Abstract
Relevance
The treatment of giant pituitary adenomas (PA) is not only the lot of neurosurgeons, but also other specialists, first of all, endocrinologists, because arising (or aggravating) after surgery or radiation therapy in almost all patients with hypopituitarism (and often panhypopituitarism) requires substitution (sometimes lifelong ) or corrective hormonal therapy (Ugryumov VM 1963, Starkova NT 1983, Vaks VV 1999, Greenman Y et al 1996). Nevertheless, there is currently no algorithm correction of hormonal disorders in patients with PA in the early and late postoperative period, meanwhile, it is the latter largely determine their quality of life. The causes of the continued growth of the tumor and its determining factors, as well as the tactics of surgical or conservative treatment in these cases, have not been clarified. Among the large number of existing classifications of PA, there are no classifications that fully reflect the size of the tumor, the ways of its spread, the nature of growth, hormonal activity, which presents certain difficulties for the development of a detailed clinical diagnosis, tactics of surgical treatment and correction of hormonal disorders. There is widespread discussion, despite the effectiveness proved in a number of papers, the feasibility of applying radiation treatment for giant PA due to a large number of complications (Sheline GE, 1979; Tsang RW et al., 1996; Sasaki R. et al., 2000), at present, the results of surgery of giant pituitary adenomas cannot be considered satisfactory, and the postoperative treatment tactics are far from perfect. Considerable postoperative lethality, a high level of disabled people dictates the need for further targeted study of this problem, aimed at improving the results of treatment [1-3]. All the above determines the relevance of this research topic. All the above was the reason for conducting this research.
Keywords:Giant Pituitary Adenomas; Clinic; Complications
The purpose of the study
To study the frequency and clinical and hormonal characteristics of patients with giant formations of the chiasmal-sellar region (CSR) according to a retrospective analysis.
Material and methods of research
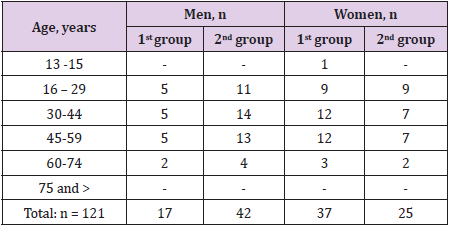
For the period from 2015 to 2017 years we performed a retrospective analysis of data from three Centers in Tashkent, namely the RSRSPC of Endocrinology of the Ministry of Public Health of the Republic of Uzbekistan, the Republican Scientific Center for Neurosurgery and the Republican Scientific Center for Emergency Medical Care. A total of 121 adult patients with macroand giant pituitary adenomas were analyzed. Of them - men - 59 (48.9%), women - 62 (51.1%). Middle age: men were 37.12 years old, women - 38, 15 years old. Disease duration ranged from 2 months to 25 years. All patients were divided into 2 groups: group 1 - patients with pituitary macroadenomas - 53 (43.8%) cases and group 2 - patients with giant pituitary adenomas - 68 cases (56.1%). The research methods included:
a) general clinical (complaints, anamnesis, filling in
the subject patient questionnaire, research of endocrine,
neurological statuses, anthropometry - height, weight, FROM,
OB, BMI).
b) instrumental (perimetry for all colors, fundus, visual
acuity, ECG, densitometry, CT/MRI (computed tomography /
magnetic resonance imaging) of the Turkish saddle and adrenal
glands, etc.).
c) hormonal blood tests (STG, IFR-1, LH, FSH, prolactin, TSH,
ACTH, free testosterone, estradiol, cortisol, free thyroxin).
d) biochemical blood tests (LDL, HDL, cholesterol,
triglycerides, calcium, chlorides, sodium, etc.). Based on the
completed studies, the following results were obtained.
The obtained data were processed using computer programs Microsoft Excel and STATISTICA_6. The significance of quantitative differences (n>12) was determined by the Wilcoxon method for unrelated ranges, to determine the reliability of small samples (n<0.05. The mean values (M), standard deviations of the mean (m) were calculated.12), the non-parametric randomization criterion for the Fisher components for independent samples was used. Differences between groups were considered statistically significant at P [4-9]. The significance of differences in the level between groups was assessed by the magnitude of the confidence interval and student’s criterion (p). Differences were considered statistically significant at p<0.05.
