Abstract
Medicinal plants have been used from prehistoric times as first source of health care to fight infectious and non infectious diseases. Most of these medicinal plants may have scientific evidence to be considered in general practice. Various parts of Bauhinia acuminata is employed in the treatment of glandular swelling, skin diseases and ulcer etc. The aim of this work was to investigate the antioxidant capacities and anti-inflammatory activities of the methanol extract of stem bark of B. acuminata and its different fractions. Crude Methanol Extract (CME) of stems and its various fractions such as Chloroform (CHF), Ethyl Acetate (EF), and Aqueous (AQF) were subjected to assay for antioxidant activity using various methods of assay like 2, 2-Di Phenyl Picryl Hydrazyl (DPPH), hydrogen peroxide and hydroxyl radical-scavenging activity. Total phenolics and flavonoids were determined by Folin-Ciocalteu and colorimetric methods. Anti-inflammatory activity was evaluated by carrageenan and formalin induced hind paw edema model.
The results showed that all the fractions and crude methanol extract were found to have variable levels of antioxidant properties in all antioxidant assays but Ethyl Acetate Fraction (EAF) had highest antioxidant activity based on DPPH (IC50=18.89 μg/ml) and hydrogen peroxide (42.78%) radical scavenging capacity. Furthermore, the stem bark contains phytochemicals like flavonoids and phenolics that are excellent free radical scavengers. The crude methanol extract and its fraction CHF significantly reduced the paw edema and WBC mobilization in rats. Among the sample CHF was found to significantly reduced the carragenan induced rat paw edema (94.29%; 250 mg/kg b.w.) as compared to standard drug diclofenac sodium. The results of the present study indicate that stem bark of B. acuminata has the potential to be a powerful antioxidant and also possess significant anti-inflammatory activity.
Keywords: Anti-oxidant; Stem Bark; Free Radical; Anti-inflammatory
Abbreviations: ROS: Reactive Oxygen Species; SOD: Superoxide Dismutase; GPx: Glutathione Peroxidase; CAT: Catalase; BHT: Butylated Hydroxytoluene, BHA: Butylated Hydroxyanisole; CME: Crude Methanol Extract; EAF: Ethyl Acetate Extract; CHF: Chloroform Extract; HF: Hexane Extract; AQF: Aqueous Extract; DPPH: 1, 1-Diphenyl-2-Picryl Hydrzyl
Introduction
Reactive Oxygen Species (ROS) results from oxidative stress due to the improper balance between ROS and antioxidant is an important factor in the pathogenesis of various diseases, such as cardiovascular disorders, neurological conditions, Parkinson’s disease, rheumatoid arthritis, and ageing [1]. Common free radical or Reactive Oxygen Species (ROS) are the hydroxyl radical, the superoxide anion radical, hydrogen peroxide, singlet oxygen, nitric oxide radical and hypochlorite radical. Antioxidants are agents and molecules that can reduce and limit oxidative damage to biological structures by scvenging the free radicals, neutralize the harmful free radicals before ROS can attack the cells and prevent damage living cells, proteins, enzymes and DNA etc. They break off the chain reactions via the removal of free radical and inhibit other oxidation reactions. Antioxidants synthesized in the body, including all antioxidant proteins and various small molecules, form a defense against oxidative stress, and their levels in the body cannot be altered by simple means.
On the other hand, the levels of antioxidant vitamins such as ascorbate, α-tocopherol, β-carotene and phytochemicals viz. polyphenols, flavonoids, can be increased easily by supplementation [2]. Enzymatic antioxidants include Superoxide Dismutase (SOD), Glutathione Peroxidase (GPx), Catalase (CAT), and micronutrients such as, selenium in glutathione, and copper and zinc in SOD as integral components of metallo enzymes [3]. Synthetic antioxidants such as Butylated Hydroxytoluene (BHT), Butylated Hydroxyanisole (BHA), tertiary butylated hydroquinone and gallic acid esters which are used in food industries now have restricted use in foods due to their prompt negative health effects [4]. So, there has been a growing interest in replacing commercial antioxidants with natural ingredients. Moreover, natural antioxidants are considered to be safe and environmentally friendly and they play a very important role in the physician’s therapeutic armamentarium. Currently, the use of plants and herbs with antioxidants is gaining importance.
The anti-oxidative activity has been confirmed contributing inflammatory preventions for its multiple functional roles. The production of oxidants is a typical event associated with aerobic metabolism. When oxygen is supplied in excess or its reduction is insufficient, reactive oxygen species or free radicals such as superoxide anions, hydroxyl radicals and hydrogen peroxide are generated [5]. Accumulation of the free radicals in body organs or tissues can cause oxidative damage to biomolecules and membranes of cell, eventually leading to many chronic diseases, such as inflammatory, cancer, diabetes, aging, cardiac dysfunction and other degenerative diseases [6]. In the last 50 years, antioxidant and anti-inflammatory activities of extracts from medicinal or food plants have been extensively investigated. Many pharmacological studies have shown that extracts of some antioxidant plant possess anti-inflammatory, anti-allergic, anti-tumor, anti-bacterial, antimutagenic and anti-viral activities to a greater or lesser extent.
