Abstract
Objective: The aim was to assess the relationship between morning fractional exhaled NO (FeNO) and nocturnal oxygen desaturation in children. Design: prospective study. Setting: The study was conducted from January 2012 to January 2018, in the division of pediatric at tertiary-care hospital.
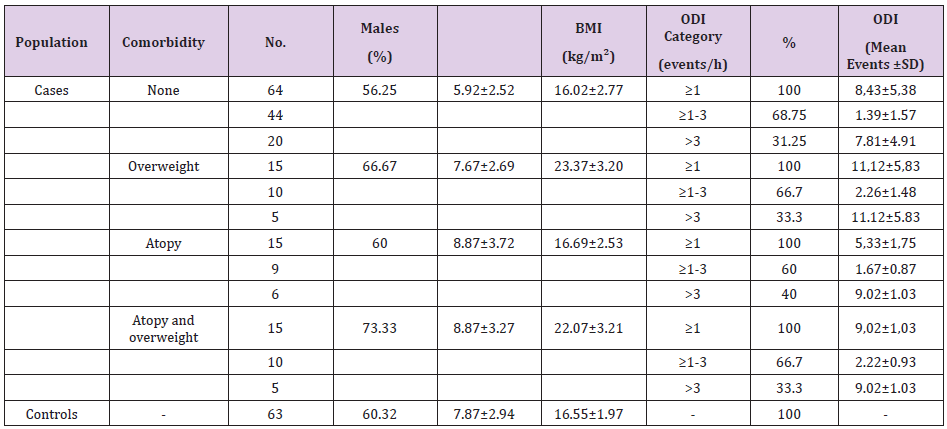
Patients: Of the total of 112 children initially enrolled, 109 (case children; 65.1% males; age 7.0± 3.1 years) met the criteria of the oxygen desaturation index (≥ 1 desaturations measured by pulse oximetry). An additional 63 healthy children (controls, 60.3% males; age 7.9±2.9 years) were enrolled as external controls. Children underwent pulse oximetry (172 subjects), were assessed for FeNO and for inhalant allergens (skin prick test). Comorbidities were allergy and obesity. Methods: Nocturnal at-home oximetry and morning FeNO levels.
Results: FeNO levels was comparable between case children without comorbidities and controls (p = 0.346). FeNO levels were much higher in case children with atopy compared to controls (p=0.0011) and in those with atopy and overweight compared to controls (p<0.0001) while FeNO levels increased in case children overweight compared to controls (p=0.0311).
Conclusion: Atopy was the main determinant of elevated FeNO levels in children with nocturnal oxygen desaturation. A novel finding is that the FeNO levels were significantly increased in obese children as compared to controls. Further studies are needed to confirm our findings and to compare FeNO levels in obese children with or without recurrent desaturation events during sleep.
Keywords: Allergy; Obesity; Pulse Oximetry; Sleep; Fractional Exhaled Nitric Oxide
Abbreviations: AHI: Apnea-Hypopnea- Index; FeNO: Fractional Exhaled Nitric Oxide; ODI: Oxygen Desaturation Index; OSAS: Obstructive Sleep Apnea Syndrome; TLC: Total Lung Capacity
Introduction
Between 1% and 5% of children suffer from intermittent upper airway obstruction during sleep [1,2]. The mean number of desaturation events per hour during sleep (no./hour) is used to calculate the Oxygen Desaturation Index (ODI), which is linked to the magnitude of Obstructive Sleep Apnea (OSA). OSA leads to a disruption of ventilation and intermittent low blood oxygenation, resulting in systemic airway inflammation and increased oxidative stress [3-5], with potential risk of cardiovascular disease, hypertension, neuropsychiatric and developmental disorders [1-3]. ODI during sleep can be measured simply using an at-home pulse oximeter. An ODI >3% measured by nocturnal pulse oximetry will have a 5% error to predict the polysomnographic Apnea- Hypopnea-Index (AHI) [6]. Nocturnal oximetry is useful in settings with limited resources because it predicts sleep-disordered breathing severity [7]. In this way, the ODI can be easily calculated to predict Obstructive Sleep Apnea (OSA) severity [8,9].
