Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Abu OD1*, Awhin EP2, Iyoha AI3 and Chukwuma AU1
Received: February 21, 2024; Published: March 01, 2024
*Corresponding author: Abu OD, Department of Biochemistry, Faculty of Life Sciences, University of Benin, Benin City, Nigeria
DOI: 10.26717/BJSTR.2024.55.008687
Aim: The present study investigated the hepatotoxicity of aqueous extract of Dialium guineense stem bark in Wistar rats.
Materials and Methods: A total of thirty-five (35) rats (mean weight = 170 ± 10 g) were divided into seven groups of five rats each. Group I rats served as control, while others received graded doses of the medicinal plant extract (200 - 5000 mg/kg body weight, bwt) for a period of 28 days. Indices of liver function were measured in plasma.
Results: There were no significant differences in plasma activities of alanine Aminotransferase (ALT) and aspartate Aminotransferase (AST) in rats exposed to 200 - 1000 mg/kg bwt aqueous extract, relative to control group (p > 0.05). However, the corresponding activities of Gamma Glutamyltransferase (GGT) were markedly reduced, when compared with control group (p < 0.05). There were no significant differences in AST/ALT and relative organ weights among the groups (p > 0.05). There were also no significant increases in the concentrations of albumin, total protein, globulins, bilirubin and plasma Malondialdehyde (MDA) in treated rats, relative to the control group (p > 0.05). Furthermore, the basal activities and concentrations of the measured indices of liver function were not significantly different from those after treatment (p > 0.05).
Conclusion: The aqueous extract of guineense stem bark did not elicit any obvious deleterious effects on liver function of rats.
Keywords: Albumin; Aminotransferases; Hepatotoxicity; Medicinal Plant; Total Protein
Abbreviations: ALT: Aminotransferase; AST: Aminotransferase; GGT: Glutamyltransferase; MDA: Malondialdehyde; BDH: British Drug House; One-Way ANOVA: One-Way Analysis of Variance; LFTs: Liver Function Tests
Toxicology deals with the deleterious impact of chemical, physical or biological agents on living organisms and the environment [1]. It encompasses toxicity and the underlying principles. The amount of a substance may account for its toxicity: dose is what distinguishes a poison from a remedy. Toxicity is the extent to which a substance can damage an organism. It could impact negatively on whole organism, as well as organelles [1,2]. The major principle of toxicology is that “dose and effect are directly proportional to each other”; seemingly harmless substance like water can intoxicate if consumed in a very high dose, while a toxic substance such as snake venom may not produce any toxicity if present in very low concentration. It has been established that toxicity is species-specific [3,4]. Toxicity measurement is carried out via meticulous observation of the effect of a substance on whole organism, organ, tissue or cell. The assessment is usually population-based [1]. Toxic effects are classified based on the site of effect. When the effect is observed at a single site, it is known as “specific target organ effect”, but, when it happens at several sites, it is called “systemic toxicity”. Acute, sub-chronic, and chronic toxicities are examples of systemic toxicity [1]. Acute toxicity occurs almost immediately after an exposure, while continuous exposure for weeks or months results in sub-chronic toxicity. As a common human exposure pattern sub-chronic toxicity assessment examines non-lethal impacts of toxicants on behavior, growth and reproduction, caused by biochemical, haematological, physiological and histopathological disturbances [5].
As the principal organ for the detoxification and distribution of drugs the liver is closely monitored to confirm the relative safety of a substance [6]. The liver is an indispensable organ involved in metabolism, detoxication and excretion. Some medicinal substances, chemicals and herbal preparations may cause liver injury [7]. Hepatotoxicity refers to injury to liver cells (hepatocytes) or liver function impairment. It means liver damage driven by chemicals. Hepatocellular damage may be caused by drugs, food additives, alcohol, chlorinated solvents, peroxidized fatty acids, fungal toxins, radioactive isotopes, environmental toxicants, and certain herbs [8,9]. Playing a key role in the transformation and clearance of chemicals the liver is highly vulnerable [10]. The organ may be injured by some medicinal substances, when taken in overdoses (for example, paracetamol) or within therapeutic ranges (for example, halothane). Hepatotoxins refer to agents that cause liver injury [11]. The aim of this study was to investigate the hepatotoxicity of aqueous extract of D. guineense stem bark in Wistar rats.
