Abstract
The advent of bioceramics materials increases the possibility of reparative and regenerative processes in dentistry, and more precisely, in endodontics. Bioceramics materials presents biocompatibility, bioactivity, resistance to leakage, sealing ability, and biomineralization activity. Mineral trioxide aggregate (MTA) is a gold standard bioceramic material indicated for reparative procedures presenting high clinical success. MTA is used for pulp capping, pulpotomy, apexification, perforation, root resorption repair, regenerative endodontics, and apical surgery. The emergence of MTA demonstrated great potential in endodontic therapies, and modifications and improvements of this bioceramic gave rise to many other materials. New bioceramic materials are now available in several countries with distinct compositions. Although there is a concern regarding the modification of compounds and formulation of bioceramic materials for reaching better properties, distinct products produce the same effects of reduced leakage, biological activity, sealing ability, marginal adaptation and bridge formation. The biological properties of bioceramic materials are well defined in literature, but new formulations must be evaluated in longitudinal clinical cases, and systematic reviews and meta-analysis are needed to compare these products better. The present review demonstrates a summary of bioceramics properties, applications, improvements, and future directions in endodontics to maintain tooth longevity.
Keywords: Bioceramics; Endodontics; MTA
Introduction
The dental structure can be affected by physical, chemical, and biological events. Dental caries and trauma are the most frequent aggressions involving teeth [1]. Both circumstances can trigger pulpal responses and culminate in inflammation and/or tissue necrosis [2]. The necrotic pulp can be infected by many microorganisms that can mediate the inflammation and destruction of the tooth surrounding tissues [3]. Pulp and periradicular inflammation can also trigger root internal/external resorption and alveolar bone resorption [4,5]. Thus, several pathological conditions can affect dental structures and, the first intervention must be based on eliminating the aggression by treating trauma and/or infection (e.g., treating caries, restor ing cavities, performing endodontic therapy). After tissue inflam mation and repair should be stimulated, and a new tissue can be formed or replaced. For these reasons, medicine and dentistry are in constant improvements involving machines, techniques, and materials for treating diseases and improving life quality. The advent of bioceramics materials increases the possibility of reparative and regenerative processes in dentistry, and more precisely, in endodontics. The present review demonstrates a summary of bioceramics properties, applications, improvements, and future directions in endodontics to maintain tooth longevity better.
Bioceramics Applications for Reparative Endodontics
Ceramics are inorganic non-metallic materials produced by heated raw minerals [6]. Bioceramics are materials composed of nanosphere particles of tricalcium silicate, tantalum pentoxide, dicalcium silicate, monobasic, amorphous silicon dioxide, and calcium phosphate [7]. Bioceramic materials include alumina and zirconia, bioactive glass, glass ceramics, calcium silicates, hydroxyapatite, resorbable calcium phosphates, and radiotherapy glasses [8]. These materials presents biocompatibility [9], bioactivity [10], resistance to leakage, sealing ability [11], and biomineralization activity [12]. The presence of calcium silicate in these materials can reduce inflammation and induce tissue repair [13]. Calcium phosphate and bioactive glass induce the formation of hard tissue [14]. In addition, bioceramic materials contribute to tissue repair once it stimulates the secretion of morphogenetic cells, proteins, and growth factors such as bone morphogenetic protein and transforming growth factor- beta 1 [12]. Endodontic applications for bioceramic materials for reparative purposes demands several physicochemical properties (short setting time, high alkaline pH and calcium ion release, high mechanical strength, high radiopacity, moderate flow, low porosity, and solubility) and also biological properties (biocompatibility, induction of pulp cell differentiation, and antibacterial activity) to ensure its effectiveness [15,16].
However, current products commercially available do not demonstrate all these desirable properties, but there are products with favorable clinical results. Mineral trioxide aggregate (MTA) is a gold standard bioceramic material indicated for reparative procedures presenting high clinical success [17]. MTA was first cited in the 1990s as a calcium silicate cement [18]. Ever since, MTA is used for pulp capping, pulpotomy, apexification, perforation, root resorption repair, regenerative endodontics, and apical surgery [19]. MTA is a biocompatible and sealing material that can induce tissue repair (bone and dentin) [20]. The recommendations for bioceramics application involve repair or regeneration of pulp tissue, periodontal tissues and also dentin, and bone tissues. MTA is recommended for direct pulp capping and pulpotomy procedures as a material to cover exposed tissue and act as a barrier. The direct contact of MTA with pulp tissue can stimulate the healing and repair of the pulp through the induction of dentin bridge formation [21].