Among other cells in the body, the bronchial epithelium produces Nitric Oxide (NO) [10]. NO regulates endotheliumdependent vasodilation of the airways [6]. A biological mediator, it is present in exhaled breath [11]. Fractional exhaled NO (FeNO) is a non-invasive marker of airway inflammation [12]; it is easy to measure and stress-free for the patient [13,14]. A recent metaanalysis reported that FeNO is significantly increased in patients with Obstructive Sleep Apnea Syndrome (OSAS) [15]. To our knowledge, no data are available regarding the relationship between morning FeNO and intermittent nocturnal hypoxic events assessed by at-home pulse oximetry. To fill this gap, we measured morning FeNO levels in children with comorbid atopy and overweight and various degrees of ODI severity during sleep.
Methods
Subjects
Between January 2016 and June 2018, 112 children attending the sleep clinic, University of Insubria, Varese, Italy, were enrolled. The parents reported that their children suffered from nocturnal and daytime symptoms that suggested sleep-disordered breathing [16]. Following at-home sleep oximetry [13,14,17], 109 children (case children; 65.14% males) met our enrolment criteria for the oxygen desaturation index (ODI ≥ 1), while 3 did not (ODI<1) and were excluded. An additional 63 children recruited from the pediatric emergency department were enrolled as controls (males 60.3%). Total numbers of children enrolled and subsequently measured were 172. Written consent was obtained from the parents or legal guardians. None of these children had night-time signs or symptoms of OSA, allergic symptoms or chronic or recurrent respiratory disease or other serious diseases. Physical examination revealed maximum grade 2 tonsils, no ogival palate or dental malocclusion, laryngomalacia, neuromuscular disorders, genetic or craniofacial anomalies. After assessment of general health, weight and height were measured and the Body-Mass Index (BMI, weight in kilograms divided by height in meters squared) was calculated.
At-Home Oximetry
The case children underwent at-home pulse oximetry [1,18]. The parents were instructed to turn the device on when the child fell asleep and turn it off when he/she woke up. A sleep diary was provided so that the parents or the child could record nocturnal awakenings. The ODI was calculated with a portable pulse oximeter (Nonin® 2500 PalmSat Digital Pulse Oximeter and Nonin Vision 5.1 software, Nonin, Plymouth, MN, USA) that records and displays oxygen desaturation, heart rate, mean and minimum peripheral capillary oxygen saturation (SpO2), and automatic exclusion of artifact areas. Desaturation was defined as significant when the blood oxygen level dropped by ≥3% [19].
Exhaled Nitric Oxide (FeNO)
The mean FeNO in exhaled air in 172 children was measured according to an on-line single-breath technique using a HypAir- FeNO system (Médisoft, Sorinnes, Belgium - SensorMedics Italia srl) [18]. Age, weight and height, and morning exhaled FeNO were recorded. The children were told to refrain from food or drinks for at least 1 hour before the exam. The expiratory pressure was maintained between 5 and 20 cm H2O, which ensured closure of the palatal veil and prevented contamination from nasal NO. If two or three measurements were within 10% of each other, the mean was taken [19]. The children are asked to inhale from the mouthpiece and expand their lungs as much as possible to Total Lung Capacity (TLC) and then to exhale at a constant flow of 50 ml/s, until a plateau of NO values was displayed on the device monitor. During expiration, the plateau phase was > 4 s for children aged under 12 years and 6 s in children over 12 years. To facilitate control of inspiratory and expiratory flow, the child was prompted by watching an animation on the monitor that served as visual feedback: when the child blew through the device mouthpiece, a dolphin appeared on the monitor and jumped through a series of hoops during expiration [13]. The onboard software processed the data and created a pressure/time graph of the mean FeNO values.
Atopy Test
A skin prick test with commercially available extracts for the most common inhalant allergens was administered to all children [20]. The positive control is a cutaneous reaction to histamine (histamine 10 mg/ml in saline solution of glycerin) which causes a wheal of at least 3 mm. A sufficient or insufficient response to histamine indicated whether the child was taking antihistamine drugs or had experienced hyperresponsiveness. A drop of allergenic extract was introduced under the skin with a sterile lancet [21,22]. The drops were left to dry for 60 s, making sure they did not overlap or contaminate each other. The reaction was read within 15-20 min and compared with the histamine reaction. A positive response to an allergen was manifested as a small, round wheal usually surrounded by erythema.