Trouillas et al. investigated the antioxidant, anti-inflammatory and anti-proliferative properties of sixteen French herbal tea and found some herbs exhibited high antioxidant, anti-inflammatory and anti-proliferative activities [7]. Antioxidant activities in twenty traditional anti-inflammatory herbs extracts were investigated. The results suggested that the anti-inflammatory activities of these extracts could be explained, at least in part, by their antioxidant properties [8]. Free radicals liberated from phagocyte cells are important in inflammatory processes, because they are implicated in the activation of nuclear factor kB, which induces the transcription of inflammatory cytokines and cyclooxygenase-2 [9]. B. acuminata is a member of the Fabaceae family locally known as Shwet Kanchan. The plant is used in the treatment of common cold and cough [10]. The bark and leaves in a decoction can relieve biliousness and asthmatic attack [11]. The bark and leaves in a decoction also used to treat stones in the bladder, venereal diseases and leprosy.
The paste of the leaves applied on the throat is beneficial for throat troubles and to treat skin diseases. The root is boiled in oil and applied to burns [12]. Khammuang et al. [13] reported that crude extract of B. acuminata seeds showed strong antibacterial activity against various pathogenic bacteria. Anti-inflammatory activity of the leaves of this plant was reported by Mohammed et al. [14]. Therefore, B. acuminata is one of the famous plant based drugs used in traditional medicine. In the present study, we evaluated the antioxidant and anti-inflammatory effect of stem bark extract for the first time using different assay system to characterize its significance as a drug in traditional medicine.
Material and Methods
A. Plant Collection and Preparation of Extracts: The stem bark of B. acuminata was collected from University of Rajshahi, during August to September. The plant was identified by Botany Department, University of Rajshahi. The stem bark of the plant was thoroughly washed under running tap water and cut into very small pieces dried under shade, pulverized in a mechanical grinder and then extracted. Cold maceration technique was used for extraction. The coarse powders (800 gm) were soaked in methanol at room temperature. After 7days, the extract was filtered under vacuum through Whatman filter paper No.1. The filtrate was combined and methanol was evaporated under vacuum, using rotary evaporator at 45°C to yield greenish-colored extract. The quantity of extract obtained was 150 gm.
B. Fractionation: Fractionation was carried out by suspending the Crude Methanol Extract (CME) in 150 ml water separately and partitioning with different organic solvents n-hexane, chloroform and ethyl acetate in order of increasing polarity by using separating funnel. All fractions of the stem bark extracts were dried by evaporating respective solvent using rotary evaporator to give finally 26 gm of Ethyl Acetate Extract (EAF), 15 gm of Chloroform Extract (CHF), 9 gm of Hexane Extract (HF) and the remaining 75 gm of Aqueous Extract (AQF). All extracts were stored at 4°C until use.
C. Total Phenolic Content and Flavonoid Content: Total polyphenols were determined by the Folin-Ciocalteu method used by Mahfuza et al. [15] in samples. 40 μl of samples were mixed with 200 μl of Folin-Ciocalteu reagent, 600 μl of 20% sodium carbonate solution and 2.16 ml of distilled water. After 2 h, absorbance at 765 nm was read in the spectrophotometer. Total polyphenol content of extracts was expressed as mg of gallic acid equivalents (mg of GAE)/gm of extract through the calibration curve of gallic acid. Total flavonoid content was measured using a colorimetric assay [16]. An aliquot (500 μl) of diluted sample or standard solution of catechin was added to 150 μl of NaNO2 solution (7%), and mixed for 6 min with 2.5 ml distilled water, before adding 300 μl AlCl3 (10%). After 5 min, 0.5 ml of NaOH solution (1 M) was added. The final volume was adjusted to 5 ml, thoroughly mixed, and the absorbance of the mixture was determined at 510 nm. Total flavonoid content was expressed as mg of catechin equivalent (mg of CE)/gm of extract, through the calibration curve of catechin.
In vitro Antioxidant Activity
The radical scavenging effects of B. acuminata stem bark extract/fractions were evaluated against DPPH, hydrogen peroxide and hydroxyl radicals.
1, 1-Diphenyl-2-Picryl Hydrzyl (DPPH) Radical-Scavenging Assay: The radical scavenging capacity was determined by the modified DPPH method [17]. A methanol solution containing 0.008% DPPH was prepared. After adjusting the blank with methanol, an aliquot of extracts and standard concentration ranging from 10 to100 μg was added to 2.5 ml of this solution and volume was adjusted to 5 ml with distilled water. The decrease in absorbance at 517 nm was measured after 30 minutes. The free radical scavenging capacity was expressed as the concentration of the samples required to reduce the original amount of free radicals by 50% (IC50).
