Abstract
Introduction: The aim of this study was to explore the association between the BMI Z score and the levels of some selected common nutrients among children under 5 years old with ventricular septal defect (VSD).
Methods: We recruited children under 5 years of age who were diagnosed with VSD from May 2017 to November 2017 at the Children’s Hospital of Nanjing Medical University as research subjects to measure their height/length, weight and serum levels of common nutrients. The nutritional status of the selected children was evaluated by the Z-score method.
Results: A total of 146 children under five years old diagnosed with VSD (78 males, 68 females) were recruited. Iron levels were significantly higher among girls than boys. Pearson correlation analysis showed a negative association between VC and the weight for height Z-score (WHZ) (r=-0.172, P=0.039), and there was a positive correlation between VC and the weight for age Z-score (WAZ) (r=0.247, P=0.003). A comparison of Z scores >0 and <0 also revealed that the VC level in the WHZ>0 group was significantly higher than in the WHZ<0 group (P=0.025). After linear regression analysis with adjusted as age, gender, and pulmonary artery pressure, there was a positive association between WAZ and VC (P=0.035).
Conclusion: Our study found that there may be a positive correlation between VC levels and the nutritional status of children with VSD.
Keywords: Ventricular septal defect; Trace elements; Vitamins; Nutritional status
Abbreviations: vsd: Ventricular Septal Defect; Whz: Weight for Height Z-Score; Nst: Nutrition Support Team; Haz: Height for Age Z-Score; Waz: Weight for Age Z-Score; Va: Vitamin A; Vb1: Vitamin B1; Vb2: Vitamin B2; Vb6: Vitamin B6; Vb9: Vitamin B9; Vb12: Vitamin B12; Vc: Vitamin C; Vd: Vitamin D; Ve: Vitamin E; Cu: Serum Copper; Zn: Zinc; Ca: Calcium; Mg: Magnesium Fe: Iron; eNOS: Endothelial NO Synthase
Introduction
Congenital heart disease refers to anatomical abnormalities of the heart and large blood vessels during the embryonic development period or abnormal heart and blood vessel function caused by no closure of the channels that should be closed after birth. Current epidemiological data [1] show that congenital heart disease accounts for approximately 28% of all major congenital anomalies [2]. On the one hand, due to the progress of society, diagnosis and screening methods have been continuously improving, and thus, increasing numbers of children with congenital heart disease are identified at birth; on the other hand, the process of industrialization and urbanization has changed the environment. Increasing numbers of women have postponed childbearing age, especially in developed countries, leading to a high incidence of congenital abnormalities. The incidence of congenital heart disease is increasing each year, so studies of congenital heart disease are receiving increased attention.
Due to the abnormal hemodynamics caused by congenital heart
disease, children with congenital heart disease often suffer from
hypoxia, hyperthyroidism, frequent respiratory infections, and
poor gastrointestinal absorption [3], and growth retardation and
malnutrition are common in children with congenital heart disease.
At present, the European Society of Pediatric Gastrointestinal
Hepatology Nutrition has called for the establishment of a “nutrition
support team (NST)” aimed at scientifically effective nutritional
management of hospitalized children and reduction of the
prevalence of malnutrition [4]. However, with the trend in younger
children with congenital heart disease becoming increasingly
obvious [1], the earlier the time for surgery is, the more favorable
it is for children to catch up with children of the same age earlier in
their growth and development [5]. Therefore, we should focus our
attention and intervention on the nutritional status of children with
congenital heart disease.
However, clinical interventions for nutrition are mostly focused
on carbohydrates and fats that can provide actual calories, and
the importance of micronutrients is often overlooked. Although
humans need a few vitamins and trace elements, they are the trace
organics necessary for maintaining body health. These substances
neither provide energy sources in the body nor constitute the raw
material of body tissues or participate in the formation of human
cells; however, these substances play a crucial role in the growth,
metabolism, and development of the human body. Due to a lack
of attention to dietary supplements and poor digestive functions
such as malabsorption and diarrhea, children with congenital heart
disease are at risk of these micronutrient deficiencies. In addition,
diuretics are used in children with congenital heart disease.
Diuretics excrete water and sodium through the kidneys to improve
circulatory function and at the same time, excrete other watersoluble
molecules, including potassium, magnesium, and vitamin C
[6]. Studies have shown that early identification of abnormalities
in micronutrients and immunodeficiency can help reduce the
incidence in children after cardiac surgery [7].