Statistical Analysis
Statistical analysis was performed using IBM SPSS Statistics 22.0 for Windows (IBM Corp. Armonk, NY, USA). The Kolmogorov- Smirnov test was used to explore normal distribution of variables and the Mann-Whitney test to compare continuous measurements. Statistical significance was set at P <0.05.
Results
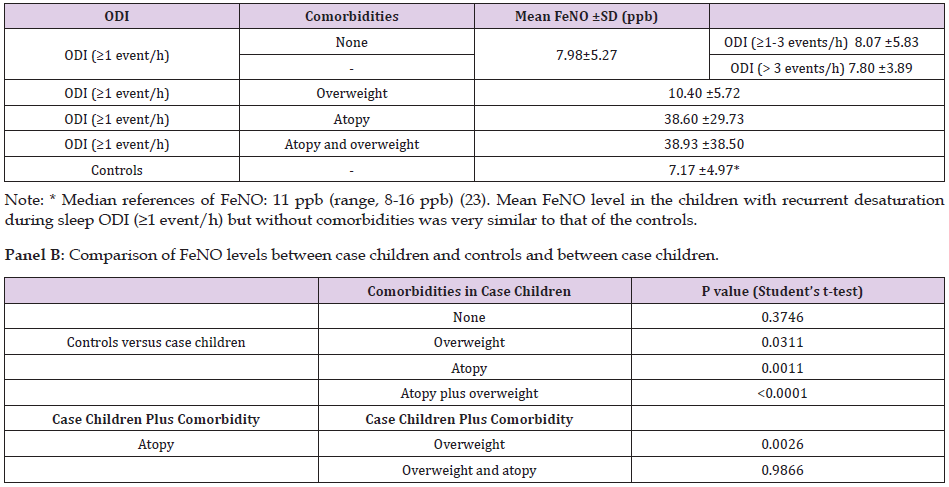
Table 1 presents the characteristics of the study population (172 subjects; 60.6% males, mean age 7.7±3.1 years). The case children (109 subjects; 65.1% males; age 7.0± 3.1 years) had a mean ODI of 7.81±4.91 events/hour. Table 2 (panel A) presents the FeNO levels. The case children were categorized as having a comorbidity or not (none, overweight, atopy, and overweight plus atopy). Table 2 (panel B) compares the case children without and with comorbidities (overweight, atopy, and overweight plus atopy) with the controls. FeNO was compares between case children without comorbidities and controls (p = 0.346). Children with desaturation during sleep had FeNO levels similar to the controls. FeNO was severely increased in the case children with atopy versus the controls (p=0.0011). FeNO was significanty increased in case children with atopy plus overweight compared to controls (p<0.0001). FeNO was also significantly increased in case children with overweight compared with controls (p=0.0311). FeNO in case children with atopy was significantly higher than in case children with overweight (p=0.0026) while FeNO was similar in case children with atopy and case children with overweight and atopy (p=0.9866).
Discussion
The main determinant of raised FeNO levels is atopy and asthma [23-25]. FeNO is an independent risk factor for asthma and allergic sensitization [26], and in school-age children it is used for clinical monitoring [27] in order to distinguish allergic rhinitis from nonallergic rhinitis [28]. Furthermore, FeNO is associated with various degrees of eosinophil inflammation of the airways [29]. In our study, FeNO levels in the case children without comorbidities were similar to those in the controls, whereas the level was significantly higher in case children with atopy than in controls. Accordingly, allergy alone, but not desaturation, may explain the increased FeNO produced by the upper airways. We did not investigate the numerous factors that affect the amount of eosinophils or differentiate the degree of exposure to allergen by the season in which the measurement was taken (a typical example is a major increase during the pollen season) or adherence to environmental prophylaxis and/or possible drug therapies (steroids). Consequently, there was no way to normalize the population of atopic children in relation to these variables and to measure the FeNO values in each child in the same condition.