Hydrogen Peroxide Scavenging Activity Assay: Hydrogen peroxide scavenging activities of the extracts were estimated by replacement titration [18]. Aliquot of 1.0 ml of 0.1 mM H2O2 and 1.0 ml of various concentrations of extracts were mixed, followed by 2 drops of 3% ammonium molybdate, 10 ml of 2M H2SO4 and 7.0 ml of 1.8 M KI. The mixed solution was titrated with 5.09 mM Na2SO3 until yellow color disappeared. Percentage of scavenging of hydrogen peroxide was calculated as:
Where, 0 V was volume of Na2SO3 solution used to titrate the control sample in the presence of hydrogen peroxide (without extract),
1 V was the volume of Na2SO3 solution used in the presence of extract.
Hydroxyl Radical Scavenging Assay: The extent of hydroxyl radical scavenging from Fenton reaction was quantified using 2’-deoxyribose oxidative degradation as described by Halliwell et al. [19]. The reaction mixture containing FeCl3 (100 mM), EDTA (100 mM), H2O2 (1 mM) and 2-deoxy- D-ribose (2.8 mM) were mixed with or without the samples at various concentrations (10-250 μg) in 1 ml final reaction volume made with potassium phosphate buffer (20 mM, pH 7.4) and incubated for 1 hr at 37.50C. The mixture was heated at 95°C in water bath for 15 min followed by the addition of 1 ml each of TCA (2.8%) and TBA (1%). Finally the reaction mixture was cooled on ice and centrifuged at 5000 rpm for 15 min. Absorbance of supernatant was measured at 532 nm. All readings were corrected for any interference from brown color of the extract or antioxidant by including appropriate controls. The negative control without any antioxidant or samples was considered 100% deoxyribose oxidation. Ascorbic acid was taken as the positive control. The % hydroxyl radical scavenging activity was calculated by the following equation.
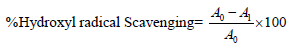
Evaluation of Anti-Inflammatory Activity
In vivo Evaluation of Anti-Inflammatory Activity: Carrageenan induced paw edema: Carrageenan-induced rat paw edema is used widely as a working model of inflammation in the search for new anti-inflammatory drug. The anti-inflammatory activity of extracts was evaluated by carrageenan-induced rat paw edema method [20]. Swiss albino rats (130–150g) of both sexes were used for the present study. After 12 h fasting, rats were divided into 6 groups of five each (n=5) prior to the day of experiment. Group l received normal saline (control group), Group II and III received 500 and 750 mg/kg b.w. of CME. Group IV & V received CHF in the doses of 250 and 500mg/kg b.w., respectively. Group VI received standard drug diclofenac sodium in in the dose of 100mg/kg. One hour after dosing (pre-emptive), the rats were challenged by a subcutaneous injection of 0.1 ml of 1% solution of carrageenan in 0.9% (w/v) sterile saline into the sub-plantar side of the left hind paw. The paw volume was measured again at 1 hr, 2 hrs, 3 hrs and 4 hrs after the challenge. The increase in paw circumference was calculated as percentage compared to the basal circumference as follows:
Where, Vt = is the average paw edema circumference of each extract treated group, as well as diclofenac sodium group;
Vc = is the paw circumference of the negative control group that only received the vehicle.
Formalin Induced Edema Assay: The formalin induced rat paw edema was carried out as described by Chau et al. [21] to evaluate acute anti-inflammatory activity of stem bark extract of Bauhinia acuminata Paw edema was induced by injecting 0.1 ml of 2.5% (w/v) formalin suspension in 0.9% (w/v) sterile saline into the plantar tissue of the right hind paw of experimental rats, one hour following oral administration of standard drug or test samples. Increase in paw circumference was measured at 0.5hr, 1hr, 2hrs, 3hrs and 4hrs after the challenge, using the method described by Tjolsen et al. [22]. The percentage inhibition of inflammation was calculated as follows:
Where, Vt = is the average paw edema circumference of each extract treated group, as well as diclofenac sodium group;
Vc = is the paw circumference of the negative control group that only received the vehicle.
WBC Migration Assay: Four hours after the administration of the inflammatory agent, the plantar aponeurosis of the inflamed paw was injected with 2% xylocaine, incised and the paw fluid of each rat aspirated (using 26G hypodermic needle) and slowly squirted into a test tube. The residual fluid was gently squeezed out, ensuring that blood did not mix with the fluid. Any fluid that had blood in it was discarded. The fluid was examined under microscope (x40) for any sign of breakages of the blood cells. Total white blood cell count in paw fluid was performed as described by Prempeh et al. 0.02 ml of paw fluid was mixed with 0.38 ml of WBC fluid (3% acetic acid with crystal violet dye), in a test tube. The mixture was transferred into a counting chamber, and the total number of WBC counted under a microscope (×40). The inhibition of migration of WBC was evaluated as percent reduction/inhibition in the treated animals relative to control animals using the relation:
% Inhibition of WBC migration = 100×(1−T / C)
Where, C = is the total WBC in control group
T= is the total WBC in treated group.
Statistical Analysis: All observations were presented as Mean ± SEM. The data was analyzed by one-way ANOVA followed by Dunnet’s test. *p< 0.05, **p< 0.01, ***p< 0.001 was considered as significant.
