Abstract
Background and Methods: The culprit site of coronary artery occlusion is a major determinant of prognosis in patients with inferior wall acute myocardial infarction (AMI). A two-step strategy was performed to evaluate whether one simple electrocardiographic (ECG) reading can be used to identify the three possible sites of coronary artery occlusion in patients with inferior wall AMI. An initial retrospective study analyzed which ECG criterion was the most appropriate criterion in determining the culprit sites of coronary artery occlusion in 107 patients with inferior wall AMI. Subsequently, a prospective study investigating the usefulness of this criterion in identifying the high risk or proximal right coronary artery (RCA) occlusion patients was assessed in another 110 patients with inferior wall AMI.
Results: The retrospective investigation found that the ratio of ST segment elevation in lead III to lead II (III/II ratio) was the most powerful criterion in discriminating the three culprit sites. The III/II ratio > 1.8 predicted proximal RCA occlusion, III/II ratio between 1.2 and 1.8 predicted distal RCA occlusion, and III/II ratio < 1.2 predicted left circumflex artery occlusion. The second validation study showed that the sensitivity, specificity, and accuracy of identifying the proximal RCA occlusion using III/II ratio > 1.8 was 61%, 86%, and 76%, respectively.
Conclusion: This study demonstrates that a simple ECG criterion (III/II ratio) can be used to identify the 3 culprit sites and the III/II ratio > 1.8 was capable of identifying the high-risk patients in inferior wall AMI.
Keywords: Acute Myocardial Infarction; Coronary Angiography; Culprit Site, Electrocardiography; High Risk
Introduction
Acute myocardial infarction (AMI) usually occurs suddenly and is associated with a considerably high mortality rate. The infarct-related artery in inferior wall AMI is usually located at the right coronary artery (RCA), less often at the left circumflex coronary artery (LCX) [1,2]. Inferior wall AMI occlusive site above the first right ventricular branch of RCA was more frequently associated with right ventricular infarction, which had a higher incidence of bradyarrhythmia, shock, and in-hospital death [3-7]. Early recognition of the site of infarct-related artery, especially in combination with right ventricular infarction may result in a significant reduction in in-hospital mortality and morbidity in the era of primary percutaneous coronary intervention [8-10]. There are several non-invasive methods to predict the culprit site, including radionuclear imaging study, echocardiography, and electrocardiography (ECG). Among these methods, ECG is one of the simplest and most useful tools. Several studies have used ST segment elevation in lead III > ST segment elevation in lead II and ST segment depression in lead I, aVL, or both, to predict that the culprit site was located at the RCA; ST segment elevation in lead I, aVL, V5 and V6, and ST segment depression in V1, V2 and V3 to predict that the culprit site was located at the LCX [11-24]. These ECG readings (including lead III, II, I, aVL, V1, V2, V3, V5 and V6), can only differentiate between RCA and LCX lesions.
However, if clinicians wished to know whether the culprit site is located at the proximal or distal RCA, further evaluation of the ST segment elevation in leads V1 and V4R,would be needed [25], which results in the algorithm involving too many leads and having difficult to remember. The aims of this study were investigating which ECG variable was the most powerful criterion for predicting culprit sites and assessing whether this variable was capable of identifying the high-risk patients in those with inferior wall AMI. A two-step study was performed to achieve these goals. First, previously used criteria, including the ST-segment elevation in each limb lead, ST-segment depression in precordial leads, ratio of STsegment depression in lead V3 to ST-segment elevation in lead III (V3/III ratio), and the ratio of ST-segment elevation in lead III to that of lead II (III/II ratio) were retrospectively studied to assess the relationship between ECG findings and sites of culprit lesions from coronary angiograms. After determining which ECG variables were capable of identifying the culprit site among the three coronary arteries, a receiver operating characteristic (ROC) curve analysis was subsequently performed to analyze which ECG variable was the most powerful criterion for predicting the culprit site of the coronary artery in patients with inferior wall AMI. Afterwards, a prospective study investigating the usefulness of this criterion in identifying the high risk or proximal RCA occlusion was assessed in another 110 patients with inferior wall AMI.