OSA is characterized by repetitive collapse of the upper airways and intermittent desaturation events, which can trigger inflammation and oxidative stress. Intermittent desaturation is the main determinant of systemic inflammation in OSA children. Children with OSA are noted to have increased pro-inflammatory cytokine levels [30,31]. Studies involving children with OSA in which FeNO was measured as an inflammatory parameter have produced contradictory results [32-35]. Methodological discrepancies have been claimed as the reason, which is why these studies cannot be easily compared [32]. A mild increase in FeNO was reported in children with OSA [33-35], whereas in our study we found no difference in FeNO between children with higher ODI, those with mild (ODI ≥ 1-3 events/h) and moderate-severe (ODI > 3 events/h) and the controls. Summarizing, OSA alone was not the cause of increased FeNO levels. The mechanism of airway inflammation is different between asthma/atopy and OSA. Eosinophil count in the sputum of asthma patients is significantly elevated [36], whereas an increased neutrophil count and a low number of eosinophils, macrophages, and lymphocytes are found in the sputum of OSA children [37].
Obesity predisposes to a pro-inflammatory state via increased inflammatory mediators. Although, the results are contradictory. Ellulu et al., reported that the inflammatory state in obesity is characterized by decreased NO and elevated reactive oxygen species leading to oxidative stress [38], while cross-sectional studies showed that obesity alone is not associated with increased FeNO levels [26,39]. On the other hand, Verhulst et al., reported increased FeNO levels in children with overweight-associated OSA [40]. Our study showed a slight albeit significant increase in FeNO levels in obese children without atopy compared to controls. Yao et al., reported that excess weight influenced FeNO levels in atopic but not in non-atopic obese children [41]. In our study, FeNO was measured in 30 case children with atopy, 15 of which had normal weight and 15 were overweight. High FeNO levels were measured in case children, with atopy or with atopy plus obesity, whereas the obese children without atopy had a mild increase in FeNO compared to the controls. This difference could suggest that obesity per se is a significant factor contributing to raised FeNO levels. A possible explanation could come from the study done by Salerno et al., that reported low eosinophil count in the sputum of OSA children and a mild increase in FeNO levels [37]. As a final point wew would also underline that the online method yielded FeNO values with excellent reproducibility, even in very young children (3 or 4 years old), in which the animation was particularly stimulating as visual feedback for.
Conclusion
In children with recurrent desaturation during sleep the FeNO levels were comparable to those of the controls. Atopy and atopy plus obesity were associated with a significant increase in FeNO levels. Atopy was the major factor contributing to elevated FeNO levels. A novel finding coming from our study is that FeNO levels in obese children with recurrent desaturation during sleep are significantly increased compared to healthy controls. Further studies are needed to confirm our findings and to compare obese children with or without recurrent desaturation events during sleep in order to determine the potential influence of overweight in raising FeNO levels.
References
- Zaffanello M, Antoniazzi F, Tenero L, Nosetti L, Piazza M, et al. (2018) Sleep-disordered breathing in paediatric setting: existing and upcoming of the genetic disorders. Ann Transl Med 6(17): 343.
- Brunetti L, Rana S, Lospalluti ML, Pietrafesa A, Francavilla R, et al. (2001) Prevalence of obstructive sleep apnea syndrome in a cohort of 1,207 children of southern Italy. Chest 120(6): 1930-1935.
- Dehlink E, Tan HL (2016) Update on paediatric obstructive sleep apnoea. J Thor Dis 8(2): 224-235.
- Dos Santos C, Samuels M, Laverty A, Raywood E (2018) Comparison of oxygen desaturation index and apnoea-hypopnoea index for categorising OSA in children. Eur Respir J 52(S62): PA549.
- Chang L, Wu J, Cao L (2013) Combination of symptoms and oxygen desaturation index in predicting childhood obstructive sleep apnea. Int J Pediatr Otorhinolaryngol 77(3): 365-371.