It is necessary to carry out studies of the preoperative nutritional
status of children with congenital heart disease, especially infants
and young children, and investigate the correlations between
nutritional status and various common trace elements and vitamins
in our country. Studies have shown that the nutritional status of
children with cyanotic and noncyanotic congenital heart disease is
significantly different: children with noncyanotic congenital heart
disease are more prone to malnutrition [8], and the nutritional
status of these children and its influencing factors deserve
more attention. Ventricular septal defect is the representative
disease type in noncyanotic congenital heart disease. It is the
most common clinical congenital heart disease, accounting for
approximately 20% of congenital heart disease [9]. Therefore, in
order to ensure the representativeness of the research sample and
effectively control for confounding, this study selected ventricular
septal defect as the research disease. This research will study the
preoperative nutritional status, common elements, and vitamin
levels of children with ventricular septal defect younger than 5
years of age and explore the relevance between them, so that we
can provide preventive measures for preoperative malnutrition and
guide postoperative nutritional intervention.
Methods
Population Included
A cross-sectional study was conducted at the Department of Cardiothoracic Surgery of Children’s Hospital of Nanjing Medical University from May 2017 to October 2017. This study comprised 146 children younger than 5 years who were diagnosed with ventricular septal defects by echocardiography and excluded children with other congenital malformations or other diseases (including genetic diseases) affecting growth or nutritional status. The study was approved by the Ethical Committee of Children’s Hospital of Nanjing Medical University, and informed consent was obtained from the guardian of the children.
Nutrition Evaluation Method
Anthropometric measurements included weight (kilograms) and length (centimeters), and they were performed according to standard World Health Organization (WHO) procedures [10]. The Z-score method provided by the National Health Statistics Center and recommended by the WHO was used as the evaluation standard. The Z-score was calculated according to the formula Z-score=(measurement value-median value of the same-age and same-sex reference standard)/standard deviation of the reference standard, including height for age Z-score (HAZ), weight for age Z-score (WAZ), and weight for height Z-score (WHZ). The latest WHO reference standard in 2006 was selected to calculate the Z value [11].
Vitamins and Elements Measurement
Serum levels of vitamin A (VA), vitamin B1 (VB1), vitamin B2 (VB2), vitamin B6 (VB6), vitamin B9 (VB9), vitamin B12 (VB12), vitamin C (VC), vitamin D (VD), and vitamin E (VE) were measured by a BH5100T Whole Blood Multielement Analyzer. Serum copper (Cu), zinc (Zn), calcium (Ca), magnesium (Mg) and iron (Fe) levels were measured by an LK3000V vitamin detector (Tianjin Lanbiao Electronic Technology Development Co., Ltd.).
Data Analysis
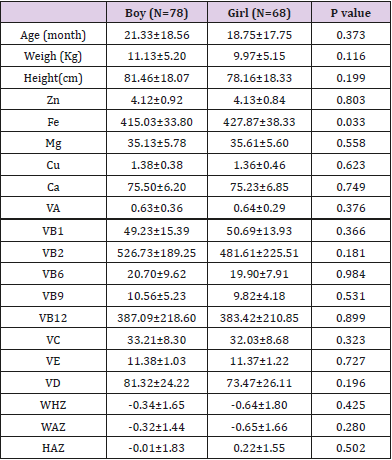
In this study, SPSS 23.0 statistical software was used for statistical analysis, and P<0.05 indicates a significant difference. General information, zinc, iron, magnesium, copper, calcium, VA, VB1, VB2, VB6, VB9, VB12, VC, VE, and VD levels of male and female children are described with the mean plus standard deviation. Mann-Whitney U tests verified whether there was a significant difference between different genders. Pearson bivariate correlation analysis was used to evaluate the correlation between Z scores (e.g., WHZ, WAZ, HAZ) and Zn, Fe, Mg, Cu, Ca, VA, VB1, VB2, VB6, VB9, VB12, VC, VE, and VD. Z-values WHZ, WAZ, and HAZ were grouped by 0, and we compared the levels of Zn, Fe, Mg, Cu, Ca, VA, VB1, VB2, VB6, VB9, VB12, VC, VE, and VD between groups, verified whether they met normality and used an independent t-test for data that satisfied normality. If the data did not satisfy normality, the Mann- Whitney U test was used. Using logistic regression analysis, the confidence interval level was 95%. After correction for age, gender, and pulmonary artery pressure, the ratios of Z-values WHZ, WAZ, HAZ and zinc, iron, magnesium, copper, calcium, VA, VB1, VB2, VB6, VB9, VB12, VC, VE, and VD were analyzed, and the 95% confidence interval was calculated.
