Abstract
Ovarian cancer has been identified as the most common cause of gynecological cancer-related death, and is the 5th leading cause of cancer-associated mortality worldwide amongst women. While there are numerous front-line agents currently being used in chemotherapy to treat ovarian cancer such as paclitaxel and doxorubicin, much more improved chemotherapeutics are desperately needed to both improve overall drug efficacy, as well as to minimize negative unintended side-effects. The use of nanocarriers such as micelles has been shown to be quite effective with respect to addressing both of these factors, which would explain the clinical success of several nanocarrier-based drug formulations currently used to treat various types of cancer. Therefore, it is not surprising that many groups are currently working on further improving these types of nanocarrier-based chemotherapeutics through the addition of targeting ligands in order to improve colocalization between the nanocarrier containing the encapsulated cytotoxic agent and cancer cells. Due in part to the specificity of antibodies for their intended upregulated cell surface receptors present on various types of cancers, one such formulation involves decorating micelles via surface- modification with antibodies or antibody fragments to generate immunomicelles, and various promising constructs have recently been reported. Here we present some of these recently reported immunomicellar- based drug formulations intended to treat ovarian cancer
Keywords: Ovarian Cancer; Micelles; Immunomicelles; Chemotherapy; Nanocarriers; Targeted Drug Delivery.
Abbreviations: IV: Intravenous; EPR: Enhanced Permeability and Retention; MAB 2C5: Monoclonal 2C5 Antibody; CDK: Cyclin-Dependent Kinase; CD 147ab: CD147 Antibody; HER2: Human Epidermal growth factor receptor 2; SIRNA: Short Interfering RNA; Bcl-xL: B-cell Lymphoma-Extra Large; STAT3, Signal Transducer and Activator of Transcription 3; MUC 16: Mucin 16.
Introduction
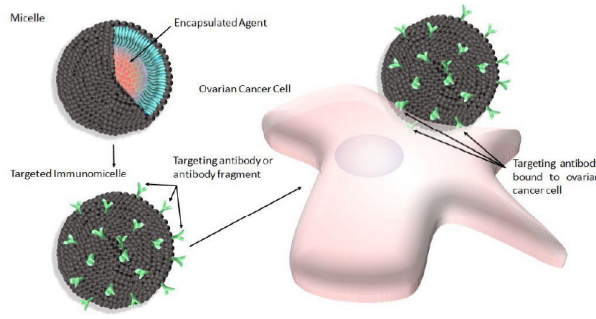
The use of nannocarriers in general for the delivery of cytotoxic agents to solid tumors is ideal as they have been shown to improve the overall pharmacological properties of otherwise low-molecular weight “free drugs”, or unencapsulated cytotoxic agents by altering drug pharmacokinetics and biodistribution/bioavailability [1]. There are numerous types of nanocarriers available for such delivery to include dendrimers, liposomes, nanoparticles made of various materials (i.e. ceramic nanoparticles), as well as micelles to name a few. While all of these nanocarriers have been used with varying levels of success in the past, micelles are particularly attractive for use as a drug delivery system for a number of reasons. For example, micelles used for this purpose are by definition selfaggregates of biocompatible surfactant molecules dispersed in a liquid colloid containing a hydrophobic core (Figure 1, upper left). Thus, they are ideal for the encapsulation of hydrophobic drugs, which most chemotherapeutics tend to be, thereby eliminating the need for potentially toxic solubilizing agents such as Cremophor EL during intravenous (i.v.) administration [2].
Figure 1: Shematic depicting micelles (upper left) modified with antibody or antibody fragments to produce targeted immunomicelles (lower left) intended to target and bind specific overexpressed cell surface receptors present on ovarian cancer cells (right).
Furthermore, the clinical success of drugs such as GenexolPM, which is produced by the Samyang Company in South Korea and is clinically approved in that country [3,4], proves that these nanocarriers can in fact be very effective in the treatment of solid tumors. This micellar-based drug formulation contains the hydrophobic mitotic inhibitor cytotoxic agent paclitaxel. It should be noted however, that clinically-approved micellar-based drugs such as Genexol-PM delivers encapsulated cytotoxic agents to solid tumors based on a “passive” form of delivery facilitated by the enhanced permeability and retention (EPR) effect [5,6], and future work intended to improve the overall efficacy of micellar-based drugs involve “active” delivery of encapsulated drugs. Generally, this latter type of delivery involves the addition of a targeting ligand to the drug formulation that is specific to certain identified upregulated cell surface receptors known to be present on various cancer cells in order to improve drug/cancer cell colocalization.