Results
Total Phenolic and Flavonoid Content
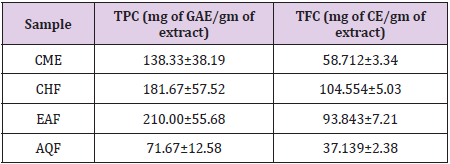
The total phenolic content (TPC) of the samples were calculated on the basis of the regression calibration curve, y = 0.0008x - 0.005 (R2=0.975) of gallic acid. The TPC of crude methanol extract (CME) and various fractions (CHF, EAF and AQF) were shown in Table 1. EAF possesses the highest phenolic content (210.00±55.68 mg of GAE/gm of sample) followed by CHF, CME and AQF. Total flavonoid content of the samples was calculated on the basis of the standard curve (y = 0.0178x + 0.0524; R2=0.9862) for catechin and results were expressed as mg of catechin equivalent (mg of CE)/gm of extract. The results were shown in Table 1. In this study, the CHF and EAF of crude methanol extract revealed the highest total flavonoid content (104.554±5.03 and 93.843±7.21 mg of CE/gm of sample, respectively).
Table 1: Total phenol and flavonoid content of the CME and various fractions of B. acuminata stem bark.
In vitro Antioxidant Assay
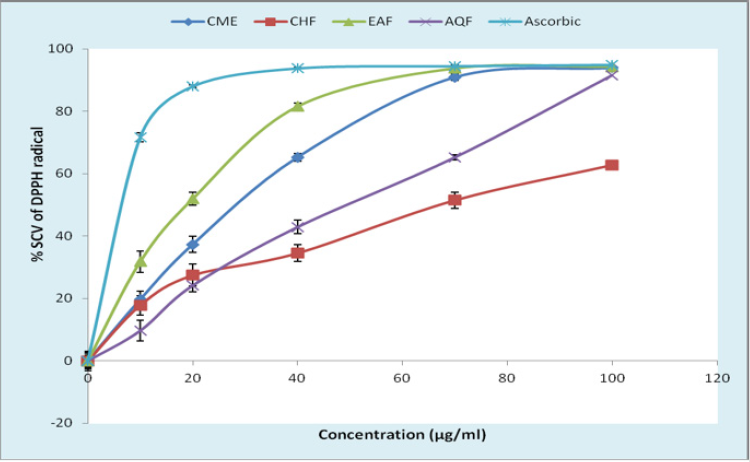
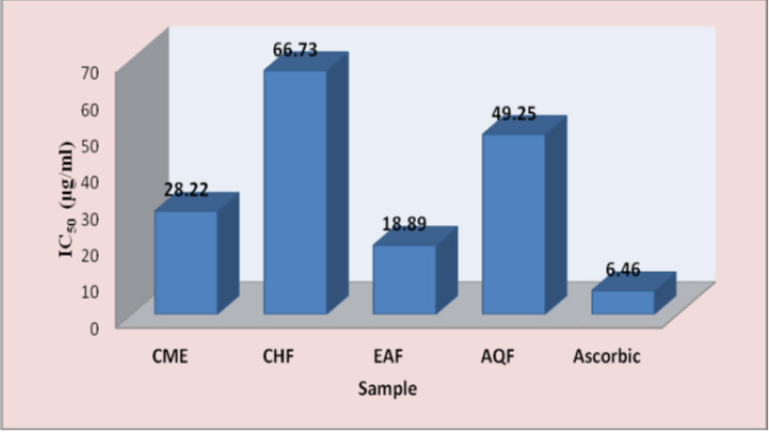
Effect on DPPH Radical-Scavenging Activity: The results of DPPH radical scavenging activity of CME and various fractions were shown in Figure 1 as comparable with reference standard ascorbic acid (AA). The scavenging activity of CME and fractions on DPPH radical was in the order of EAF>CME>AQF>CHF. At concentration of 100 μg/ml, the values for % scavenging of DPPH ranged between 62.78±2.60 to 94.33±0.68% and values for IC50 was in the range of 18.89 to 66.73 μg/ml, while at the same concentration that of AA was 94.90±0.39% with IC50 of 6.46 μg/ml (Figure 2). Standard AA had higher scavenging activity than CME and all the fractions but EAF showed DPPH scavenging activity which was close to standard AA.
Figure 2: Comparison of IC50 values of CME and various fractions of B. acumiata bark in DPPH radical scavenging assay.
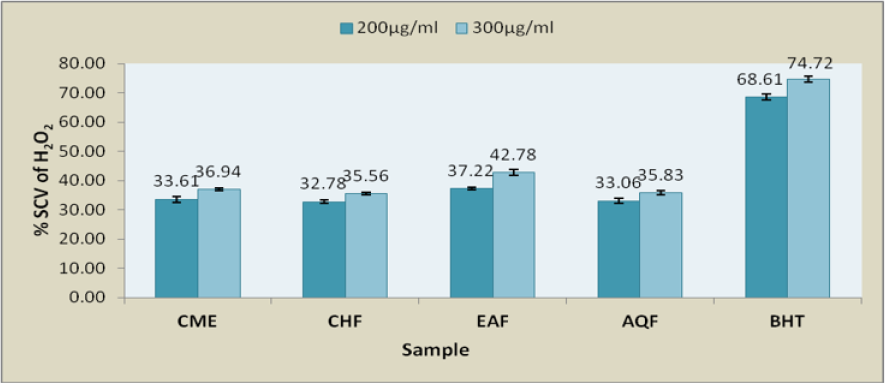
Hydrogen Per-Oxide Scavenging Assay: The results shown in Figure 3, CME extract of B. acuminata stem bark and its fractions demonstrated hydrogen peroxide scavenging activity in a concentration dependent manner. All the fractions including CME had moderate hydrogen peroxide scavenging activity compare with reference standard butylated hydroxyl toluene (BHT) (Figure 3).
