (bone growth) and mechanical properties.
Bio-Compatible Ti6Al4V
Ti6Al4V alloys are extensively used as biomedical implant due to its many beneficial assets, such as, low stiffness, high specific strength-to-weight ratio, satisfactory biocompatibility, superior corrosion resistance, good fatigue resistance, and so on [10]. So, Ti6Al4V have been commonly used in biomedical application, such as oral repair, artificial joints, scaffold and hip implant [11-13]. The elastic modulus of Ti6Al4V is about 110 GPa, relatively low related with CoCr alloys and stainless steel, however very high relative to human bones (0.3–20GPa). The mismatch of stiffness between metallic implant and natural bone is a main problematic of Ti6Al4V implants in orthopedic application, that may cause stress shielding and our bone become inadequate for bearing loading. Consequently, the result of stress shielding is ultimate loosening of the implants because of the crack in our bone. Furthermore, some Ti6Al4V alloys will release toxic elements such as vanadium and aluminum, that are very hurtful to human nerves; those problems have restricted the development of Ti6Al4V in medical applications. To handle the higher stiffness of Ti6Al4V, cellular structures is progressively more developing as a technique to reduce high stiffness of titanium alloy and attain steady long-term fixation with full ingrowth bone tissue. A number of study have also investigated a number of novel titanium alloys with no harmfulness or toxicity, such as Ti6Al7Nb, which belongs toα+βtype titanium alloys; Ti13Nb13Zr, Ti12Mo6Zr, Ti30ZrMo, Ti35Nb5Ta7Zr [11], and Ti-24Nb4Zr7.9Sn (Ti-2448), that similar to the novel β-type Ti alloys, to overawed the capacity toxic effects of based Titanium alloys.
Conclusion
In spite of the fact that few AM technologies are used for biomedical applications, SLM process can print cellular implant biodegradable / biocompatible implant with higher resolution and high accuracy dimension. The mechanical and biological properties are the main concern while Ti6Al4V cellular implant fabricating using SLM process.Presently, cellular metallic implant manufactured by SLM, according to in vitro biological evaluation, are not achieved very clear conclusion, this is because of design cellular implant have a lot variable such as pore shape, pore size, porosity, interconnectivity and so on. This means that any change in one variables can affect the performance of the implant. Therefore, the benefit of using permeability as variable design could the combination of the effect of the aforementioned parameters (pore size, porosity etc.) this revealed that the higher permeability implant was suitable for bone growth.
References
- Abate KM, Nazir A, Chen JE, Jeng YJ (2019) Design, Optimization, and Evaluation of Additively Manufactured Vintiles Cellular Structure for Acetabular Cup Implant.
- Melchels FPW, Barradas AMC, Van Blitterswijk CA (2010) Effects of the architecture of tissue engineering scaffolds on cell seeding and culturing. Acta Biomater 6(11): 4208-4217.
- Butscher A, Bohner M, Hofmann S (2011) Structural and material approaches to bone tissue engineering in powder-based three-dimensional printing. Acta Biomater 7(3): 907-920.
- Hollister S (2006) Porous scaffold design for tissue engineering. Nat Mater 5: 590.
- Hutmacher DW (2000) Scaffolds in tissue engineering bone and cartilage. Biomaterials 21: 2529-2543.
- Dias MR, Guedes JM, Flanagan CL, ScottHollister, Paulo R Fernandes (2014) Optimization of scaffold design for bone tissue engineering: A computational and experimental study. Med Eng Phys 36(4): 448-457.
- Bhate D (2019) Four questions in cellular material design. Materials (Basel) 12.
- Yoo J, Medinafranco JL (2011) Discovery and Optimization of Inhibitors of DNA Methyltransferase as Novel Drugs for Cancer Therapy.
- Nguyen DS, Vignat F (2016) A method to generate lattice structure for Additive Manufacturing. IEEE Int Conf Ind Eng Eng Managpp.966-970.
- Niinomi M (2008) Mechanical biocompatibilities of titanium alloys for biomedical applications. J Mech Behav Biomed Mater (1): 30-42.
- Arabnejad S, Burnett Johnston R, Pura JA (2016) High-strength porous biomaterials for bone replacement: A strategy to assess the interplay between cell morphology, mechanical properties, bone ingrowth and manufacturing constraints. Acta Biomater 30: 345-356.
- Von Doernberg MC, Von Rechenberg B, Bohner M,GamalBaroud, JörgAuera, et al. (2006) In vivo behavior of calcium phosphate scaffolds with four different pore sizes. Biomaterials 27: 5186-5198.
- De Krijger J, Rans C, Van Hooreweder B (2017) Effects of applied stress ratio on the fatigue behavior of additively manufactured porous biomaterials under compressive loading. J Mech Behav Biomed Mater 70: 7-16.

 Mini Review
Mini Review