An additional distinct advantage of using nanocarriers such as micelles for this type of delivery is the ease with which they can be surface-modified to accommodate such targeting ligands, thereby eliminating the need for direct conjugation between the targeting ligand and the drug, which can potentially negatively alter the cytotoxicity of the drug [7,8]. Various targeting ligands have been used in order to confer targeting capabilities to micelles, and have recently been reported with varying levels of success. For example, small molecules including vitamins, carbohydrates, peptides and proteins have all been used to generate targeted micelles intended to treat various types of cancers [9-12]. However, due to the fact that specificity between the targeting ligand and intended upregulated cell surface receptor is a very important consideration when selecting a targeting ligand, many groups have elected to use antibodies or antibody fragments to generate immunomicelles (Figure 1, lower left). Furthermore, ovarian cancer in particular has specifically been the focus of many recent studies involving immunomicelles as the clinically approved micellar-based drug Genexol-PM contains encapsulated paclitaxel [13], which is known to be a standard chemotherapeutic front-line agent for the treatment of ovarian cancer [14,15]. Furthermore, paclitaxel, along with other drugs such as the antineoplastic drug doxorubicin which is also a front-line agent used in the treatment of ovarian cancer [16], are drugs that are very commonly-used in micellar based formulations. Additionally, as previously mentioned, ovarian cancer is the most common cause of gynecological cancerrelated death and one of the leading overall causes of cancerassociated mortalities amongst women worldwide [17,18], and therefore much more efficacious drugs are desperately needed with respect to this type of cancer. These future drugs may in fact involve immunomicelles, and while we recognize that it is not possible to present every immunomicellar-based drug preparation intended for the treatment of ovarian cancer considering this large and rapidly growing field, rather here we present some of the more recently reported promising formulations.
Immunomicelles Intended to Target Ovarian Cancer
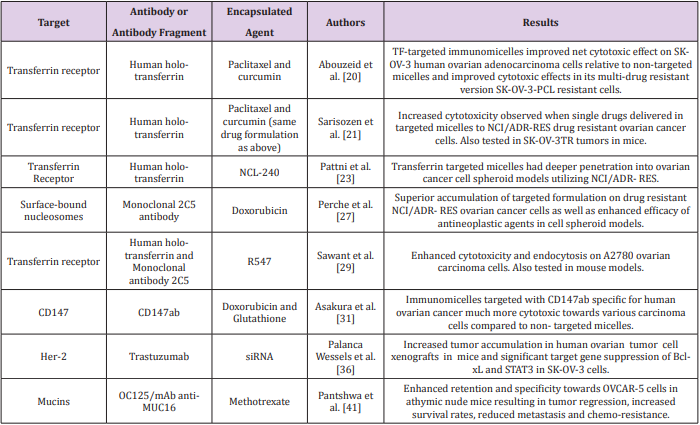
In general, antibodies would seem to be ideal targeting ligands due to their highly specific nature. However, there are several complications associated with the use of full antibodies as targeting ligands in drug formulations to include immunogenicity resulting in rapid elimination and low circulation times in vivo [19]. Therefore in some cases, antibody fragments are used instead of full antibodies in order to generate a less immunogenic drug formulation. In any event, the advantage of using antibodies or antibody fragments as targeting ligands to generate immunomicelles specific for ovarian cancer (Figure 1, right) can clearly be seen in the literature, and many recent reports seem to be very promising (Table 1). For example, Abouzeid et al. [20,21] have recently reported a drug formulation involving holo-transferrin-immunomicelles containing encapsulated paclitaxel and curcumin, which are specific for the overexpressed transferrin receptor [20].