Bioceramics as MTA is indicated for regenerative endodontic therapy due to its potential to assist root development. The ability to seal, induce cell proliferation, differentiation, and biomineralization makes these materials the most suitable for blood clot top-sealing [22]. MTA allows adhesion, supports cell proliferation, and induce mesenchymal stem cell migration [23]. MTA is also recommended for the repair of areas where periodontal communication occurs (e.g., perforations and root-end filling) [21]. This recommendation is based on the properties of biocompatibility, dimensional stability, and sealing [24]. Indeed, MTA can block communications of the root canal and surrounding tissues. The first commercial formulation of MTA was developed as a gray powder. However, gray MTA was developed into white MTA to overcome tooth discoloration [25]. Tooth discoloration occurs due to the presence of bismuth oxide, a radiopacifier that can produce metallic bismuth and oxygen [26,27]. Other disadvantages of MTA include long setting time, difficulties in handling, and high cost [13,28]. In addition, its properties also depend on manipulation. The ratio powder/water and entrapped air during mixing can be interfering and contribute to the waste of material [18].
Bioceramics Evolution in Endodontics
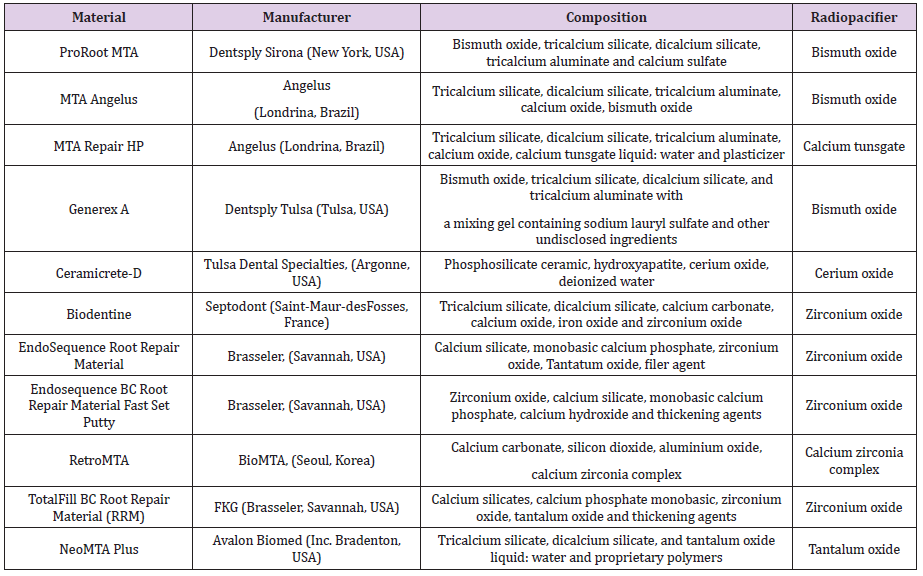
Over the years, many materials have been recommended to induce tissue repair of dental and periapical tissues (e.g., eugenol zinc oxide cements, glass ionomer cement, composite resins, amalgam, gutta-percha, calcium hydroxide) [29]. The emergence of MTA demonstrated great potential in endodontic therapies, and modifications and improvements of this bioceramic gave rise to many materials. New bioceramic materials are available in several countries with distinct compositions. Table 1 demonstrates some examples of bioceramic materials similar to MTA. Endosequence Bioceramic Root Repair Material (Brasseler, Savannah, USA) was developed as a premixed cement into a syringeable fast set putty to improve manipulation better and discoloration disadvantages [25]. Endosequence contains zirconium dioxide as a radiopacifier that do not form a precipitate in contact with collagen [27]. However, results regarding its potential of discoloration are distinct [30]. RetroMTA (BioMTA, Seoul, Korea) also presents a modification on its composition compared to MTA and do not present Portland cement, and used hydraulic calcium zirconia as radiopacifier [31]. This product has calcium carbonate and may present fast setting time (3 minutes) compared to MTA but it still demonstrated some discoloration potential [32]. Generex A (Dentsply Tulsa, Tulsa, USA) is similar to ProRoot MTA (Dentsply Sirona, New York, USA), but is mixed with gels instead of water [33]. Some studies demonstrated that tricalcium silicate from Generex A stimulates osteoblast growth and contributes to the formation of bone apatite in a similar manner of MTA [33,34]. Ceramicrete-D (Tulsa Dental Specialties, Argonne, USA) is composed of hydroxyapatite powder, phosphosilicate ceramics, and radiopaque cerium oxide, although it may also contain bismuth oxide as a radiopacifying agent [34,35]. It demonstrated sealing ability and alkalinity [35], handling properties similar to ProRoot MTA (Dentsply Sirona, New York, USA) and setting time of 150 minutes [33]. However, its biocompatibility is still controversial [34].