Methods
Study Patients
In the first step, we retrospectively analyzed data from January 2015 to August 2017. Two hundred and thirty-one patients with a discharge diagnosis of AMI were evaluated. Of these, 140 patients suffering from first inferior wall AMI but only 107 patients with symptoms to the ECG examination less than 12 hours were enrolled in this study. The diagnosis of acute inferior AMI includes: 1) chest pain > 30 minutes; 2) ST elevation > 1mm in at least two of the three inferior lead (II, III, AVF); and 3) a 2-fold increase in serum creatine kinase or troponin I levels. At least 2 of the above 3 criteria are necessary to confirm the diagnosis. Right ventricular (RV) infarction was diagnosed by the presence of an ST-segment elevation of 0.1 mV in the V3R or V4R lead (recorded in 89 patients with inferior wall MI) in the ECG performed immediately upon arrival at the emergency department. Exclusion criteria included previous AMI or coronary artery bypass surgery, ECG evidence of bundle branch block, undetermined culprit site by coronary angiography, or the first ECG obtained more than 12 hours after the onset of symptoms. Patients’ demographic variables, important risk factors, and clinical outcomes, including atrioventricular block, arrhythmia, shock, and mortality during the first hospital days were recorded.
All patients received dual anti-platelets with aspirin and clopidegrol, and anti-coagulant (low molecular weight heparin or unfractured heparin) regimen. Most patients received primary percutaneous coronary intervention, and a GpIIb/IIIa antagonist (tirofiban) was administered at 30 minutes before the procedure. In the second step, from September 2017 to October 2018, we prospectively recruited 110 patients who matched the above inclusion criteria. We then studied the sensitivity, specificity, positive and negative predictive values, and accuracy in detecting the culprit sites of the coronary artery in inferior wall AMI patients using the same ECG criteria.
Electrocardiography (ECG)
Standard 12-lead ECG was performed within 12 hours after onset of chest pains in all study patients. ST-segment deviation from the isoelectric line was measured with a caliper at the point of 80ms after the J point. The preceding TP-segment was considered as the isoelectric line. The magnitudes of ST-segment deviation in 12-lead ECG, V3R and V4R were also assessed.
Coronary Angiography
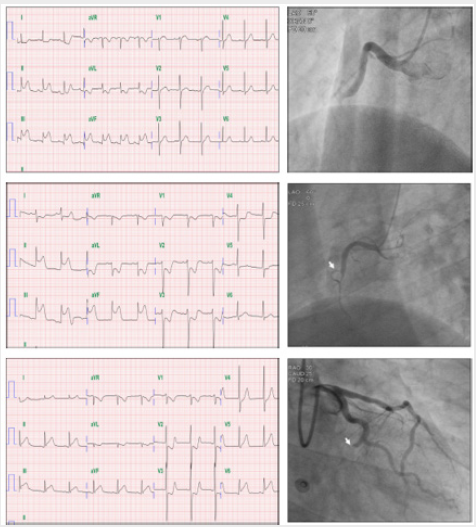
Coronary angiography was performed during the first day of admission. The cineangiography films were reviewed by 2 interventional cardiologists who were blinded to the ECG findings. The culprit artery was defined as total occlusion (Thrombolysis in Myocardial Infarction, TIMI grade 0) or significant stenosis of RCA or LCX and/or of their major branches, with intraluminal thrombosis or supply to hypokinetic territory. The occlusion site of the infarctrelated coronary artery determined by coronary angiography was classified into three groups: proximal RCA: lesion proximal to the first right ventricular branch of the RCA; distal RCA: lesion distal to the first right ventricular branch of the RCA; and LCX. Figure 1 showed the examples of the ECGs and coronary angiograms in patients with proximal RCA, distal RCA, and LCX artery occlusions, respectively.
Figure 1: Showing the examples of electrocardiograms and coronary angiograms in patients with proximal right coronary artery (RCA), distal RCA, and left circumflex coronary artery (LCX) occlusions (Upper, middle, and lower panels) respectively. Upper left panel showed that the magnitude of ST -segment elevation in lead III and II was 2.0 mm and 1.0 mm, respectively. The ratio of III/II is 2.0. Upper right panel was the angiogram of the same patient, which demonstrated that the culprit artery was proximal to first right ventricular branch of RCA. Middle left panel showed that the degree of ST-segment elevation in lead III and II were 3.0 mm and 2.0 mm, respectively. The ratio of III/II is 1.50. Middle right panel showed that the culprit artery was distal to first (white arrow) right ventricular branch of RCA. Lower left panel showed that the extent of ST-segment elevation in lead III and II were 2 mm and 2.5 mm, respectively. The ratio of III/II was 0.8. Lower right panel illustrated that the culprit artery was distal LCX, white arrow point out the occlusive lesion.
