SUMMARY
In this article the authors discussed the results of an investigation of 106 patients with delay of puberty in age 11-17 years old. They noted that this patients have hypogonadotropic hypogonadism (HH). In all age periods of puberty by Tanner patients have low mean levels of LSH, FSH, and total testosterone with some hyperprolactinemia. It was diagnosed 3 degree of severity of HH: easy (11.3%), middle (16.04%) and hard (72.6%).
Keywords: Delay of Puberty; Teenagers
Background
In girls, the first physical marker of the onset of puberty is most often the transition from the B1 to B2 Tanner breast stage, which includes early growth of breast tissue. In boys, the relevant marker is a change from G1 to G2 Tanner genital stage, including testicular enlargement (i.e. achieving >3 ml volume or ≥25 mm length testis) [1,2]https://scholar.google.com/scholar_lookup?journal=Eur+- J+Pediatr&title=IGSF1+variants+in+boys+with+familial+delayed+ puberty&author=SD+Joustra&author=K+Wehkalampi&author= W+Oostdijk&author=NR+Biermasz&author=S+Howard&volume= 174&issue=5&publication_year=2015&pages=687692&pmid= 25354429&. Initially, Marshall and Tanner reported a mean (±SD) puberty of 11.15 (±1.10) years for girls and 11.64 (±1.07) years for boys. Research done in Switzerland [3,4] https://scholar. google.com/scholar_lookup?journal=Eur+J+Pediatr&title=IGSF1+- variants+in+boys+with+familial+delayed+puberty&author=SD+- Joustra&author=K+Wehkalampi&author=W+Oostdijk&author=N R+Biermasz&author=S+Howard&volume=174&issue=5&publication_ year=2015&pages=687692&pmid=25354429&,USA[5] https://scholar.google.com/scholar_lookup?journal=Eur+J+Pediatr& title=IGSF1+variants+in+boys+with+familial+delayed+- puberty&author=SD+Joustra&author=K+Wehkalampi&author= W+Oostdijk&author=NR+Biermasz&author=S+Howard&volume= 174&issue=5&publication_year=2015&pages=687692&pmid= 25354429&and Denmark [6]
https://scholar.google.com/scholar_lookup?journal=Eur+- J+Pediatr&title=IGSF1+variants+in+boys+with+familial+delayed+ puberty&author=SD+Joustra&author=K+Wehkalampi&author= W+Oostdijk&author=NR+Biermasz&author=S+Howard&volume= 174&issue=5&publication_year=2015&pages=687692&pmid= 25354429&showed approximately the same mean age of onset of puberty. Although the average age of onset can be uniform, the onset of puberty occurs over a wide range of ages in normal, healthy adolescents.
Some pathological conditions directly or indirectly affect the timing of puberty and contribute to this discrepancy, but the vast majority of differences in the timing of puberty cannot be attributed to any clinical disorder. In most populations, 95% of girls begin puberty between 8.5 and 13 years of age, and the same percentage of boys between 9 and 13.5 years of age (9–14 years old).). These findings led to the traditional definition of delayed puberty as the absence of development of secondary sexual characteristics by age 13 in girls and by age 14 in boys. However, these restrictions do not apply to all ethnic groups. Due to the downward trend in puberty, as discussed in more detail below, some but not all reports from the US [7,8-11]
https://scholar.google.com/scholar_lookup?journal=Eur+- J+Pediatr&title=IGSF1+variants+in+boys+with+familial+delayed+ puberty&author=SD+Joustra&author=K+Wehkalampi&author= W+Oostdijk&author=NR+Biermasz&author=S+Howard&volume= 174&issue=5&publication_year=2015&pages=687692&pmid= 25354429&and other countries [12,13]
https://scholar.google.com/scholar_lookup?journal=Eur+- J+Pediatr&title=IGSF1+variants+in+boys+with+familial+delayed+ puberty&author=SD+Joustra&author=K+Wehkalampi&author= W+Oostdijk&author=NR+Biermasz&author=S+Howard&volume= 174&issue=5&publication_year=2015&pages=687692&pmid= 25354429&, some argue for earlier age limits for the general population as well. However, the long-term changes reported in the general population at the onset of puberty have not always been observed in adolescents with late development, and therefore there may not be a need to adjust age-specific definitions of delayed puberty in males.