In this study, immunomicelles decorated with CD147ab proved to be much more cytotoxic towards various carcinoma cell lines compared to non-targeted micelles. Human epidermal growth factor receptor 2 (HER2) is yet another known potential target for targeted chemotherapeutics due to its overexpression in various types of cancers, resulting in increased tumor aggressiveness and a relatively poor prognosis [33,34]. However, the frequency of positive HER2 overexpression in ovarian cancer is quite variable [35], and therefore it is unclear as to how widely used HER2 targeted drugs would be amongst ovarian cancer patients in particular. Nonetheless, such a drug formulation would potentially be of great benefit to ovarian cancer patients with positive HER2 overexpression. In fact, Palanca Wessels et al. [36] have recently reported immunomicelles decorated with trastuzumab, which is a targeting ligand specific for HER2, containing encapsulated short interfering RNA (siRNA) [36].
Increased tumor accumulation in human ovarian tumor cell xenographs in mice and significant target gene suppression of Bcl-xL and STAT3 in SK-OV-3 ovarian cancer cells was observed when the targeted formulation was used compared to its nontargeted counterpart. These two genes in particular were chosen in this study as Bcl-xL gene expression allows cancer cells to escape apoptotic signals resulting from treatment, and STAT3 mediates the expression of various genes involved in cellular survival function [37,38]. Interestingly, STAT3 can be activated by MUC16, which is a transmembrane glycoprotein mucin that has been shown to be overexpressed in ovarian cancer, and therefore it is also a potential target in targeted drug delivery [39,40]. In fact, Pantshwa et al. [41] have recently reported a drug formulation involving mAb antiMUC16 immunomicelles containing encapsulated methotrexate to treat ovarian cancer cells [41]. In this study, the targeted formulation resulted in tumor regression, increased survival rates, reduced metastasis, and reduced chemoresistance compared to the non-targeted system when tested in mice.
Conclusion
While there are many types of targeting ligands that can in theory be used along with micellar-based drugs, antibodies or antibody fragments are particularly attractive due in part to their specificity for the various known upregulated/overexpressed cell surface receptors present on ovarian cancer cells. This combination of using micelles decorated with antibodies or antibody fragments to form immunomicelles, designed to treat ovarian cancer specifically in this case, have been shown to be quite promising and many recently reported drug formulations have been described here. While it is not possible to describe every immunomicellarbased drug formulation intended for the treatment of ovarian cancer, rather here we report some promising drug formulations involving immunomicelles based in part on the encouraging results recently reported in the literature.
Acknowledgement
This work was supported by funds generously provided by the Welch Foundation (grant # AE-0025), the Ross Wilson Organization, as well as the Killgore Research Center through the Research Enhancement and Killgore Research grant program at West Texas A&M University.
References
- Allen TM, Cullis PR (2004) Drug delivery systems: Entering the mainstream. Science 303(5665): 1818- 1822.
- Gelderblom H, Verweij J, Nooter K, Sparreboom A (2001) Cremophor EL: the drawbacks and advantages of vehicle selection for drug formulation. Eur J Cancer 37(13): 1590-1598.
- Fan Z, Chen C, Pang X, Yu Z, Qi Y, et al. (2015) Adding vitamin E-TPGS to the formulation of Genexol-PM: specially mixed micelles improve drug-loading ability and cytotoxicity against multidrug-resistant tumors significantly. PloS one 10(4): e0120129.
- Werner ME, Cummings ND, Sethi M, Wang EC, Sukumar R, et al. (2013) Preclinical evaluation of Genexol-PM, a nanoparticle formulation of paclitaxel, as a novel radiosensitizer for the treatment of non-small cell lung cancer. Int J Radiat Oncol Biol Phys 86(3): 463-468.
- Matsumura Y, Maeda H (1986) A new concept for macromolecular therapeutics in cancer chemotherapy: mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs. Cancer Res 46(12 Pt 1): 6387-6392.
- Khan DR, Yarbrough JC, Woodyard JD, Phelps SM (2018) Recent Advancements Involving Immunoliposomes to Target Breast Cancer. Journal of Cancer Science and Clinical Oncology 5(2): 203.