Statistical Analysis
Data of ECG findings, patients’ characteristics, and clinical outcomes were presented as mean ± standard deviation for continuous variables and frequency distribution for discrete variables. Chi-square analysis was used to assess the association of patients’ characteristics with sites of culprit lesions. One way ANOVA with Bonferroni post hoc test were used to assess the relation between the sites of culprit lesions and ECG findings. The value of P ≤ 0.05 was considered statistically significant. The cut-off point of the ECG criteria for predicting culprit sites of coronary artery was determined using the receiver operating characteristic (ROC) curve. The diagnostic validity of each cut-off point in predicting the culprit lesions was further tested using ROC curve analysis, by plotting sensitivity against 1-specificity. Two experienced cardiologists (Chou LP and Huang WC) were assigned to a blind measurement of ST segment deviation in all cases. The inter-observer agreement for locating the culprit site of coronary artery using the determined criteria was tested by kappa statistics [26].
Results
Patient Characteristics
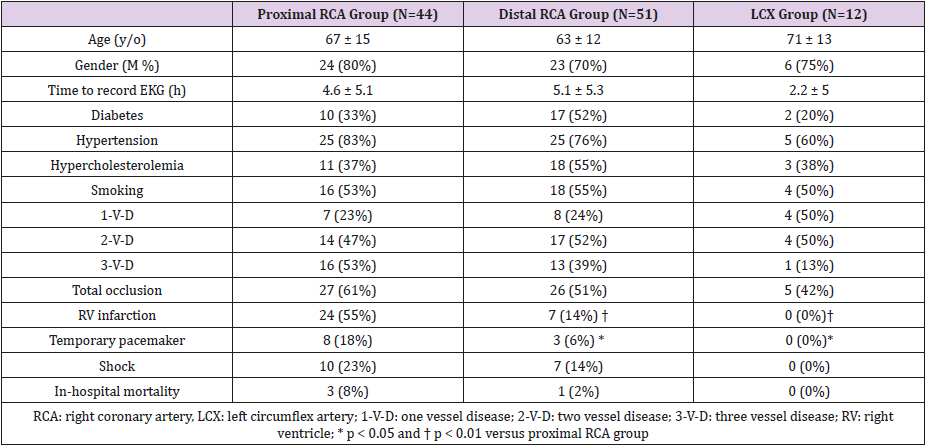
The initially studied 107 patients included 44 patients in the proximal RCA group; 51 in the distal RCA group; and 12 in the LCX group. The patients’ characteristics, vessel numbers of coronary artery disease, and clinical outcomes were summarized in Table 1. There were no significant differences in sex, age, diabetes, hypertension, hypercholesteremia, smoking, duration of chest pain to first ECG, vessel number of coronary diseases. Total occlusions in the infarct related arteries were noted in 58 patients, which included 27 in the proximal RCA group (61%), 26 in the distal RCA group (51%) and 5 in the LCX group (42%). There were no significant differences in the rates of total occlusion among these 3 groups. RV infarction was noted in 31 patients. The proximal RCA group had significantly greater incidences of RV infarction (55% vs. 14%, 0%, p<0.01) and temporary pacemaker implantation (18% vs. 6%, 0%, p<0.05) than the distal RCA and LCX groups. The proximal RCA group also tended to suffer from higher incidences of shock (23% vs. 14%, 0%) and had a greater in-hospital mortality rate (8% vs. 2%, 0%) when compared to the distal RCA and LCX groups.