Table 1: Bioceramic materials from distinct countries and with distinct composition and formulations.
Biodentine (Septodont, Saint-Maur-desFosses, France) is composed of a mixture of tricalcium silicate, dicalcium silicate, calcium carbonate, iron oxide, and zirconium oxide, and a water-soluble polymer calcium chloride [36]. The radiopacity is attributed to zirconium oxide, but it might not provide a significant radiographic contrast [37,38]. Biodentine presents a short setting time (12 minutes) due to the presence of smaller particles, and the use of calcium chloride, an accelerator of chemical reactions [28]. This material releases calcium hydroxide induces restorative dentin synthesis, and also demonstrated antimicrobial activity [39,40]. Total Fill BC Root Repair Material (RRM, FKG Brasseler, Savannah, USA) is based on calcium phosphate, possess prolonged setting time (4 hours) but presented some limitations of handling and sealing ability compared to ProRoot MTA (Dentsply Sirona, New York, USA) [41]. NeoMTA Plus (Avalon Biomed, Inc. Bradenton, USA) is a tricalcium silicate, dicalcium silicate, and calcium sulfate powder mixed with a water-based gel. This product is based on tantalum oxide as a radiopacifying agent and possesses prolonged setting time for a prolonged effect of calcium and hydroxyl ions release [42]. There are other bioceramic materials available in the market with similar compositions and properties. Although there is a concern regarding the modification of compounds and formulation of bioceramic materials for reaching better properties, distinct products produce the same effects of reduced leakage [24,43], biological activity [14,44], sealing ability, marginal adaptation [41] and bridge formation [7].
Future Directions
A great vast of studies regarding MTA and other bioceramic materials as regenerative materials for endodontics demonstrated its effectiveness. However, the emergence of new biomaterials is needed in dentistry, aiming to prolong tooth life expectancy and also systemic life quality. The improvements of formulation (e.g., the premixed formulation in a syringe, putty formulation) consist of significant advancement and also the concerns regarding tooth discoloration and compound modifications. However, manufacturers and researchers need to attend cases of reintervention were the removal of these materials is needed. In addition, high costs also must be a great concern to disseminate the beneficial use of bioceramics in daily practice better. The biological properties of bioceramic materials are well defined in many in vitro and in vivo studies, but new formulations must be evaluated in longitudinal clinical cases, and systematic reviews and meta-analysis are needed to compare these products better. Finally, several products present effective results even when compared to gold standard MTA, and the bioceramic choice should be based on professional experience [45].
Conflicts of Interests
none
References
- DK Rechenberg, JC Galicia, OA Peters (2016) Biological Markers for Pulpal Inflammation: A Systematic Review. PLoS One 11(11): e0167289.
- B Hashemi Beni, M Khoroushi, MR Foroughi, S Karbasi, AA Khademi, et al. (2017) Tissue engineering: Dentin - pulp complex regeneration approaches (A review). Tissue Cell 49(5): 552-564.
- PN Nair (2004) Pathogenesis of apical periodontitis and the causes of endodontic failures. Crit Rev Oral Biol Med 15(6): 348-381.
- M Yoshpe, S Einy, N Ruparel, S Lin, AY Kaufman (2019) Regenerative Endodontics: A Potential Solution for External Root Resorption (Case Series). J Endod.
- Y Yang, L Huang, Y Dong, H Zhang, W Zhou, et al. (2014) In vitro antibacterial activity of a novel resin-based pulp capping material containing the quaternary ammonium salt MAE-DB and Portland cement. PLoS One 9(11): e112549.
- A Shenoy, N Shenoy (2010) Dental ceramics: An update. Journal of conservative dentistry: JCD 13(4): 195-203.