Figure 3: Comparison of hydrogen peroxide scavenging activity of CME and different fractions with reference standard BHT.
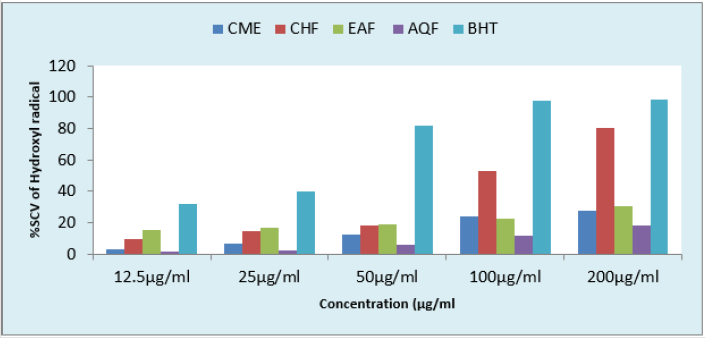
Hydroxyl Radical Scavenging Activity: The results of hydroxyl radical scavenging assay of various fractions and standard BHT were shown in Figure 4. The activity of the test samples was in the following order: CHF >EAF>CME>AQF. The highest scavenging activity were found in CHF and EAF. On the other hand, CME and AQF showed moderate scavenging activity. The results demonstrated that all the fractions and CME of bark of B. acuminata possessed significant hydroxyl free radical scavenging activity as compare to reference standard BHT.
Figure 4: Comparison of hydroxyl radical scavenging activity of CME and its various fractions at different concentrations with respect to BHT.
In vivo Anti-inflammatory Activity
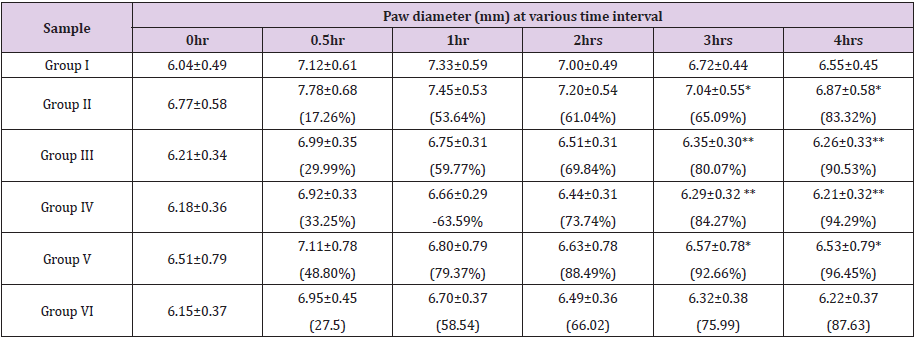
Carrageenan-Induced Paw Edema Test: Table 2 shows the results of antiedematous effects of orally administered CME and CHF of stems and standard drug as compared to carrageenan control at different hours on carrageenan-induced paw edema in rats. In the control group, the onset of edema occurred as early as 0.5 h after carrageenan injection and was sustained through the 4th hour of observation. Both CME and CHF caused a sharp decrease in paw edema from 1st hour until the 4th hour of treatment in a similar manner as that of the reference drug diclofenac. The most inhibition was obtained with CHF at 500 mg/kg dose (92.66% inhibition) followed CME (65.09% inhibition). Diclofenac sodium at a dose of 100 mg/kg p.o. prevented carrageenan induced paw edema with a percentage inhibition of 75.99% at 3 hours.
Table 2: Effect of CME, CHF and standard diclofenac sodium on inhibition of paw edema induced by carrageenan.
Values are mean ± SEM, (n=5); ** p<0.0001; *p<0.001 compared to reference standard drug treated group (one-way ANOVA followed by Dunnett’s test). Group l received normal saline (control group), Group II and III received 500 and 750 mg/kg b.w. of CME. Group IV & V were treated with 250 and 500mg/kg b.w. of CHF. Group VI received 100mg/kg of standard drug diclofenac sodium.