The Results of the Study and their Discussion
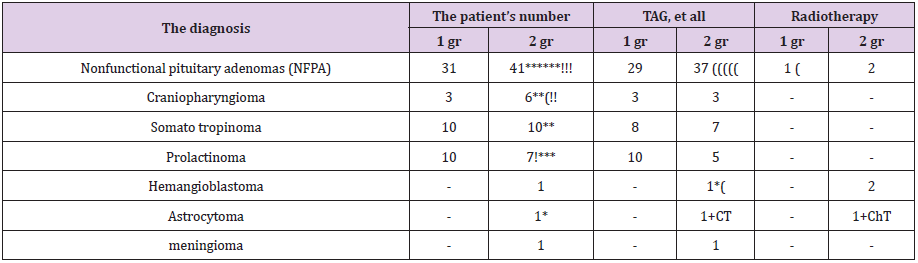
Table 1 shows the distribution of patients by sex and age. Distribution of patients according to the topographic-anatomical classification of the side of growth of the pituitary adenoma by Kadashev B.A. (2007) showed that pituitary adenomas with a total growth variant were most often observed - 27 cases (22.4%), infra-suprastosellar growth - 28 cases (23%), infra-suprasellar growth - 28 cases (23%) . It was these cases that differed more vivid clinical symptoms, due to the direction of tumor growth. Table 2 shows the distribution of patients according to the nature of the formation of the sellar region. From Table 2 NFPA was the most frequent - 72 cases out of 121 (56%). At the same time, 51 patients (98%) out of 53 in group 1 underwent surgical treatment - transnasal pituitary adenomectomy (TAG). 55 patients (80%) of 68 in group 2 were subjected to surgical treatment - transnasal adenomectomy of the pituitary gland. A total of 106 TAGs were performed in both groups. Repeated operations on the pituitary gland were performed in 8 patients (7%) of both groups. Radiation therapy was received by 6 (5%) patients and 1 by chemotherapy. It was found that panhypopituitarism was the most common in group 2 patients - in 32 of 68 patients (44.4%), postoperative panhypopituitarism occurred in 59 of 68 patients (55.5%), while in group 1 panhypopituitarism did not was observed. Bitemporal hemianopsia was also observed with a greater frequency in patients of group 2-41 cases out of 68 (61.1%). In addition, secondary amenorrhea was also more frequent in patients of group 2-27 cl (33.3%). Such disorders as secondary osteopenia, delayed physical and sexual development were identified only in group 2 patients. Thus, the most pronounced neuroendocrine and ophthalmologic disorders occurred in patients of group 2.
Table 2: The distribution of patients according to the nature of the formation of the sellar region.
Note: TAG - Trans nasal adenectomy of the pituitary gland, - relapse growth! - stromal hemorrhage, (- repeat RT operation - number of patients who received radiation therapy, chemotherapy, CT - combination therapy.
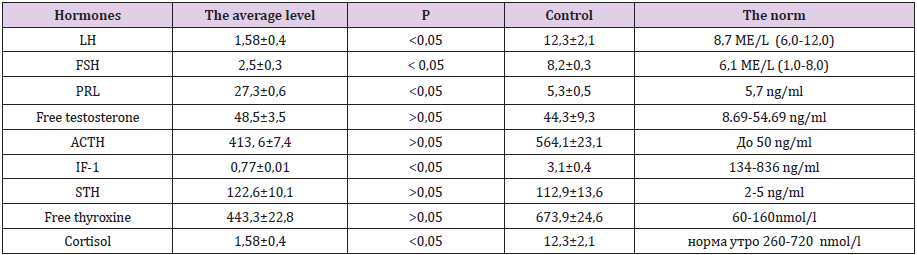
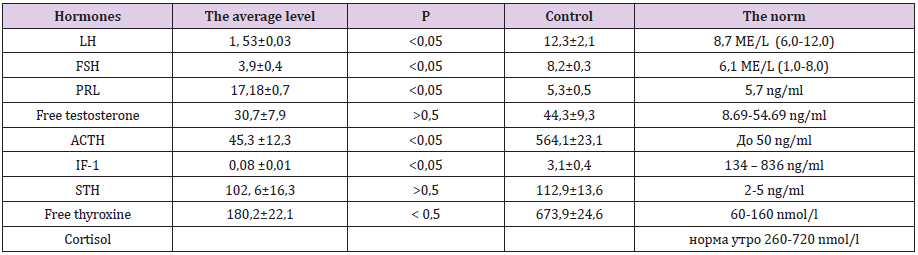
The most pronounced disorders of the neurological status - endocrine encephalopathy, hallucinations (1 case), inadequate condition (1 cases), amaurosis (9 caces), ischemic stroke (2 caces), pathological reflexes, impaired muscle tone - decreased - were observed in patients 2 during the germination of a tumor in the brain, the groups are the anterior, middle, or posterior cranial fossa, with the growth of the tumor in the ventricles of the brain and both cavernous sinus [5-10].The most pronounced violations of the visual fields in the form of bitemporal hemianopsia were observed in patients of both groups with endo-suprasellar growth - 18 caces (14.8%), with a total growth variant - 36 cl (30%), with endosupra- infrasellar growth - 28 sl (23%) and supra-infra-laterosellar growth - 28 caces (23%). Complications such as hemorrhage into the tumor in 6 out of 58 (10%), tumor recurrence in 14 out of 58 (24%) were found only in 2 grams of patients. Tables 3,4 give the average basal plasma hormone levels by group. As can be seen from Tables 3,4, the most significant decrease in the mean values of basal levels of plasma tropic hormones - GH, LH, FSH, ACTH - was recorded in patients of group 2. In addition, these patients also significantly reduced levels of cortisol, while in patients of group 1 plasma cortisol was on average within the normal range [11-15]. Thus, the most pronounced neuroendocrine complications were found in patients with giant pituitary adenomas.