Relation of The ECG Findings to Culprit Lesions
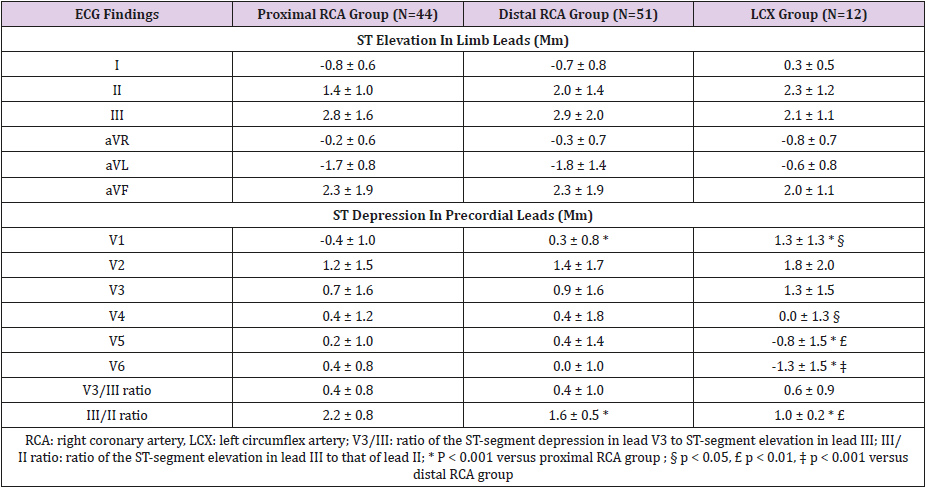
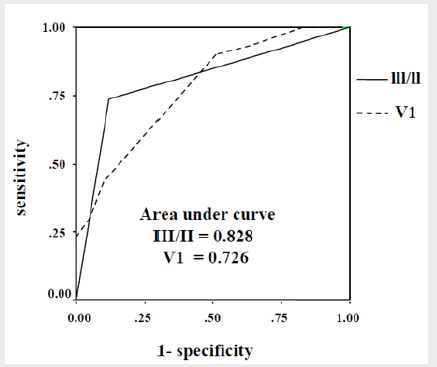
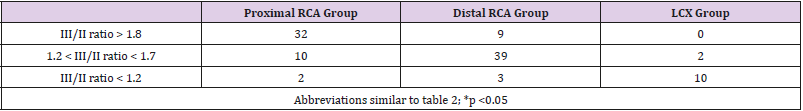
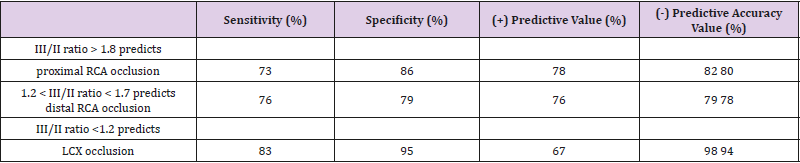
The data of ST- segment deviation in 12-lead ECG were shown in Table 2. Significant differences among the 3 groups were observed for the ST depression in lead V1, and the ratio of ST-segment elevation of lead III to that of lead II (III to II ratio). The value of the III/II ratio was higher in the proximal RCA group (2.2 ± 0.8), followed by the distal RCA group (1.6 ± 0.5), and the LCX group (1.0 ± 0.2). However, the ratio of the ST- segment depression in lead V3 to ST-segment elevation in lead III (V3/III ratio) was not statistically different among these 3 groups. The area under the ROC curve for predicting the infarct-related artery using ST depression in lead V1 was 0.726 (95% CI: 0.630-0.821), while it was 0.828 (95% CI: 0.747-0.910) using III/II ratio (Figure 2). Based on the ROC plot, III/ II ratio > 1.8 was chosen to predict culprit lesions at the proximal RCA, III/II ratio between 1.2 and 1.8 to predict the distal RCA, and III/II ratio < 1.2 to predict the LCX. The relation of III/II ratio to the site of infarct related coronary arteries in the inferior wall AMI was shown in Table 3. The sensitivity, specificity, and positive and negative predictive value, and accuracy of the III/II ratio for predicting the sites of coronary artery occlusions were shown in Table 4. Sensitivity of the III/II ratio for predicting proximal RCA occlusion, distal RCA occlusion, and LCX occlusion was 73%, 76%, and 83%, respectively.
Figure 2: Receiver operating characteristic (ROC) curve analysis for the culprit lesions using the ST depression of lead V1 and ratio of ST-segment elevation of lead III to that of lead II (III/II ratio). The III/II ratio had a greater area under the ROC curve than that of the V1 (0.828 vs. 0.726) in predicting the culprit lesions.