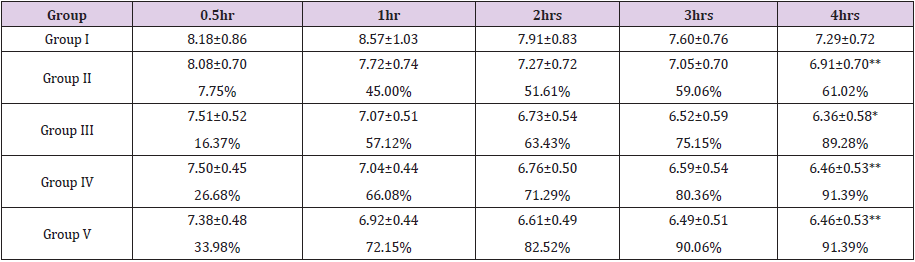
Formalin-Induced Paw Edema Test: CME administered at a dose of 500 mg/kg, showed 45.00%, 51.61%, 59.06%, and 61.02% inhibition at 0.5-4th hour, respectively while CHF at a same dose prevented paw edema with a percentage inhibition of 72.15%, 82.52%, 90.06%, 96.81% (Table 3). The percentage inhibition of standard anti-inflammatory drug was within the range of 61.08 to 91.07% (100 mg/kg, b.w). Each value represents the mean ± SEM from 5 animals in each group. Experimental group were compared to reference standard drug treated group; ** p<0.0001; *p<0.001 (one-way ANOVA followed by Dunnett’s test). Group l received normal saline (control group), Group II and III received 500 and 750 mg/kg b.w. of CME. Group IV & V were treated with 250 and 500mg/kg b.w. of CHF. Group VI received 100mg/kg of standard drug diclofenac sodium.
Table 3: Effect of CME, CHF and reference drug diclofenac on inhibition of paw edema induced by formalin.
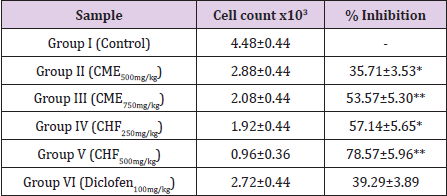
Total WBC Count in Paw Fluid: The results showed that the total number of WBC in the inflammatory exudates increased significantly after inflammation induced by carrageenan. CHF at higher dose (500 mg/kg) significantly (p<0.001) reduced the total WBC count in paw fluid by 78.57±5.96% on the other hand at dose of 750 mg/kg of CME, WBC count was reduced by 53.57±5.30%. The standard drug diclofenac sodium (100mg/kg) showed 39.29±3.89% inhibition (Table 4).
Table 4: Effect of CME and CHF on carrageenan induced WBC migration in rat paw edema fluid.
** p<0.0001; *p<0.001 compared to reference standard drug treated group
Discussion
The present study showed the antioxidant and antiinflammatory activity of crude methanol extract of the stem bark of B. acuminata in Wistar albino rats. Free radicals are capable of reacting with membrane lipids, nucleic acids, proteins and enzymes, and other small molecules, resulting in cellular damage. Several studies proposed that phenolic compounds are responsible for effective free radical scavenging and antioxidant activities [23]. Therefore, determination of the quantity of phenolic compounds is very important in order to determine the antioxidant capacity of plant extracts [24]. In the present study, higher concentration of polyphenols was found in EAF and CHF, suggesting the importance of EAF and CHF for effective antioxidant activities in traditional medicine. A number of methods are used to determine the radical scavenging effects of antioxidants. The DPPH method is a preferred method because it is fast, easy and reliable and does not require a special reaction and device. DPPH is a stable free radical, capable of accepting an electron or hydrogen radical to become a stable diamagnetic molecule [25].
Antioxidant compounds specially, phenolics present in the extract lose hydrogen which is received by DPPH and discoloration occurs due to the decreasing quantity of DPPH radicals in the environment. The discoloration of the DPPH therefore reflects the radical scavenging activity of the analyzed extract [26]. The higher content of total phenolic compounds and flavonoid in the extracts might have contributed towards the scavenging of free radicals [27]. CHF, which showed 2nd highest for the content of total phenol and highest of flavonoid content in this study but was weak to scavenge DPPH. This may be the other constituents in CHF that could antagonize the activity of this fraction. The presence of hydrogen peroxide inside the cell is non-reactive, but the conversion of hydrogen peroxide to hydroxyl radical is toxic, since hydroxyl radical reacts with other biomolecules in the cell, which eventually causes tissue damage.
It is reported that hydrogen donating ability of the plant extracts prevent the toxic effects of H2O2 by converting them into water [28]. The results demonstrated that EAF and other fractions may have the constituents which can protect tissue damage by preventing the toxic effect of hydrogen peroxide. Of all the reactive oxygen species, the hydroxyl (OH) is known to be one of the most reactive and physiologically harmful. As scavengers of hydroxyl radical, stem bark of B. acuminata can protect humans from a number of diseases. Antioxidation and anti-inflammation properties of plants are routinely investigated and are considered to be among the primary health benefits of natural products. In addition, antioxidative agents prevent the formation of a number of diseases including inflammatory diseases. Carragenan induced paw edema is one of the most widely used test procedure to screen anti-inflammtory drugs.