[https://scholar.google.com/scholar_lookup?journal=Eur+- J+Pediatr&title=IGSF1+variants+in+boys+with+familial+delayed+ puberty&author=SD+Joustra&author=K+Wehkalampi&- author=W+Oostdijk&author=NR+Biermasz&author=S+Howard& volume=174&issue=5&publication_year=2015&pages= 687692&pmid=25354429& [14,15] The pathogenesis of delayed puberty includes several conditions but is most often associated with constitutional delay in puberty (also known as constitutional growth and puberty retardation). There are three main groups of differential diagnoses for constitutional delayed puberty: functional hypogonadism, disorders causing primary hypogonadism, and GnRH deficiency leading to hypogonadotropic hypogonadism (HH), although up to 30 different etiologies underlying delayed puberty have been identified. [16] https://scholar. google.com/scholar_lookup?journal=Eur+J+Pediatr&title=IGSF1+- variants+in+boys+with+familial+delayed+puberty&author=SD+- Joustra&author=K+Wehkalampi&author=W+Oostdijk&author=NR+ Biermasz&author=S+Howard&volume=174&issue=5&publication_ year=2015&pages=687692&pmid=25354429& The absence of a pathological history, signs and symptoms, and a positive family history of delayed puberty in one or both parents suggest a diagnosis of constitutional delayed puberty; however, major pathological conditions must be ruled out before a diagnosis can be made.
These include the aforementioned differential diagnoses of delayed puberty: functional HH, in which late puberty is associated with delayed maturation along the HHY axis secondary to chronic disease (found in about 20% of subjects with delayed puberty), malnutrition, excessive exercise and psychological or emotional stress; hypergonadotropic hypogonadism with primary gonadal insufficiency, leading to an increase in the level of gonadotropin due to the lack of negative feedback (found in approximately 7% of male patients and in 26% of female patients with delayed puberty); and permanent HH, characterized by low levels of LH and FSH (9% of boys and up to 20% of girls) [17]
https://scholar.google.com/scholar_lookup?journal=Eur+- J+Pediatr&title=IGSF1+variants+in+boys+with+familial+delayed+ puberty&author=SD+Joustra&author=K+Wehkalampi&author= W+Oostdijk&author=NR+Biermasz&author=S+Howard&volume= 174&issue=5&publication_year=2015&pages=687692&pmid= 25354429& All of the above formed the basis for this study.
Purpose of the study
To study the characteristics of hormonal disorders in adolescents (boys) with hypogonadotropic hypogonadism in the Kashkadarya region according to screening data.
Material and Research Methods
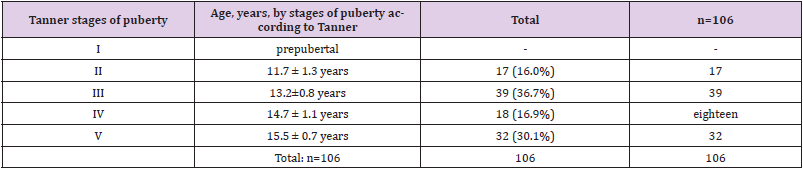
Of the examined 523 schoolchildren in Karshi were subjected to screening studies at the age of 11 to 16 years. Further, from among the examined adolescents, we identified 106 (20.3%) adolescents aged 11-16 years suffering from delayed sexual development. All 106 patients were subjected to studies, including the study of somatic, endocrine status, clinical, biochemical, hormonal (STH, LH, FSH, prolactin, TSH, testosterone, cortisol, free thyroxine, etc.), in addition, anthropometric studies were carried out in of all adolescents (height and weight deficit, projected height, parental height, centile, SDS of height, weight, etc.) Based on the international Tanner-Weithaus height-weight chart, height and weight deficit was assessed, and the stage of puberty was assessed by Tanner stages (using tables and orchidometer). If necessary, X-ray (X-ray of the hand and Turkish saddle), ultrasound examination of the thyroid gland and genital organs were prescribed. Hormonal studies were performed in the laboratory of the RSNPMCE of the Ministry of Health of the Republic of Uzbekistan. The obtained data were processed using computer programs Microsoft Excel and Statistica_6. Differences between groups were considered statistically significant at p<0.05. Mean values (M) were calculated, medium standard deviations (m). Research results. (Table 1) shows the distribution of patients by age.