- S Azimi, M Fazlyab, D Sadri, MA Saghiri, B Khosravanifard, et al. (2014) Comparison of pulp response to mineral trioxide aggregate and a bioceramic paste in partial pulpotomy of sound human premolars: A randomized controlled trial. International endodontic journal 47(9): 873-881.
- A Nasseh (2009) The rise of bioceramics. Endodontic Practice 2: 17-22.
- T Takita, M Hayashi, O Takeichi, B Ogiso, N Suzuki, et al. (2006) Effect of mineral trioxide aggregate on proliferation of cultured human dental pulp cells. Int Endod J 39(5): 415-422.
- MG Gandolfi, P Taddei, A Tinti, C Prati (2010) Apatite-forming ability (bioactivity) of ProRoot MTA. Int Endod J 43(10): 917-929.
- J Camilleri, MG Gandolfi, F Siboni, C Prati (2011) Dynamic sealing ability of MTA root canal sealer. Int Endod J 44(1): 9-20.
- EM Rodrigues, AL Cornelio, LB Mestieri, AS Fuentes, LP Salles, et al. (2016) Human dental pulp cells response to mineral trioxide aggregate (MTA) and MTA Plus: cytotoxicity and gene expression analysis. Int Endod J 50(8): 780-789
- I Chen, I Salhab, FC Setzer, S Kim, HD Nah, et al. (2016) A New Calcium Silicate-based Bioceramic Material Promotes Human Osteo- and Odontogenic Stem Cell Proliferation and Survival via the Extracellular Signal-regulated Kinase Signaling Pathway. Journal of endodontics 42(3): 480-486.
- S Utneja, RR Nawal, S Talwar, M Verma (2015) Current perspectives of bio-ceramic technology in endodontics: calcium enriched mixture cement - review of its composition, properties and applications. Restorative dentistry & endodontics 40(1): 1-13.
- Z Wang (2015) Bioceramic materials in endodontics. Endodontic Topics 32(1): 3-30.
- J Ma, Y Shen, S Stojicic, M Haapasalo (2011) Biocompatibility of two novel root repair materials. Journal of endodontics 37(6): 793-798.
- I Tsesis, E Rosen, S Taschieri, Y Telishevsky Strauss, V Ceresoli, et al. (2013) Outcomes of surgical endodontic treatment performed by a modern technique: an updated meta-analysis of the literature. Journal of endodontics 39(3): 332-339.
- SJ Lee, M Monsef, M Torabinejad (1993) Sealing ability of a mineral trioxide aggregate for repair of lateral root perforations. Journal of endodontics 19(11): 541-544.
- G De Deus, A Canabarro, GG Alves, JR Marins, AB Linhares, et al. (2012) Cytocompatibility of the ready-to-use bioceramic putty repair cement iRoot BP Plus with primary human osteoblasts. International endodontic journal 45(6): 508-513.
- PN Nair, HF Duncan, TR Pitt Ford, HU Luder (2008) Histological, ultrastructural and quantitative investigations on the response of healthy human pulps to experimental capping with mineral trioxide aggregate: a randomized controlled trial. International endodontic journal 41(2): 128-150.
- J Mente, S Hufnagel, M Leo, A Michel, H Gehrig, et al. (2014) Treatment outcome of mineral trioxide aggregate or calcium hydroxide direct pulp capping: long-term results. J Endod 40(11): 1746-1751.
- N Wongwatanasanti, J Jantarat, H Sritanaudomchai, KM Hargreaves (2018) Effect of Bioceramic Materials on Proliferation and Odontoblast Differentiation of Human Stem Cells from the Apical Papilla. J Endod 44(8): 1270-1275.
- VD Anto, MP Di Caprio, G Ametrano, M Simeone, S Rengo, et al. (2010) Effect of mineral trioxide aggregate on mesenchymal stem cells. J Endod 36(11): 1839-1843.
- S Shi, DD Zhang, X Chen, ZF Bao, YJ Guo, et al. (2015) Apical sealing ability of bioceramic paste and mineral trioxide aggregate retrofillings: a dye leakage study. Iranian endodontic journal 10(2): 99-103.
- S Alsubait, S Al Haidar, N Al Sharyan (2017) A Comparison of the Discoloration Potential for EndoSequence Bioceramic Root Repair Material Fast Set Putty and ProRoot MTA in Human Teeth: An In Vitro Study. Journal of esthetic and restorative dentistry: official publication of the American Academy of Esthetic Dentistry 29(1): 59-67.