Results
Demographic Characteristics of the Participants
There were 68 females and 78 males; for females, the mean age was 18.75±17.75 months, mean weight was 9.97±5.15 kg, and mean height was 78.16±18.33 cm, and for males, the mean age was 21.33±18.56 months, mean weight was 11.13±5.20 kg, and mean height was 81.46±18.07 cm. The iron levels of females were significantly higher than those of males (male vs female: iron, 415.03±33.80 vs 427.87±38.33, P=0.033) (Table 1). However, no statistically significant differences of other vitamins and trace elements were existing in males and females.
Correlation Analysis of Elements and Vitamins and Z Scores
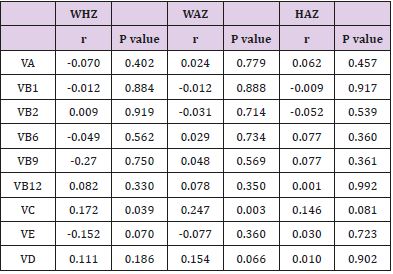
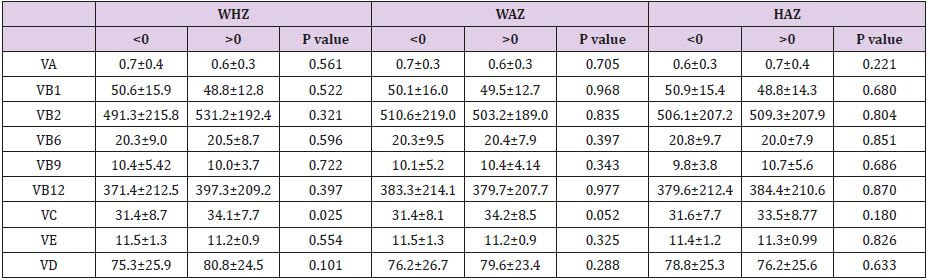
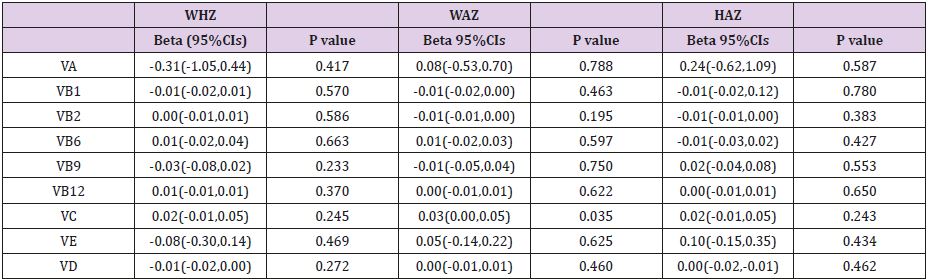
Pearson correlation analysis (Table 2) showed that there was a significant correlation between VC and WHZ (r=-0.172, P=0.039), and there was also a significant correlation between VC and WAZ (r=0.247, P=0.003). While, no significant correlation was found between Zn, Fe, Mg, Cu, Ca levels and the Z-value scores WHZ, WAZ, and HAZ. A comparison of Z scores >0 and <0 showed that VC levels in the WHZ>0 group were significantly higher than those in the WHZ<0 group (P=0.025), as shown in (Table 3). As a result, we conducted a linear regression after adjusting gender and age and found VC was positively associated with between WAZ. The P value was 0.035, as shown in (Figure 1) and (Table 4).
Figure 1: A scatter plot and a fitted line with 95% CI of the relationship between vitamin C and weight for age Z-score in children with ventricular septal defect.
Table 4: Partial vitamin and Z value regression analysis.
Substances are Log10
Corrected gender, age, pulmonary artery pressure.