Table 4: Sensitivity, Specificity, Positive and Negative Predictive Value of III/II Ratio for Predicting the Site of Coronary Artery Occlusion.
The specificity for predicting the 3 culprit sites was 86%, 79%, and 95%, respectively. The positive predictive value ranged from 78% (predicting proximal RCA occlusion) to 67% (predicting LCX occlusion), while the highest and lowest negative predictive value was 98% (predicting LCX occlusion) and 79% (predicting distal RCA occlusion), respectively. Accuracy of the III/II ratio for predicting proximal RCA occlusion, distal RCA occlusion, and LCX occlusion was 80%, 78%, and 94%, respectively. Inter-observer agreement for locating the culprit site of the coronary artery using the III/II ratio with a weighted kappa value was 0.80 (95% CI, 0.65-0.94).
Validation of The Criteria
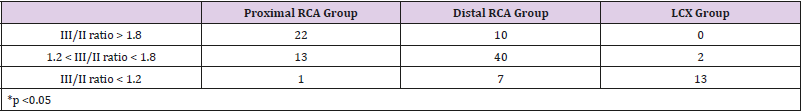
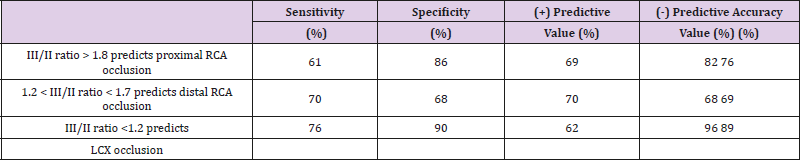
From the initial study we understood that the III/II ratio > 1.8, 1.2 ~ 1.8, and <1.2 could identify the three culprit sites of coronary artery stenosis in patients with inferior wall AMI. We then validated these criteria in assessing the culprit sites of coronary artery stenosis in another group of 110 patients with acute inferior AMI, prospectively. Among these 110 patients with inferior wall AMI, the culprit site was found at the proximal RCA in 36 cases, distal RCA in 57 cases, and LCX in 17 cases. The results of this prospective study revealed that these criteria could detect the three culprit sites. The sensitivity, specificity, (+) and (-) predictive value, and accuracy of identifying the three culprit sites could be refer to Tables 5 and 6, respectively. Furthermore, the sensitivity, specificity, and accuracy of identifying the high risk or proximal RCA occlusion using III/II ratio > 1.8 was 61%, 86%, and 76%, respectively
Table 6: Sensitivity, Specificity, Positive and Negative Predictive Value of III/II Ratio for Predicting the Site of Coronary Artery Occlusion.
Discussion
In this study, we found that inferior wall AMI with culprit lesion from the proximal RCA had the most adverse outcome when compared to the distal RCA and LCX (Table 1). Using the newly developed single ECG criteria (III/II ratio), the second prospective study demonstrated that this criteria not only could differentiate the three culprit sites but also provided an acceptable degree of sensitivity, specificity, and accuracy in quickly identify the high risk patients. Furthermore, this study found that the kappa value (κ) for inter-observer agreement was 0.80, which suggested that our method for locating the culprit sites of coronary artery occlusion using the III/II ratio had a high degree of inter-observer agreement [26]. Some previous studies using various ECG criteria to predict infarct-related coronary artery in inferior wall AMI showed good predictive accuracy [11-22], but such accuracy was not replicated in other reports [23, 24]. The main reason for such discrepancies can be attributed to a heterogeneous inclusion criteria adopted in the earlier studies, including the time from chest pain to first ECG recording, severity of the culprit lesion (total or non-total occlusion), the number of diseased coronary arteries, dominant vessels and so on. With the clearly defined inclusion criteria, our study demonstrated that the “III/II ratio”, can be used to predict the locations of culprit sites of inferior wall AMI patients both in the retrospective and prospective studies.