The time course of edema development in carragenan induced edema is a biphasic event, stimulating the release of histamine, serotonin and bradykinin in the first phase (30 min after carrageenan administration) and prostaglandin-like substances (PXs) in the second phase (0.5 to 4 h) [29]. These chemical substances produced increase in vascular permeability, thereby promoting accumulation of fluid in tissues that accounts for the oedema [30]. Formaldehyde which is also a potent oedematous agent can produce inflammation through the release of several inflammatory mediators including prostaglandins. The ability of the extract to reduce the size of oedema produced by carragenan and formaldehyde, suggests that it contained chemical components that may be active against inflammatory conditions. There is a significant percentage inhibition of paw edema from the 1st hour to 4th hour by both CHF and CME. CHF at the dose of 200 mg/kg body weight showed the percentage of inhibition of 42.6%.
At 400 mg/kg produced a highly significant 80% reduction in the volume of paw edema at the 4th hour. The percentage inhibition of standard anti-inflammatory drug diclofenac sodium was 58.% at 1st hour and 75.99% at 4th hour; 100 mg/kg, bw). Percentage of inhibition of mean paw volume of CHF at the dose of 200mg/kg was comparatively higher than that of the standard drug diclofenac sodium and CME. Since the extract (CHF and CME) may inhibit the paw edema both in early and late phase of carragenan induced inflammation. Therefore the possible inhibitory effect of extract in carragenan induced inflammation may be due to inhibition of cycloxygenase leading to inhibition of prostaglandin synthesis as well as inhibition of histamine and serotonin release at the 1st phase of inflammation. This effect is similar to that produced by non-steroidal anti-inflammatory drugs such as diclofenac and indomethacin, whose mechanism of action is inhibition of the cyclooxygenase enzyme.
The chemical constituents found in B. acuminata were vitamin C (leaves), β-sitosterol, lupeol, kamepferol, 3, 5, 7-dehydroxy and 5, 7 dimethoxy-flavanone-4-O-alpha-L-rhamnopyranosyl-β-D-glucopyranosides. Flavonoids showed presence of kamepferol, quercetin, and apigenin. Derivatives of quercetin i.e quercetin-3-glycoside was present in B. acuminata [31]. Several chemical compound including palmitic acid, three phthalic acid esters, phthalic acid, gallic acid, ursolic acid, were identified from the leaves of Bauhinia acuminate. These molecules have a wide range of biological activities. Phytochemical screening showed the presence of carbohydrate, phenolic compounds, saponins, flavonoids, oils and fats in leaves and stems of B. acuminata. Various flavonoids, glycosides and aglycones were previously reported as having potent anti-inflammatory and antioxidant activity. In the present study, we found that the CHF contains higher concentration (104.55 mg of CE/gm of extract) of flavonoids.
The ability of the crude methanol extract (CME) and its fraction CHF of stem bark of B. acuminata extract to reduce the size of oedema produced by carrageenan and formaldehyde, suggests that stem bark of this plant contained above mentioned chemical components that may be active against inflammatory conditions. On the other hand, the antioxidant potential of the extract can participate in fighting inflammation through the scavenging of reactive oxygen species produced at the site of inflammation by migrating WBC (neutrophils).
Conclusion
On the basis of results in this study, it can be concluded that the methanol extracts specially, EAF/CHF of B. acuminata stem bark is capable of scavenging a wide range of synthetic and naturally occurring free radicals and could be utilized as a good natural source of antioxidants. In addition, the results indicate that phenolic compounds might be major contributors to the antioxidant and anti-inflammatory activities of stem bark. Hence, this study supports the idea that B. acuminata stem bark can be a promising sources of potential antioxidants that can participate in fighting inflammation through the scavenging of reactive oxygen species. This preliminary study also warrants a need for further research to isolate, identify and characterize the specific phenolic compounds which may contribute in the therapy of diseases related to oxidative stress.
References
- Chen J, He J, Hamm L, Vatuman V, Whelton PK (2002) Serum antioxidant vitamins and blood pressure in the United States population. Hypertension 40(6): 810-816.
- Ray G, Husain SA (2002) Oxidants, antioxidants and carcinogenesis. Ind J Exp Biol 40(11): 1213-1232.
- Madhavi DL, Deshpande SS, Salunkhe DK (1995) Toxicological aspects of food antioxidants. In: Food antioxidants. New York, Marcel Dekker Inc, pp: 267-290.
- Kris-Etherton PM, Lefevre M, Beecher GR, Gross MD, Keen CL, et al. (2004) Bioactive compounds in nutrition and health-research methodologies for establishing biological function: the antioxidant and anti-inflammatory effects of flavonoids on atherosclerosis. Annu Rev Nutr 24: 511-538.
- Wang S, Konorev EA, Kotamraju S, Joseph J, Kalivendi S, et al. (2004) Doxorubicin induces apoptosis in normal and tumor cells via distinctly different mechanisms, intermediacy of H2O2 and P53-dependent pathways. J Biol Chem 279(24): 25535-25543.
- Trouillas P, Calliste CA, Allais DP, Simon A, Marfak A, et al. (2003) Antioxidant, anti-inflammatory and anti-proliferative properties of sixteen water plant extracts used in the Limousin countryside as herbal teas. Food Chem 80(3): 399-407.
- Schinella GR, Tournier HA, PJrieto JM, Mordujovich de Buschiazzo P, Rios JL (2003) Antioxidant activity of anti-inflammatory plant extracts. Life Sci 70(9): 1023-1033.