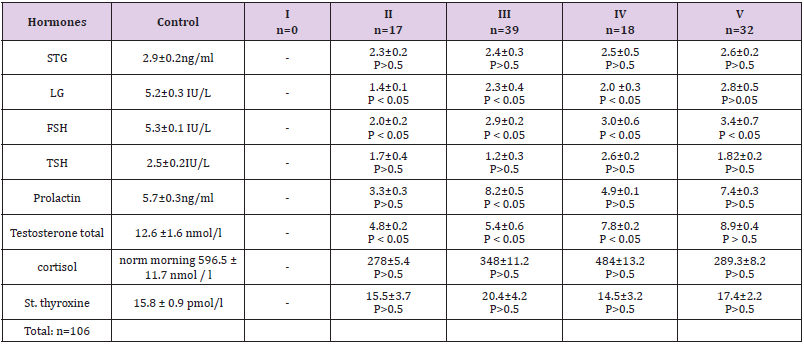
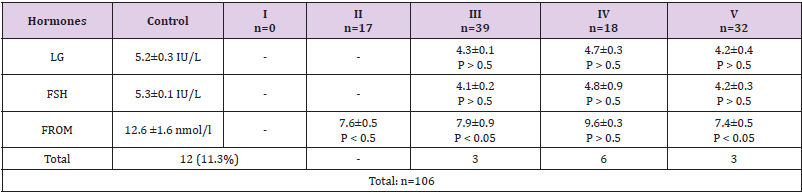
As can be seen from (Table 1), the most frequently examined patients were patients aged from 13.2±0.8 years and 15.5 ± 0.7 years (36.7% and 30.1%). (Table 2) gives the average values of various hormones in patients with PD by stages of sexual development. From (Table 2) it follows that in all age periods of sexual development in the examined patients there was hypogonadotropic hypogonadism (HH): there was a significant decrease in the average levels of LH, FSH, total testosterone - WC (p < 0.05). At the same time, these values were the lowest in patients with the 2nd stage of puberty according to Tanner, that is, at the age11.7 ± 1.3 years (n = 17) against the background of normoprolactinemia. Mean prolactin levels were not significantly elevated in Tanner stage 3 and 5 patients (P>0.5). It should be emphasized that the levels of growth hormone, TSH and free thyroxine, as well as cortisol were within the normal range in all patients (n = 106). P - significance of differences compared with the control group. The table for comparison shows fluctuations in hormone levels from 11 to 16 years of age in the control group (healthy individuals). When comparing stages puberty and hormonal data, it was found that as the age increases, the average values of LH, FSH, total testosterone also increase, although they remain significantly reduced.
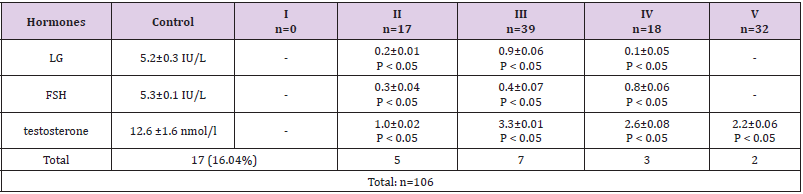
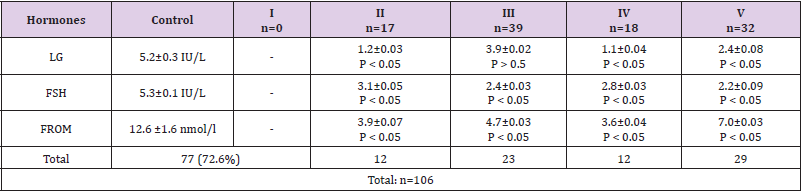
Next, we analyzed the cases of the lowest values of LH, FSH and total testosterone, namely, when the levels of LH/FSH ranged from 0.1 to 0.9 IU/L, OT - from 1 to 3 nmol/l (severe degree of HH) , cases of average values of gonadotropins and OT, when the levels of LH / FSH were in the range from 1 to 4 IU / L, OT - from 3 to 7 nmol / L (moderate severity of HH), as well as cases with mild HH, when the levels LH/FSH fluctuated from 4 and above IU/L, OT - from 7 nmol/l and above (mild severity of HH). Thus, this characteristic allowed us to identify 3 groups of patients with 3 degrees of severity HH - moderate, moderate, and severe - depending on the average values of LH, FSH, OT. These data are given in (Tables 3-5). (Table 3) shows the number of patients with severe by Tanner stages. The total number of these patients was39 (36.7%). As can be seen from the data in (Table 3), when analyzing the lowest values of LH, FSH and total testosterone, namely, when the levels of LH/FSH ranged from 0.1 to 0.9 IU/L, OT - from 1 to 3 nmol/l (severe HH) the total number of cases was 17(16.04%). In this group, the significance of differences was the highest (P < 0.05). (Table 4) shows the number of patients with moderateGG by Tanner stages. The total number of these patients was77 (72.6%). When analyzing cases of moderate severityGH we proceeded from those average values of gonadotropins and OT, when the levels of LH/FSH were in the range from 1 to 4 IU/L, OT - from 3 to 7 nmol/l (moderate severity of GH). In total, there were 77 such cases. (72.6%) and this group was dominant (P < 0.05 and also P > 0.5).