Kosuge et al. [27] reported the first ECG criteria for predicting the 3 different sites of coronary artery occlusion in inferior wall AMI. They selected the criteria of V3/III ratio < 0.5 to identify proximal RCA occlusion, V3/III ratio > 1.2 to identify LCX occlusion, and that from 0.5 to 1.2 to identify distal RCA occlusion. However, the criteria used in Kosuge and his colleagues’ report were unable to effectively differentiate the 3 groups classified in our patient population (Table 2). Previous reports [11-15,18,23-24], more frequently used ST-segment elevation in lead III than lead II (or lead III > II) as an indicator of infarct-related coronary artery at the RCA. One recent meta-analysis also demonstrated that ST segment elevation in lead III > II provided better prediction of right coronary artery as the culprit artery in inferior myocardial infarction [28]. However, they have not described whether their variable could be used in differentiating the proximal versus distal RCA occlusion. In our study, we further demonstrated that the value of both lead III/II ratio and ST depression in V1 can differentiate the 3 groups of patients who presented with proximal and distal RCA and LCX arteries as the infarct-related coronary artery (Table 2). Moreover, we noticed that the “area under the ROC curve” was greater in lead III/II ratio than that of ST depression in V1 (0.828 versus 0.726). Thus, we found that this lead III/II ratio is a better and simple ECG criterion that can be used to discriminate the three culprit sites of the coronary artery in inferior wall AMI.
Previous report had claimed that the ST segment elevation 1 mm in lead V4R was a reliable sign of right ventricle (RV) infarction. RV infarction usually reflected the presence of proximal RCA occlusion [29,30]. However, ST segment elevation in the right precordial leads was short lived, having disappeared within 10 hours after the onset of chest pain in half of inferior AMI patients with RV involvement [30]. Unless the ECG was studied early enough, the detecting rate of RV involvement using ST segment elevation at V4R may be underestimated. In our initial retrospective study, 89 patients had received V4R examinations, the percentage of RV infarction was presented in 31 (35%) of these 89 patients. Our data was consistent with previous article which reported that about 25% to 50% of inferior wall AMI patients had a RV infarction in their series [31]. Since the goal of the second step prospective study was to address the effectiveness of using the determined criterion (or the III/II ratio >1.8) in identifying the high-risk patients, we had not analyzed the data relevant to the ST segment deviation in lead V4R. Hamon et al. [32] reported a meta-analysis which included 22 relevant studies involving a total of 7,136 patients with AMI at baseline, of whom 1,936 (27.5%) had right ventricular infarction. An overall pooled relative risk increase of 2.59 (95% confidence interval, 2.02-3.31) was found. Another study from Assali et al. [33], after adjustment with CALLIAC score, had an odds ratio for 30-day morbidity of 5.2 (95% confidence interval 1.6-17, P = 0.005) for patients with right ventricular infarction versus those without right ventricular infarction.
Within the group of patients with right ventricular infarction, complete revascularization of the right coronary artery, including the major right ventricular branch, was associated with higher rates of right ventricular function recovery by echocardiography and improved 30-day mortality rates (odds ratio 0.4, 95% confidence interval 0.1-1.05, P = 0.06). Our analysis, although of a relatively small patient population, also demonstrated that proximal RCA as infarct-related coronary artery in inferior wall AMI had the worst clinical outcome. In other words, this group patients had significantly greater incidences of RV infarction, severe bradycardia, shock, and in-hospital mortality rates than the other 2 groups. (Table 1) The patients in the LCX group as infarct-related coronary artery has the best clinical outcome, and the distal RCA group had an outcome status midway between the other two. Thus, early identification of the site of infarct-related coronary artery in inferior wall AMI is very important at the time of primary angioplasty for AMI, which can facilitate properly choosing the guiding catheter and shortening the intervention time.
Several limitations of this study should be mentioned. First, we intended to extend our clinical applications, so we did not restrict these patients with multivessel disease, total or subtotal occlusion, or dominant RCA or LCX vessels. Although it is clinically more practical, it may underestimate the accuracy of prediction as compared to findings from previous studies. Second, the measurement of ECG deviations of S-T segment by manual method can produce some unavoidable errors, including quality of recording and visual difference between observers. However, our results showed substantial agreement in classification of occlusive sites (the weighted kappa value was 0.80), which means that our measurement was reliable. Nevertheless, newly developed computer-assisted measurement will further reduce this bias. Third, this study enrolled relatively a small number of patients, a larger prospective study to validate this criterion may be needed.