Discussion
The results of our study revealed a linear regression relationship
between VC and WAZ, with a P value of 0.035, suggesting a possible
correlation between VC and the nutritional status of children with
congenital heart disease. In people with malnutrition caused by
chronic diseases, poor eating habits, malabsorption, and other
factors, obvious VC deficiency can often be seen [12]. Some animal
experiments have shown that VC is an important vitamin for
maintaining normal growth and physiological function [13], playing
an important role in the growth process [9], collagen formation [14,
15], iron metabolism and hematology [9], reproduction [16], stress
response [17], and wound healing [18]. Wang et al. [19]. found that
dietary supplementation with VC can significantly increase the daily
weight of juvenile ducks. A study by Zhou et al. [20] also showed that
increases in body weight, specific growth ratio, protein efficiency
ratio, and feed efficiency of VC-deficient fish were lower than those
of fish supplemented with VC. In addition, growth performance,
antioxidant capacity, and humoral immune parameters of juvenile
cobia supplemented with VC were significantly improved [19, 20].
Heart failure increased pulmonary arterial pressure, and
anemia are risk factors for protein energy malnutrition in children
with congenital heart disease [9], and some physiological functions
of VC can alleviate these risk factors. A considerable amount of
research is currently focused on the use of VC to prevent heart
failure [21], mainly because of its ability to prevent oxidative
damage. Reactive oxygen or nitrogen species (ROS/RNS) are the
most common oxidants in the human body and play a key role
in aggravating human heart failure [9]. Studies have shown that
ROS and RNS can change the properties of lipids, proteins, and
nucleic acids, leading to myocardial dysfunction, and myocardial
oxidative damage is closely related to congestive heart failure [22].
On the other hand, ascorbate regulates vasodilation by increasing
NO synthesis and improving bioavailability [23]. Endothelial NO
synthase (eNOS) produces NO, which then diffuses into the smooth
muscle cell layer of the vascular wall and causes vasodilation
through interaction with soluble guanylate cyclase. Another clinical
application of VC is to increase the absorption of nonheme iron
[12]. In the small intestine, VC can effectively increase dietary iron
transport in intestinal epithelial cells. When anemia patients are
supplemented with iron and VC, the synthesis of hemoglobin can
be promoted. In addition, studies have also shown [24] that a longacting
VC derivative called ascorbic acid 2-phosphate stimulates cell
growth and increases mRNA type III collagen in human osteogenic
MG-63 cells and normal human osteoblasts, as well as expression in
human bone marrow mesenchymal stem cells, thereby stimulating proliferation and differentiation of human osteoblast-like cells.
In summary, we speculate that VC can indirectly influence the
nutritional and growth status of children with congenital heart
disease by exerting its biological functions.
VC is mainly related to WAZ, which can reflect acute malnutrition in patients, and we have previously speculated that some trace elements may be related to HAZ, reflecting chronic malnutrition in patients. However, in our research, no correlation was found between Cu, Fe, Zn, Mg, Ca levels and WHZ, WAZ, HAZ; the reasons need further research. Our findings suggest that there is a correlation between VC and WAZ. WAZ is an indicator of acute malnutrition, reflecting the problem of malnutrition at this stage. It is more marked by factors such as disease, feeding method, food choice, and poor nutritional habits [25]. It can be speculated that supplementation with VC can alleviate acute nutritional problems soon.
Conclusion
This study revealed a positive correlation between VC and the nutritional status of children with ventricular septal defects. Further, our results may have implications from the perspective of VC on the nutritional status of children with clinical congenital heart disease intervention, to prevent malnutrition in children before surgery, while guiding clinical staff to carry out postoperative nutritional support in children with congenital heart disease.
Acknowledgement
This work was supported by funding from The National Key Research and Development Program of China (2017YFC1308105, 2016YFC1101001), the Maternal and Child Health Research Project of Jiangsu Province (F201755), the China Postdoctoral Science Foundation (2018M630585), the Key Project of Science and Technology Development Fund of Nanjing Medical University (2017NJMUZD060, 2017NJMU067), Nanjing Medical University School Project (NMUC2018012A), and Key Project supported by Medical Science and technology development Foundation, Nanjing Department of Health (YKK18139, 201723008).
Competing Interests
The authors declare that they have no competing interests.
Compliance with Ethical Standards
Conflict of Interest
The authors have no conflicts of interest to declare.
Ethical Approval
All procedures performed in study were in accordance with the ethical standards of national research committee and with the 1964 Helsinki Declaration and its later amendments.