- Tippu Z, Pengo M, Kosky C, Steier J (2014) Comparison of the polysomnographic apnoea-hypopnea-index with the oxygen desaturation index in patients with obstructive sleep apnoea. Eur Respir J 44(S58):
- Villa MP, Pietropaoli N, Supino MC, Vitelli O, Rabasco J, et al. (2015) Diagnosis of Pediatric Obstructive Sleep Apnea Syndrome in Settings with Limited Resources. JAMA otolaryngology- head neck surgery 141(11): 990-996.
- Temirbekov D, Güneş S, Yazıcı ZM, Sayın İ (2018) The Ignored Parameter in the Diagnosis of Obstructive Sleep Apnea Syndrome: The Oxygen Desaturation Index. Turkish Archives of Otorhinolaryngology 56(1): 1-6.
- Baena EM, Meneses PL, Camara JF, Calvo RE, Mazarro AP, et al. (2013) Nocturnal intermittent hypoxemia: utility for the evaluation of severity sleep apnea and cardiovascular comorbidity. Sleep Medicine 14: e67.
- Frank TL, Adisesh A, Pickering AC, Morrison JF, Wright T, et al. (1998) Relationship between exhaled nitric oxide and childhood asthma. Am J Respir Crit Care Med 158(4): 1032-1036.
- Ferrante G, Malizia V, Antona R, Corsello G, Glutta S (2013) The value of FeNO measurement in childhood asthma: uncertainties and persective. Multidiscip Respir Med 8(1): 50.
- Munakata M (2012) Exhaled nitric oxide (FeNO) as a non-invasive marker of airway inflammation Allergology international: official journal of the Japanese Society of Allergology. Allergol Int 61(3): 365-372.
- Pavone M, Cutrera R, Verrillo E, Salerno T, Soldini S, et al. (2 013) Night-to-night consistency of at-home nocturnal pulse oximetry testing for obstructive sleep apnea in children. Pediatr Pulmonol 48(8): 754-760.
- Mason DG, Iyer K, Terrill PI, Wilson SJ, Suresh S (2010) Pediatric obstructive sleep apnea assessment using pulse oximetry and dual RIP bands. Conference proceedings: Annual International Conference of the IEEE Engineering in Medicine and Biology Society IEEE Engineering in Medicine and Biology Society Annual Conference. Conf Proc IEEE Eng Med Biol Soc 2010: 6154-6157.
- Zhang D, Luo J, Qiao Y, Xiao Y, Huang R, et al. (2017) Measurement of exhaled nitric oxide concentration in patients with obstructive sleep apnea: A meta-analysis. Medicine 96(12): e6429.
- Joosten KF, Larramona H, Miano S, Van Waardenburg D, Kaditis AG, et al. (2017) How do we recognize the child with OSAS? Pediatr Pulmonol 52(2): 260-271.
- Kirk V, Baughn J, DAndrea L, Friedman N, Galion A, et al. (2017) American Academy of Sleep Medicine Position Paper for the Use of a Home Sleep Apnea Test for the Diagnosis of OSA in Children. J Clin Sleep Med 13(10): 1199-1
- Zaffanello M, Piacentini G, Pietrobelli A, Fava C, Lippi G, et al. (2017) Ambulatory clinical parameters and sleep respiratory events in a group of obese children unselected for respiratory problems. World J Pediatr 13(6): 577-583.
- Nigro CA, Dibur E, Rhodius E (2011) Pulse oximetry for the detection of obstructive sleep apnea syndrome: can the memory capacity of oxygen saturation influence their diagnostic accuracy? Sleep Disorders 2011: 427028.
- Heinzerling L, Mari A, Bergmann KC, Bresciani M, Burbach G, et al. (2013) The skin prick test - European standards. Clinical and Translational Allergy 3(1): 3.
- Dreborg S, Belin L, Eriksson NE, Grimmer O, Kunkel G, et al. (1987) Results of biological standardization with standardized allergen preparations. Allergy 42(2): 109-116.
- (1993) Position paper: Allergen standardization and skin tests. The European Academy of Allergology and Clinical Immunology. Allergy 48(14S): 48-82.
- Silva R, Cruz L, Vieira T, Leblanc A, Ferreira A, et al. (2010) Prevalence of aeroallergen sensitization and increased exhaled nitric oxide values in schoolchildren of different socioeconomic status. J Invest Aller Clin Immunol 20(3): 210-213.