Conclusion
Given its non-invasiveness and easy-to-use, we conclude that the simple ECG criterion (III/II ratio) can be used to identify the 3 culprit sites and rapidly recognizes the high-risk cases in patients with inferior wall AMI, which is useful in the decision making.
Acknowledgement
This study was supported by Tainan Sinlau Christian Hospital, Grant No.: SLH09518, and Yuan’s General Hospital, Grant No.: YGH17 – 010.
References
- Blanke H, Cohen M, Schlueter GU, Karsch KR, Rentrop KP, et al. (1984) Electrocardiographic and coronary arteriographic correlations during acute myocardial infarction. Am J Cardiol 54(3): 249-55.
- Berger PB, Ryan TJ (1990) Inferior myocardial infarction: high risk subgroups. Circulation 81: 401-411.
- Braat SH, Brugada P, den Dulk K, van Ommen V, Wellens HJ, et al. (1984) Value of lead V4R for recognition of the infarct coronary artery in acute inferior myocardial infarction. Am J Cardiol 53(11): 1538-1541.
- Braat SH, de Zwaan C, Brugada P, Coenegracht JM, Wellens HJ, et al. (1984) Right ventricular involvement with acute inferior wall myocardial infarction identifies high risk of developing atrioventricular nodal conduction disturbances. Am Heart J 107(6): 1183-1187.
- Bueno H, Lopez-Palop R, Bermejo J, Lopez-Sendon JL, Delcan JL, et al. (1997) In-hospital outcome of elderly patients with acute inferior myocardial infarction and right ventricular involvement. Circulation 96: 436-441.
- Bueno H, Lopez-Palop R, Bermejo J, Lopez-Sendon JL, Delcan JL, et al. (1998) Combined effects of age and right ventricular involvement on acute inferior myocardial infarction prognosis. Circulation 98: 1714- 1720.
- Mehta SR, Eikelboom JW, Natarajan MK, Diaz R, Yi C, et al. (2001) Impact of right ventricular involvement on mortality and morbidity patients with inferior myocardial infarction. J Am Coll Cardiol 37(1): 37-43.
- Assali AR, Teplitsky I, Ben-Dor I, Solodky I, Brosh D, et al. (2007) Prognostic importance of right ventricular infarction in an acute myocardial infarction cohort referred for contemporary percutaneous reperfusion therapy. Am Heart J 153(2): 231-237.
- Hamon M, Agostini D, Page OL, Riddell JW, Hamon M, et al. (2008) Prognostic impact of right ventricular involvement in patients with acute myocardial infarction: meta-analysis. Crit Care Med 36(7): 2023-2033.
- Owens CG, McClelland AJ, Walsh SJ, Smith BA, Stevanson M, et al. (2009) In-hospital percutaneous coronary intervention improves in-hospital survival in patients with acute inferior myocardial infarction particularly with right ventricular involvement. J Invasive Cardiol 21 (2): 40-44.
- Assali AR, Herz I, Vaturi M, Adler Y, Solodky A, et al. (1999) Electrocardiographic criteria for predicting the culprit artery in inferior wall acute myocardial infarction. Am J Cardiol 84(1): 87-89.
- Herz I, Assali AR, Alder Y, Solodky A, Sclarovsky S. et al. (1997) New electrocardiographic criteria for predicting either the right or left circumflex artery as the culprit coronary artery in inferior wall acute myocardial infarction. Am J Cardiol 80(10): 1343-1345.
- Zimetbaum PJ, Josephson ME (2003) Use of the electrocardiogram in acute myocardial infarction. N Engl J Med 348: 933-940.
- Bayram E, Atalay C (2004) Identification of the culprit artery involved in inferior wall acute myocardial infarction using electrocardiographic criteria. J Inter Med Res 32(1): 39-44.
- Fiol M, Cygankiewicz I, Carrillo A, Bayes-Genis A, Santoyo O, et al. (2004) Value of electrocardiographic algorithm based on “ups and downs” of ST in assessment of a culprit artery in evolving inferior wall acute myocardial infarction. Am J Cardiol 94(6): 709-714.
- Birnbaum Y, Sclarovasky S, Mager A, Sirasberg B, Rechavia E. et al. (1993) ST segment depression in aVL: a sensitive marker for acute inferior myocardial infarction. Eur Heart J 14(1): 4-7.