Table 3: The average value of plasma hormones in patients of group 1 (n = 53).
Note. P - significance of differences compared with the control group.
Conclusions
a) In total, over the period 2012-2017, 68 patients with giant
pituitary adenomas were treated in three Centers in Tashkent
city, among which inactive pituitary adenoma was the most
common - 41 cl (60%), less often - somatotropinoma - 10 cl
(15%), prolactinoma - 7 cl (10%), craniopharyngioma - 6 cl
(9%).
b) The most pronounced neuroendocrine and ophthalmologic
disorders occurred in patients of group 2 with giant pituitary
adenomas.
c) Neuroendocrine (GHD, panhypopituitarism, hypopituitarism,
infertility, secondary amenorrhea), ophthalmological
(GD, amaurosis, etc.) and stem disorders (pyramidal symptoms,
reduced reflexes and muscle tone diffusely) were observed in
patients with a total growth variant in giant hymenopaque patients
with giant adrenal genes, reduced reflexes and muscle
tone diffusely) were observed in patients with a total growth
variant in giant hybrids, reflexes and muscle tone diffusely).
d) In patients with giant pituitary adenomas, a decrease in the
level of GH, FSH, LH, ACTH (55%), that is, panhypopituitarism
on the background of cerebral and stem symptoms, is primarily
noted.
e) Neurovisualization marker of tumor aggressiveness can
be different variants of its total growth, namely: supra-infralatersellar
growth, supra-infrasellar growth observed in 56
patients (46.2%) of both groups.
Clinic-hormonal description of patients with giant pituitary adenomas”. In this scientific article, the authors discussed the results of studies of 121 patients with various volume formations of the sellar area, of which 59 men (48.9%) and 62 women (51.1%) women [16-21]. Depending on the size of the pituitary adenoma, detected on CT/MRI, the patients were divided into two groups: group 1 patients - macroadenomas (from 20 to 30mm) - 53 (43.8%), and group 2 patients - giant (more 30mm) - 68 (56.1%). Pituitary adenomas with a total growth pattern were most often observed - 27 cases (22.4%), infra-suprastoserellar growth - 28 cases (23%), infra-suprasellar - 28 cases (23%), It was these cases that differed brighter clinical symptoms due to the direction of tumor growth. NFPA was the most common - 72 cases out of 121 (56%). At the same time, 51 patients (98%) out of 53 in group 1 underwent surgical treatment - transnasal pituitary adenomectomy (TAG). 55 patients (80%) of 68 in group 2 were subjected to surgical treatment - transnasal adenomectomy of the pituitary gland. A total of 106 TAGs were performed in both groups. Repeated operations on the pituitary gland were performed in 8 patients (7%) of both groups. Radiation therapy was received by 6 (5%) patients and 1 by chemotherapy. It was found that the most pronounced neuroendocrine complications were found in patients with giant pituitary adenomas.