Chemicals and Reagents
Analytical grade reagents were used in this study. Kits used to perform liver integrity tests were products of Randox Laboratories Limited (United Kingdom). The other chemicals/reagents were bought from Pyrex Scientific Limited (United Kingdom), Merck (Germany), British Drug House (BDH) (England), and Sigma-Aldrich Ltd. (USA).
Plant Material and Authentication
Freshly harvested stem barks of D. guineense were collected from Auchi, Etsako West, Edo State, Nigeria. Their identification and authentication took place at the University of Benin herbarium in the Department of Plant Biology and Biotechnology, Faculty of Life Sciences (No. UBHD330).
Plant Extraction
The ground plant material was extracted via maceration for 72 h [12]. Exactly 500 g of the pulverized stem bark was steeped in 5 L distilled water. The aqueous extract produced after filtration was freeze-dried via lyophilization.
Animals
Thirty-five (35) male Wistar rats (mean weight = 170.00 ± 10.00 g) were bought from the Animal House of the Department of Anatomy, University of Benin, Benin City, Nigeria. The rats were kept in metal cages under standard laboratory conditions. They had unrestricted access to feed and water. The rats were acclimatized to the laboratory environment for seven days, prior to commencement of the study. Standard experimental procedure was adopted for this study.
Subchronic Toxicity Study
The rats were randomly divided into 7 groups (5 rats/group): “Group I served as control, while others received graded doses of extract (200 - 5000 mg/kg bwt) for 28 days”. Basal samples were collected before the commencement of treatment. At the expiration of the 28-day treatment the rats were euthanized after an overnight fast. The collected blood was centrifuged at 3000 rpm for 10 min to obtain clear plasma.
Organ Function Tests
Liver function tests (LFTs) such as ALT, AST, GGT, total protein, albumin, globulins and bilirubin were carried out in rat plasma [13-17].
Determination of Lipid Peroxidation in Liver
Malondialdehyde (MDA) concentration was measured in 20 % liver homogenate [18].
Statistical Analysis
Data obtained in this study are expressed as mean ± standard error of mean (SEM, n = 5). One-Way Analysis of Variance (One-Way ANOVA) was performed with SPSS (version 20). Groups were compared using Duncan multiple range test. Values of p < 0.05 were considered statistically significant.
Percentage Weight Increase and Relative Organ Weight of Rats
Percentage increases in body weights of rats treated with aqueous extract of D. guineense stem bark were significantly reduced, when compared with control group (p < 0.05). However, there were no significant differences in the corresponding relative organ weights among the groups (p > 0.05; (Table 1). Data are percentage weight increase and relative liver weight, and are expressed as mean ± SEM (n = 3). ap < 0.05, when compared with control group; bp < 0.05, when compared with the other treatment groups. Liver Function of Rats Treated with Varied Doses of Plant Extract “There were no significant differences in the activities of plasma ALT and AST in rats exposed to doses ranging from 200 - 1000 mg/kg bwt aqueous extract, relative to control group (p > 0.05). However, the corresponding activities of GGT were significantly reduced, when compared with control group (p < 0.05). While the activities of ALT in groups VI and VII, and AST in groups V, VI and VII were significantly increased (p < 0.05), the corresponding activities of GGT were not significantly different from that of control group (p > 0.05). There were no significant differences in AST/ALT among the groups (p > 0.05; (Table 2). There were also no significant increases in the concentrations of albumin, total protein, globulins, bilirubin and plasma MDA in treated rats, relative to the control group (p > 0.05; (Tables 3-5). Furthermore, the basal activities and concentrations of the measured indices of liver function were not significantly different from those after treatment (p > 0.05; (Tables 2-4)”.
Table 3: Concentrations of Liver Proteins in Rats Treated with Aqueous Extract of D. guineense Stem Bark.