- Yu B, Tai HC, Xue W, Lee LJ, Lee RJ (2010) Receptor-targeted nanocarriers for therapeutic delivery to cancer. Molecular membrane biology 27(7): 286-298.
- Allen TM (2002) Ligand-targeted therapeutics in anticancer therapy. Nat Rev Cancer 2(10): 750-763.
- Muthu MS, Kutty RV, Luo Z, Xie J, Feng SS (2015) Theranostic vitamin E TPGS micelles of transferrin conjugation for targeted co-delivery of docetaxel and ultra bright gold nanoclusters. Biomaterials 39: 234-248.
- Lin WJ, Lee WC, Shieh MJ (2017) Hyaluronic acid conjugated micelles possessing CD44 targeting potential for gene delivery. Carbohydrate polymers 155: 101-108.
- Ran D, Mao J, Shen Q, Xie C, Zhan C, et al. (2017) GRP78 enabled micellebased glioma targeted drug delivery. J Control Release 255: 120-131.
- Nicolas J, Mura S, Brambilla D, Mackiewicz N, Couvreur P (2013) Design, functionalization strategies and biomedical applications of targeted biodegradable/biocompatible polymer-based nanocarriers for drug delivery. Chemical Society reviews 42(3): 1147-1235.
- Keam B, Lee KW, Lee SH, Kim JS, Kim JH, et al. (2019) A Phase II Study of Genexol-PM and Cisplatin as Induction Chemotherapy in Locally Advanced Head and Neck Squamous Cell Carcinoma. oncologist.
- Kumar S, Mahdi H, Bryant C, Shah JP, Garg G, et al. (2010) Clinical trials and progress with paclitaxel in ovarian cancer. International journal of women’s health 2: 411-427.
- Kampan NC, Madondo MT, McNally OM, Quinn M, Plebanski M (2015) Paclitaxel and Its Evolving Role in the Management of Ovarian Cancer. BioMed research international 2015: 413076.
- Cagel M, Bernabeu E, Gonzalez L, Lagomarsino E, Zubillaga M, et al. (2017) Mixed micelles for encapsulation of doxorubicin with enhanced in vitro cytotoxicity on breast and ovarian cancer cell lines versus Doxil((R)). Biomedicine & pharmacotherapy 95: 894-903.
- Jayson GC, Kohn EC, Kitchener HC, Ledermann JA (2014) Ovarian cancer. Lancet 384(9951): 1376- 1388.
- Sapiezynski J, Taratula O, Rodriguez Rodriguez L, Minko T (2016) Precision targeted therapy of ovarian cancer. J Control Release 243: 250- 268.
- Richards DA, Maruani A, Chudasama V (2017) Antibody fragments as nanoparticle targeting ligands: a step in the right direction. Chem Sci 8(1): 63-77.
- Abouzeid AH, Patel NR, Sarisozen C, Torchilin VP (2014) Transferrintargeted polymeric micelles co-loaded with curcumin and paclitaxel: efficient killing of paclitaxel-resistant cancer cells. Pharm Res 31(8): 1938-1945.
- Sarisozen C, Abouzeid AH, Torchilin VP (2014) The effect of co-delivery of paclitaxel and curcumin by transferrin-targeted PEG-PE-based mixed micelles on resistant ovarian cancer in 3-D spheroids and in vivo tumors. Eur J Pharm Biopharm 88(2): 539-550.
- Daniels TR, Bernabeu E, Rodriguez JA, Patel S, Kozman M, et al. (2012) The transferrin receptor and the targeted delivery of therapeutic agents against cancer. Biochim Biophys Acta 1820(3): 291-317.
- Pattni BS, Nagelli SG, Aryasomayajula B, Deshpande PP, Kulkarni A, et al. (2016) Targeting of Micelles and Liposomes Loaded with the ProApoptotic Drug, NCL-240, into NCI/ADR-RES Cells in a 3D Spheroid Model. Pharm Res 33(10): 2540-2551.
- Pattni BS, Jhaveri A, Dutta I, Baleja JD, Degterev A, et al. (2017) Targeting energy metabolism of cancer cells: Combined administration of NCL240 and 2-DG. Int J Pharm 532(1): 149-156.