- Kontos MC, Desai PV, Jesse RL, Ornato JP (1997) Usefulness of the admission electrocardiogram for identifying the infarct-related artery in inferior wall acute myocardial infarction. Am J Cardiol 79(2): 182-184.
- Kabakci G, Yildirir A, Yildiran L, Batur MK, Cagrikul R, et al. (2001) The diagnostic value of 12-lead electrocardiogram in predicting infarctrelated artery and right ventricular involvement in acute inferior myocardial infarction. A N E 6(3):229-235.
- Wong TW, Huang XH, Liu W, Ng K, Ng KS, et al. (2004) New electrocardiographic criteria for identifying the culprit artery in inferior wall acute myocardial infaction-usefulness of T-wave amplitude ration in Leads II/III and T-wave polarity in the right V5 lead. Am J Cardiol 94(9): 1168-71.
- Hasdai D, Birnbaum Y, Herz I, Sclarovsky S, Mazur A, et al. (1995) ST-segment depression in lateral limb leads in inferior wall acute myocardial infarction: implications regarding the culprit artery and the site of obstruction. Eur Heart J 16(11): 1549-1553.
- Bairey CN, Shah PK, Lew AS, Hulse S (1987) Electrocardiographic differientiation of occlusion of the left circumflex versus the right coronary artery as a cause of inferior acute myocardial infarction. Am J Cardiol 60(7): 456-459.
- Huey BL, Beller GA, Kaiser DL, Gibson RS (1998) A comprehensive analysis of myocardial infarction due to left circumflex artery occlusion: comparison with infarction due to right coronary artery and left anterior descending artery occlusion. J Am Coll Cardiol 12(5): 1156-1166.
- Chia BL, Yip JWL, Tan HC, Lim YT (2000) Usefulness of ST elevation II/ III ratio and ST deviation in lead I for identifying the culprit artery in inferior wall acute myocardial infarction. Am J Cardiol 86(3): 341-343.
- Nair R, Glancy DL (2002) ECG discrimination between right and left circumflex coronary arterial occlusion in patients with acute inferior myocardial infarction: value of old criteria and use of lead aVR. Chest 122(1): 134-139.
- Witters PGJ, Cools FJ (2006) Evaluation of electrocardiographic algorithms in the assessment of the infarct-related artery in acute myocardial infarction. Acta Cardiol 61(4): 446-453.
- Landis JR, Koch GG (1977) The measurement of observer agreement for categorical data. Biometrics 33(1): 159-174.
- Kosuge M, Kimura K, Ishikawa T, Hongo Y, Mochida Y, et al. (1998) New electrocardiographic criteria for predicting the site of coronary artery occlusion in inferior wall acute myocardial infarction. Am J Cardiol 82(11): 1318-1322.
- Liang H, Wu L, Li Y, Zeng Y, Hu Z, et al. (2018) Electrocardiogram criteria of limb leads predicting right coronary artery as culprit artery in inferior wall myocardial infarction: A meta-analysis. Medicine 97(24).
- Croft CH, Nicod P, Corbett JR, Lewis SE, Huxley R, et al. (1982) Detection of acute right ventricular infarction by right precordial electrocardiography. Am J Cardiol 50(3): 421-427.
- Braat SH, Brugada P, de Zwaan C, Coenegracht JM, Wellens HJ, et al. (1983) Value of electrocardiogram in diagnosing right ventricular involvement in patients with an acute inferior wall myocardial infarction. Br Heart J 49(4): 368-372.
- Kakouros N, Cokkinos DV (2010) Right ventricular myocardial infarction: pathophysiology, diagnosis, and management. Postgrad Med J 86(1022):719-728.
- Hamon M, Agostini D, Page OL, Riddell JW, Hamon M, et al. (2008) Prognostic impact of right ventricular involvement in patients with acute myocardial infarction: meta-analysis. Crit Care Med 36(7): 2023-2033.
- Assali AR, Teplitsky I, Ben-Dor I, Solodky I, Brosh D, et al. (2007) Prognostic importance of right ventricular infarction in an acute myocardial infarction cohort referred for contemporary percutaneous reperfusion therapy. Am Heart J 153(2): 231-237.

 Research Article
Research Article