References
- DedovII, Melnichenko GA (2009) Endocrinology. National leadership city. Moscow,pp.597.
- Marova EI (1999) Neuroendocrinology. Clinical essays. Moscow pp. 380-401.
- Serov NK (1993) Neuro-ophthalmological syndromes in the clinic of tumors of the basal-diencephalic localization (craniopharyngiomas, gliomas of the chiasm and bottom of the third ventricle). Thesis for the degree of doctor of honey. Sciences, Moscow.
- Ulitin AYu (2008) Giant adenomas of the pituitary gland: clinical features and treatment. Diss. on the competition uch. Art. specialty - Eye diseases, St. Petersburgpp. 302.
- Fayzullaev RB (2009) Giant adenomas of the pituitary (clinic, diagnosis, surgical treatment). Diss. on the competition uch.st. Dr. honey. sciences in the specialty - Neurosurgery, Moscowpp. 337.
- Shesterikov AA (2011) Surgical methods for the prevention of postoperative complications in the treatment of pituitary tumors. Diss. on the competition uch. Art. Cand. honey. sciences on spec- Surgery, Omsk, pp. 130.
- Shahvorost NP (2009) Improving the organization of medical care for patients with pituitary adenomas. Abstract for a student account. degree cms using cipher code 14.00.33, Moscow p. 30.
- Espinosa E, Sosa E, Mendoza V, Ramirez C, Melgar V, et al. (2016) Giant prolactinomas: are they really different from ordinary macroprolactinomas? Endocrine52(3):652-659.
- Chohan MO, Levin AM, Singh R, Zhou Z, Green CL, et al. (2016) Three-dimensional volumetric measurements in defining endoscope-guided giant adenoma surgery outcomes.Pituitary19(3):311-321.
- Besser, MOThorner, SReichlin (1994) Slide atlas of clinical endocrinology. 2nd (Edn.), UK pp. 316.
- Black PM, Zervas NT, Candia G (1988) Management of large pituitary adenomas by transsphenoidal surgery.Surgical Neurology29(6):443-447.
- Han S, Gao W, Jing Z, Wang Y, Wu A (2017) How to deal with giant pituitary adenomas: transsphenoidal or transcranial, simultaneous or two-staged?J Neurooncol132(2):313-321.
- Graillon T, Castinetti F, Fuentes S, Gras R, Brue T, et al. (2017) Transcranial approach in giant pituitary adenomas: results and outcome in a modern series. J Neurosurg Sci.
- Komninos J, Vlassopoulou V, Protopapa D, Korfias S, kontogeorgos G, et al. (2004) Tumors metastatic to the pituitary gland: case report and literature review. J Clin Endocrinol Metab 89(2): 574-580.
- Landolt AM (2017) History of pituitary surgery. In: Greenblatt SH (Eds.), A History of Neurosurgery. In Its Scientific and Professional Contexts. Park Ridge, IL.American Association of Neurological Surgeons pp. 373-400.
- Marques Pedro, KorbonitsMárta(2017) Genetic Aspects of Pituitary Adenomas. Endocrinology and Metabolism Clinics of North America 46(2): 335-337.
- MelmedShlomo(2015) Pituitary Tumors. Endocrinology and Metabolism Clinics of North America 44(1): 1-9.
- RaverotGérald, Vasiljevic Alexandre,Jouanneau Emmanuel,Trouillas J(2015) Prognostic Clinicopathologic Classification of Pituitary Endocrine Tumors. Endocrinology and Metabolism Clinics of North America 44(1): 11-18.
- Yano S, Hide T, Shinojima N (2017) Efficacy and Complications of Endoscopic Skull Base Surgery for Giant Pituitary Adenomas.World Neurosurg99:533-542.
- Yosef L, Ekkehard KM, Shalom M (2016) Giant craniopharyngiomas in children: short- and long-term implications. Childs Nerv Syst32(1):79-88.
- Zakir JC, Casulari LA, Rosa JW, de Mello PA, de Maghalhaes AV, et al. (2016) Prognostic Value of Invasion, Markers of Proliferation, and Classification of Giant Pituitary Tumors, in a Georeferred Cohort in Brazil of 50 Patients, with a Long-Term Postoperative Follow-Up.Int J Endocrinol2016:7964523.

 Research Article
Research Article