- Winrow VR, Winyard PG, Morris CJ, Blake DR (1993) Free radicals in inflammation: second messengers and mediators of tissue destruction. Br Me Bull 49(3): 506-522.
- Khare CP (2001) Indian medicinal plants-An Illustrated Dictionary Springer-Verlag Berlin: 85.
- Silja VP, SamithaVarma K, Mohanan KV (2000) Ethnomedical plant knowledge of the Mullu Kuruma tribe of Wayanad district, Kerala, Indian. J Trad Knowl 7(4): 604-612.
- Phansri K, Sarnthima R, Thammasirirak S, Boonchalee P, Khammuang S (2011) Antibacterial Activity of acuminata Seed Protein Extract with Low Hemolytic Activity Against Human Erythrocytes. Chiang Mai J Sci 38: 242-251.
- Ferdous MR, Akhter S, Khan MZ, Khan ME, Islam MA, et al. (2014) Ex-Vivo anti-inflammatory and antimicrobial activities of the leaves of Bauhinia acuminate. Am J Life Science 2: 267-270.
- Khatoon M, Islam E, Islam R, Rahman AA, Alam K, et al. (2013) Estimation of total phenol and in vitro antioxidant activity of Albizia procera leaves. BMC Research Notes 6: 121.
- Nandita D, Islam ME, Nusrat J, Islam MS, Alam K, et al. (2014) Antioxidant activities of ethanol extracts and fractions of Crescentia cujete leaves and stem bark and the involvement of phenolic compounds. BMC Com and Alt Me 14: 45.
- Nusrat J, Parvin MS, Nandita D, Islam MS, Islam ME (2014) Studies on the antioxidant activity of ethanol extract and its fractions from Pterygota alata leaves. J of Acute Med 4(3): 1-6.
- Tjolsen A, Berge OG, Hunskaar S, Rosland JH, Hole K, (1992) The formalin test an evaluation of the method. Pain 51(1): 5-7.
- Velioglu YS, Mazza G, Gao L, Oomah BD, (1998) Antioxidant activity and total phenolics in selected fruits, vegetables and grain products. J Agric Food Chemistry 46: 4113-4117.
- Das NP, Pereira T (1990) Effects of flavonoids on thermal autoxidation of palm oil: Structure-activity relationships. J Am Oil Chem Soc 67(4): 255.
- De Gaulejac NSC, Glories Y, Vivas N (1999) Free radical scavenging effect of anthocyanins in red wines. Food Res Int 32(5): 327-333.
- Hatano T, Edamatsu R, Hiramatsu M, Mori A, Fujita Y (1989) Effects of the interaction of tannins with co-existing substances. VI: effects of tannins and related polyphenols on superoxide anion radical and on 1,1-diphenyl-2-picrylhydrazyl radical. Chem Pharm Bull 38(5): 2016-2021.
- Baumann J, Wurn G, Bruchlausen FV (1980) Prostaglandin synthetase inhibiting O2-radical scavenging properties of some flavonoids and related phenolic compounds. Naunyn-Schmiedebergs Arch Pharmacol 313(4): 27-32.
- Guo XY, Wang J, Wang NL, Kitanaka S, Yao XS (2007) 9, 10 Dihydrophenanthrene derivatives from Pholidota yunnanensis and scavenging activity on DPPH free radical. J Asian Nat Prod Res 9(2): 165-174.
- Molyneux P (2004) The use of the stable free radical diph- enylpicrylhydrazyl(DPPH) for estimating antioxidant activity. Songklanakarin J Sci Technol 26(2): 211-219.
- Williams TJ, Morley J (1973) Prostaglandins as potentiators of increased vascular permeability in inflammation. Nature 246(5430): 215-217.
- White M (1999) Mediators of inflammation and inflammatory process. Journal of Allergy and Clinical Immunology 103(3pt2): 5378-5381.
- Sinha KN, Sinha T (2013) Phytochemical screening and electrophoretic study of seed storage proteins of Bauhinia acuminata and Cassia occidentalis. Int J Sci Res 4: 1812 -1816.
- Nag S, Paul A, Datta RP (2013) Phytochemical analysis of some medicinal plants. Int J Sci Res Publicat3(4): 1648.
- Augusto O, Kunze KL, Montellano PR (1982) Phenylprotoporphyrin formation in the haemogolobinphenylhydrazine reaction. J Biol Chem 257: 6231-6241.
- Ferrali M, Signorni C, Ciccoli L, Comporti M (1992) Iron release and membrane damage in erythrocytes exposed to oxidizing agents, phenylhydrazine, divicine and isouramil. Biochem J 285(Pt1): 295-301.
- Anosike C, Obidoa O, Ezeanyika L (2012) Membrane stabilization as a mechanism of the anti-inflammatory activity of methanol extract of garden egg (Solanum aethiopicum). Daru 20(1): 76.
- Dongray A, Irrchariya R, Chanchal D, Chaudhary S (2015) Phytochemical and pharmacological properties of Bauhinia acuminate. World J Pharm Res 5: 531-546.

 Research Article
Research Article