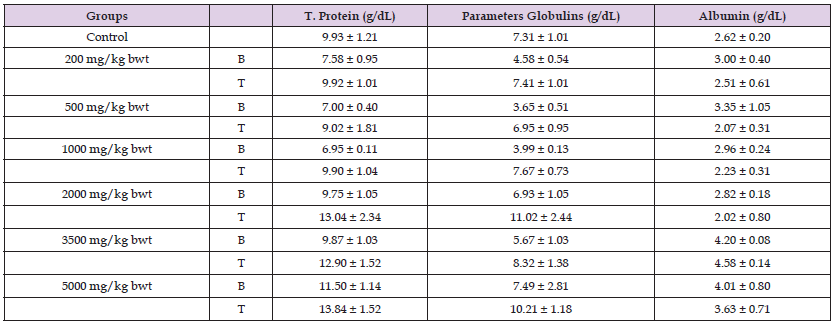
Table 4: Concentrations of Bilirubin in Rats Treated with Aqueous Extract of D. guineense Stem Bark.

Data are indices of liver function, and are expressed as mean ± SEM (n = 5). Data are indices of liver function, and are expressed as mean ± SEM (n = 5). Data are indices of liver function, and are expressed as mean ± SEM (n = 5). Data are indices of liver function, and are expressed as mean ± SEM (n = 5). T. Bilirubin = total bilirubin; D. Bilirubin = direct bilirubin; and Ind. Bilirubin = indirect bilirubin. Data are concentrations of liver MDA and are expressed as mean ± SEM (n = 5).
The liver in vertebrates and other animals’ function in detoxification, protein synthesis, and synthesis of other important biomolecules. Since there is no way to compensate for its absence in the long-term the liver is necessary for survival [6]. The organ is involved in metabolism: glycogen storage, hemolysis, synthesis of plasma proteins/hormones, and detoxification. Located below the diaphragm of the abdomen the liver secretes bile, an alkaline substance that facilitates digestion (that is, lipids emulsification) [6]. The specialized tissues of the organ control a number of biochemical/physiological reactions [19]. Karat parenchymal and non-parenchymal are the two principal cells found in liver lobes. Commonly referred to as hepatocytes, parenchymal cells constitute approximately 80 % of liver volume, while non-parenchymal cells makeup 40 % of the total number of liver cells, but only 6.5 % of its volume. Sinusoidal hepatic endothelial, Kupffer and hepatic stellate cells are some of the non-parenchymal cells that line liver sinusoid [20]. Liver function is assessed in blood. Liver Function Tests (LFTs) can reveal the degree of liver injury [21]. These tests are employed to screen for liver disease, speculate the possible cause, determine the severity, evaluate prognosis and monitor effectiveness of therapy [22]. Aminotransferases, Alkaline Phosphatase (ALP), GGT, 5’-nucleotidase, and leucine aminopeptidase are LFTs that detect injury to hepatocytes. Serum and urine bilirubin are tests of the liver’s capacity to metabolize drugs and transport organic anions. The hepatocyte’s ability to carry out synthetic function is determined with albumin [22].
During cellular necrosis and increased plasma membrane permeability, liver enzymes released into systemic circulation are used as diagnostic tool to evaluate liver damage. Most proteins in plasma are synthesized by cells of the liver and released into systemic circulation. A sharp decrease in plasma albumin level below normal range suggests a marked decline in the liver’s protein synthetic function. Liver injury or damage results in elevated bilirubin (breakdown product of haemoglobin) level. Total bilirubin elevation caused by reduced uptake and conjugation of bilirubin is as a result of hepatocyte dysfunction, while elevated levels of direct/conjugated bilirubin is due to declined secretion from liver or obstruction of bile ducts. Elevated albumin, bilirubin and total protein levels due to hemolysis or liver disease or both can result in jaundice of the skin or brain kernicterus [23,24]. In this study, treatment of Wistar rats with graded doses of aqueous extract of the medicinal plant did not produce any observable elevation in liver enzymes, proteins and metabolites to subject hepatotoxicity. Even in few cases were ALT and AST were elevated, they were not significantly different from the basal values. The hepatoprotective capacity of extracts of D. guineense stem bark has been reported [24-26]. The results of this study are in agreement with those of previous reports [27-48]. Studies have demonstrated important biological effect of extracts of the medicinal plant [49-58].
The results of this study indicate that the aqueous extract of D. guineense stem bark may not be deleterious to the liver at dose not exceeding 5000 mg/kg bwt. This study has provided first-time evidence as to the relative safety of the plant extract.