- Rao DR, Phipatanakul W (2016) An Overview of Fractional Exhaled Nitric Oxide and Children with Asthma. Expert Rev Clin Immunol 12(5): 521-530.
- Rickard K, Jain N, MacDonald Berko M (2018) Measurement of FeNO with a portable, electrochemical analyzer using a 6-second exhalation time in 7-10-year-old children with asthma: comparison to a 10-second exhalation. J Asthma 19: 1282-1287.
- Cibella F, Cuttitta G, La Grutta S, Melis MR, Bucchieri S, et al. (2011) A cross-sectional study assessing the relationship between BMI, asthma, atopy, and eNO among schoolchildren. Ann Aller Asthma Immunol 107(4): 330-336.
- Pijnenburg MW (2019) The Role of FeNO in Predicting Asthma. Frontiers Pediatr 7: 41.
- Liu D, Huang Z, Huang Y, Yi X, Chen X (2015) Measurement of nasal and fractional exhaled nitric oxide in children with upper airway inflammatory disease: Preliminary results. Int J Pediatr Otorhinolaryngo l 79(12): 2308-23
- Verini M, Consilvio NP, Di Pillo S, Cingolani A, Spagnuolo C, et al. (2016) FeNO as a Marker of Airways Inflammation: The Possible Implications in Childhood Asthma Management. J Allergy 2010: 6914-6925.
- Huang YS, Guilleminault C, Hwang FM, Cheng C, Lin CH, et al. (2016) Inflammatory cytokines in pediatric obstructive sleep apnea. Medicine 95(41): e4944.
- Kheirandish Gozal L, Gozal D (2019) Obstructive Sleep Apnea and Inflammation: Proof of Concept Based on Two Illustrative Cytokines. Int J Mol Sci 20(3): E459.
- Bikov A, Losonczy G, Kunos L (2017) Role of lung volume and airway inflammation in obstructive sleep apnea. Resp Invest 55(6): 326-333.
- Bikov A, Hull JH, Kunos L (2016) Exhaled breath analysis, a simple tool to study the pathophysiology of obstructive sleep apnoea. Sleep Med Rev 27: 1-8.
- Barreto M, Montuschi P, Evangelisti M, Bonafoni S, Cecili M, et al. Comparison of two exhaled biomarkers in children with and without sleep disordered breathing. Sleep Med 45: 83-88.
- Duarte RLM, Rabahi MF, Oliveira ESA TS, Magalhaes da Silveira FJ, Mello FCQ, et al. (2019) Fractional Exhaled Nitric Oxide Measurements and Screening of Obstructive Sleep Apnea in a Sleep-Laboratory Setting: A Cross-Sectional Study. Lung 197(2): 131-137.
- Koc Gunel S, Schubert R, Zielen S, Rosewich M (2018) Cell distribution and cytokine levels in induced sputum from healthy subjects and patients with asthma after using different nebulizer techniques. BMC Pul Med 18(1):
- Salerno FG, Carpagnano E, Guido P, Bonsignore MR, Roberti A, et al. (2004) Airway inflammation in patients affected by obstructive sleep apnea syndrome. Respir Medicine 98(1): 25-28.
- Ellulu MS, Patimah I, Khazaai H, Rahmat A, Abed Y (2017) Obesity and inflammation: the linking mechanism and the complications. Arch Med Sci: AMS 13(4): 851-863.
- Santamaria F, Montella S, De Stefano S, Sperli F, Barbarano F, et al. (2007) Asthma, atopy and airway inflammation in obese children. J Allergy Clin Immunol 120(4): 965-967.
- Verhulst SL, Aerts L, Jacobs S, Schrauwen N, Haentjens D, et al. (2008) Sleep-disordered breathing, obesity, and airway inflammation in children and adolescents. Chest 134(6): 1169-1175.
- Yao TC, Tsai HJ, Chang SW, Chung RH, Hsu JY, et al. (2017) Obesity disproportionately impacts lung volumes, airflow and exhaled nitric oxide in children. PLoS One 12(4):

 Research Article
Research Article