Informed Consent
Informed consent was obtained from all guardians of patients included in the study.
References
- (2014) 100 Family Single Column Electromechanical Universal Test Machine. 100 Family Single Column Electromechanical Universal Test Machine. Web.
- Clarke C, Azari P, Huh B (2011) Damaged insulation mimicked symptoms of occipital stimulator lead migration. Neuromodulation 14(6): 539-540.
- Cruccu G, Aziz T, Garcia Larrea L, Hansson P, Jensen T, et al. (2007) EFNS guidelines on neurostimulation therapy for neuropathic pain. European Journal of Neurology 14(9): 952-970.
- Fiume D, Sherkat S, Callovini G, Parziale G, Gazzeri G (1995) Treatment of the failed back surgery syndrome due to lumbo-sacral epidural fibrosis. Advances in stereotactic and functional neurosurgery 11: 116-118.
- Kalia V, Bizzell C, Obray R, Obray J, Lamer T, et al. (2010) Spinal cord stimulation: The types of neurostimulation devices currently being used, and what radiologists need to know when evaluating their appearance on imaging. Current Problems in Diagnostic Radiology 39(5): 227-233.
- Kumar K, Taylor RS, Jacques L, Eldabe S, Meglio M, et al. (2007) Spinal cord stimulation versus conventional medical management for neuropathic pain: A multicentre randomised controlled trial in patients with failed back surgery syndrome. Pain 132(1): 179-188.
- Coffey RJ (2009) Deep brain stimulation devices: A brief technical history and review. Artificial Organs 33(3): 208-220.
- (2010) Medtronic Soletra: Lead Kit for Deep Brain Stimulation. Implant Manual. Metronic, Inc.
- McGrath, J Pain therapies. Medtronic, Inc.
- Peterson JT, Deer TR (2013) A history of neurostimulation. Comprehensive treatment of chronic pain by medical, interventional, and integrative approaches pp. 583-586.
- Eljamel S (2013) Appendix II: Troubleshooting malfunctioning neurostimulators. Neurostimulation: Principles and Practice 222-230.
- Woods DM, Hayek SM, Bedder M (2007) Complications of neurostimulation. Techniques in Regional Anesthesia and Pain Management 11(3): 178- 182.
- Jenney C, Ta, J, Karicherla A, Burke J, Helland J (2005) A new insulation material for cardiac leads with potential for improved performance. Heart Rhythm 2(5): S318-S319.
- Borleffs CJ, Van Erven L, Van Bommel RJ, Van der Velde ET, Van der Wall EE, et al. (2009) Risk of failure of transvenous implantable cardioverter-defibrillator leads. Circulation: Arrhythmia and Electrophysiology 2(4): 411- 416.
- (2010) Medtronic Soletra: Neurostimulator for deep brain stimulation. Implant Manual. Metronic, Inc.
- Mercado L, Carney JK, Ebert MJ, Hareland SA. Bashir R (2009) Digital health and bio-medical packaging. Materials for advanced packaging pp. 681-712.
- Mullett K, Starkebaum W (1994) Spinal cord stimulator: Design and function. Spinal cord stimulation pp. 65-73.
- Nazzaro JM, Lyons KE, Pahwa R, Ridings LW (2011) The importance of testing deep brain stimulation lead impedances before final lead implantation. Surgical Neurology International 2: 131.
- Ondo WG, Meilak C, Vuong KD (2007) Predictors of Battery Life for the Activa Soletra 7426 Neurostimulator. Parkinsonism Related Disorders 13(4): 240-242.
- (2004) Patente US7831311 - Reduced Axial Stiffness Implantable Medical Lead. Google Books.
- Raphael JH, Mutagi H, Hanu Cernat D, Gandimani P, Kapur S (2009) A cadaveric and in vitro controlled comparative investigation of percutaneous spinal cord lead anchoring. Neuromodulation 12(1): 49-53.
- Rosenow JM, Stanton-Hicks M, Rezai AR, Henderson JM (2006) Failure modes of spinal cord stimulation hardware. Journal of Neurosurgery Spine 5(3): 183-190.
- Sarwat AM, Karanth KS, Sutcliffe JC (2000) A rare complication of hardware failure in neurostimulation: Report of two cases. Journal of Neurosurgery Spine 93(2): 330-331.

 Research Article
Research Article