- M Valles, M Mercade, F Duran-Sindreu, JL Bourdelande, M Roig (2013) Color stability of white mineral trioxide aggregate. Clinical oral investigations 17(4): 1155-1159.
- MA Marciano, RM Costa, J Camilleri, RF Mondelli, BM Guimaraes, MA Duarte (2014) Assessment of color stability of white mineral trioxide aggregate angelus and bismuth oxide in contact with tooth structure. Journal of endodontics 40(8): 1235-1240.
- P Kogan, J He, GN Glickman, I Watanabe (2006) The effects of various additives on setting properties of MTA. Journal of endodontics 32(6): 569-572.
- AK Lagisetti, P Hegde, MN Hegde (2018) Evaluation of bioceramics and zirconia-reinforced glass ionomer cement in repair of furcation perforations: An in vitro J Conserv Dent 21(2): 184-189.
- H Beatty, T Svec (2015) Quantifying Coronal Tooth Discoloration Caused by Biodentine and EndoSequence Root Repair Material. Journal of endodontics 41(12): 2036-2039.
- LC Souza, M Yadlapati, SO Dorn, R Silva, A Letra (2015) Analysis of radiopacity, pH and cytotoxicity of a new bioceramic material. Journal of applied oral science: revista FOB 23(4): 383-389.
- SH Kang, YS Shin, HS Lee, SO Kim, Y Shin, et al. (2015) Color changes of teeth after treatment with various mineral trioxide aggregate-based materials: an ex vivo study. Journal of endodontics 41(5): 737-741.
- ML Porter, A Berto, CM Primus, I Watanabe (2010) Physical and chemical properties of new-generation endodontic materials. J Endod 36(3): 524-528.
- JT Washington, E Schneiderman, R Spears, CR Fernandez, J He, et al. (2011) Biocompatibility and osteogenic potential of new generation endodontic materials established by using primary osteoblasts. J Endod 37(8): 1166-1170.
- KC Tay, BA Loushine, C Oxford, R Kapur, CM Primus, et al. (2007) In vitro evaluation of a Ceramicrete-based root-end filling material. J Endod 33(12): 1438-1443.
- MO Daltoe, FW Paula-Silva, LH Faccioli, PM Gaton-Hernandez, A De Rossi, et al. (2016) Expression of Mineralization Markers during Pulp Response to Biodentine and Mineral Trioxide Aggregate. J Endod 42(4): 596-603.
- J Camilleri, F Sorrentino, D Damidot (2013) Investigation of the hydration and bioactivity of radiopacified tricalcium silicate cement, Biodentine and MTA Angelus. Dent Mater 29(5): 580-593.
- G Caron, J Azerad, MO Faure, P Machtou, Y Boucher (2014) Use of a new retrograde filling material (Biodentine) for endodontic surgery: two case reports. Int J Oral Sci 6(4): 250-253.
- J Camilleri (2013) Investigation of Biodentine as dentine replacement material. J Dent 41(7): 600-610.
- P Laurent, J Camps, I About (2012) Biodentine (TM) induces TGF-beta1 release from human pulp cells and early dental pulp mineralization. Int Endod J 45(5): 439-448.
- P Lertmalapong, J Jantarat, RL Srisatjaluk, C Komoltri (2019) Bacterial leakage and marginal adaptation of various bioceramics as apical plug in open apex model. Journal of investigative and clinical dentistry 10(1): e12371.
- F Siboni, P Taddei, C Prati, MG Gandolfi (2017) Properties of NeoMTA Plus and MTA Plus cements for endodontics. International endodontic journal 50 Suppl 2: e83-e94.
- F Leal, G De-Deus, C Brandao, AS Luna, SR Fidel, et al. (2011) Comparison of the root-end seal provided by bioceramic repair cements and White MTA. International endodontic journal 44(7): 662-668.
- WR Hirschman, MA Wheater, JS Bringas, MM Hoen(2012) Cytotoxicity comparison of three current direct pulp-capping agents with a new bioceramic root repair putty. Journal of endodontics 38(3): 385-388.
- NG de Oliveira, PR de Souza Araujo, MT da Silveira, APV Sobral, MV Carvalho (2018) Comparison of the biocompatibility of calcium silicate-based materials to mineral trioxide aggregate: Systematic review. European journal of dentistry 12(2): 317-326.

 Mini Review
Mini Review