- Khan DR, Favela SJ, Muniz AE, Blakeman AN (2013) Recent Patents on Common Modifications Made to Traditional Micellar-Based Chemotherapeutics Designed to Improve Drug Delivery. Recent Patents on Nanomedicine 3(1): 21-25.
- Torchilin VP, Lukyanov AN, Gao Z (2006) Micelle Delivery System Loaded with a Pharmaceutical Agent. US.
- Perche F, Patel NR, Torchilin VP (2012) Accumulation and toxicity of antibody-targeted doxorubicin- loaded PEG-PE micelles in ovarian cancer cell spheroid model. J Control Release 164(1): 95-102.
- Elbayoumi TA, Torchilin VP (2008) Tumor-specific antibody-mediated targeted delivery of Doxil reduces the manifestation of auricular erythema side effect in mice. Int J Pharm 357(1-2): 272-279.
- Sawant RR, Jhaveri AM, Koshkaryev A, Zhu L, Qureshi F, et al. (2014) Targeted transferrin-modified polymeric micelles: enhanced efficacy in vitro and in vivo in ovarian carcinoma. Molecular pharmaceutics 11(2): 375-381.
- DePinto W, Chu XJ, Yin X, Smith M, Packman K, et al. (2006) In vitro and in vivo activity of R547: a potent and selective cyclin-dependent kinase inhibitor currently in phase I clinical trials. Mol Cancer Ther 5(11): 2644-2658.
- Asakura T, Yokoyama M, Shiraishi K, Aoki K, Ohkawa K (2018) Chemotherapeutic Effect of CD147 Antibody-labeled Micelles Encapsulating Doxorubicin Conjugate Targeting CD147-Expressing Carcinoma Cells. Anticancer research 38(3): 1311-1316.
- Yang H, Zou W, Chen B (2013) Overexpression of CD147 in ovarian cancer is initiated by the hypoxic microenvironment. Cell Biol Int 37(10): 1139-1142.
- Stewart J, James J, McCluggage GW, McQuaid S, Arthur K, et al. (2015) Analysis of wntless (WLS) expression in gastric, ovarian, and breast cancers reveals a strong association with HER2 overexpression. Mod Pathol 28(3): 428-436.
- Slamon DJ, Leyland Jones B, Shak S, Fuchs H, Paton V, et al. (2001) Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. The New England journal of medicine 344(11): 783-792.
- Serrano Olvera A, Duenas Gonzalez A, Gallardo Rincon D, Candelaria M, De la Garza Salazar J (2006) Prognostic, predictive and therapeutic implications of HER2 in invasive epithelial ovarian cancer. Cancer treatment reviews 32(3): 180-190.
- Palanca Wessels MC, Booth GC, Convertine AJ, Lundy BB, Berguig GY, et al. (2016) Antibody targeting facilitates effective intratumoral siRNA nanoparticle delivery to HER2-overexpressing cancer cells. Oncotarget 7(8): 9561-9575.
- Cardenas C, Montagna MK, Pitruzzello M, Lima E, Mor G, et al. (2017) Adipocyte microenvironment promotes Bclxl expression and confers chemoresistance in ovarian cancer cells. Apoptosis: an international. journal on programmed cell death 22(4): 558-569.
- Hirano T, Ishihara K, Hibi M (2000) Roles of STAT3 in mediating the cell growth, differentiation and survival signals relayed through the IL-6 family of cytokine receptors. Oncogene 19(21): 2548-2556.
- Felder M, Kapur A, Gonzalez Bosquet J, Horibata S, Heintz J, et al. (2014) MUC16 (CA125): tumor biomarker to cancer therapy, a work in progress. Mol Cancer 13: 129.
- Kufe DW (2009) Mucins in cancer: function, prognosis and therapy. Nat Rev Cancer 9(12): 874-885.
- Pantshwa JM, Rhoda K, Clift SJ, Pradeep P, Choonara YE, et al. (2018) Chemotherapeutic Efficacy of Implantable Antineoplastic-Treatment Protocols in an Optimal Mouse Model for Human Ovarian Carcinoma Cell Targeting. International journal of molecular sciences 19(10).

 Review Article
Review Article