Table 3: The number of patients with severe HH depending on the average values of LH, FSH and OT in patients according to 5 stages of Tanner).
Note: OT is total testosterone. P - significance of differences compared to control (1)
Table 4: The number of patients with moderate HH severity depending on the average values of LH, FSH and OT in patients according to 5 stages of Tanner).
Note: OT is total testosterone. P - significance of differences compared to control (1)
Table 5: The number of patients with mild HH depending on the average values of LH, FSH and OT in patients according to 5 stages of Tanner).
Note: OT is total testosterone. P is the significance of differences compared to control (1).
(Table 5) shows the number of patients with mild HH by Tanner stages. The total number of these patients was 12 (11.3%). Cases with mild HH, when LH/FSH levels fluctuated from 4 and above IU/L, OT - from 7 nmol/l and above (mild severity of HH) amounted to12 patients (11.3%). In this group of patients, the significance of differences in the content of H, FSH, and WC in blood plasma was less significant, while being in the range from P < 0.05 to P > 0.5. Thus, the analysis of hormonal results showed that the most frequently observed moderate severity HH (72.6%), while severe (16.04%) and light (11.3%) of his degree. Only 1 (0.9%) patient had hypergonadotropic hypogonadism.
Conclusion
1) In all age periods of sexual development, the examined patients had hypogonadotropic hypogonadism: there was a significant decrease in the average levels of LH, FSH, total testosterone (p < 0.05). Only 1 (0.9%) patient had hypergonadotropic hypogonadism.
2) When comparing the stages of puberty and hormonal data, it was found that as the age increases, the average values of LH, FSH, and total testosterone also increase, although they remain significantly reduced.
3) 3 degrees of severity are established hypogonadotropic hypogonadism: mild (11.3%), moderate (16.04%) and heavy (72.6%).
4) This category of patients’ needs further examination (magnetic resonance imaging of the pituitary gland, ultrasound of the genital organs, etc.) and treatment.
References
- Marshall WA, Tanner JM (1970) Variations in the pattern of pubertal changes in boys. Arch DisChild 45(239): 13-23.
- Marshall WA, Tanner JM (1969) Variations in pattern of pubertal changes in girls. Arch DisChild 44(235): 291-303.
- Largo RH, Prader A (1983) Pubertal development in Swiss girls. Helv Paediatr Acta 38(3): 229-243.
- Largo RH, Prader A (1983) Pubertal development in Swiss boys. Helv Paediatr Acta 38(3): 211-228.
- Roche AF, Wellens R, Attie KM, Siervogel RM (1995) The timing of sexual maturation in a group of US white youths. J Pediatr Endocrinol Metab 8(1): 11-18.
- Juul A, Teilmann G, Scheike T, Hertel NT, Holm K, et al. (2006) Pubertal development in Danish children: Comparison of recent European and US data. Int J Androl 29(1): 247-255.
- Sun SS, Schubert CM, Chumlea WC, Roche AF, Kulin HE, et al. (2002) National estimates of the timing of sexual maturation and racial differences among US children. Pediatrics 110(5): 911-919.
- Buck Louis GM, Gray LE Jr, Marcus M, Ojeda SR, Pescovitz OH, et al. (2008) Environmental factors and puberty timing: Expert panel research needs. Pediatrics 121(Suppl 3): S192-S207.
- Herman-Giddens ME, Slora EJ, Wasserman RC, Bourdony CJ, Bhapkar MV, et al. (1997) Secondary sexual characteristics and menses in young girls seen in office practice: A study from the Pediatric Research in Office Settings network. Pediatrics 99(4): 505-512.
- Herman-Giddens ME, Steffes J, Harris D, Slora E, Hussey M, et al. (2012) Secondary sexual characteristics in boys: Data from the Pediatric Research in Office Settings Network. Pediatrics 130(5): e1058-e1068.
- Karpati AM, Rubin CH, Kieszak SM, Marcus M, Troiano RP (2002) Stature and pubertal stage assessment in American boys: The 1988-1994 Third National Health and Nutrition Examination Survey. J Adolesc Health 30(3): 205-212.
- Aksglaede L, Sørensen K, Petersen JH, Skakkebaek NE, Juul A (2009) Recent decline in age at breast development: The Copenhagen Puberty Study. Pediatrics 123(5): e932-e939.
- Sørensen K, Aksglaede L, Petersen JH, Juul A (2010) Recent changes in pubertal timing in healthy Danish boys: Associations with body mass index. J Clin Endocrinol Metab 95(1): 263-270.
- Sasha R Howardand Leo Dunkel (2019) Delayed Puberty—Phenotypic Diversity, Molecular Genetic Mechanisms, and Recent Discoveries. Endocr Rev 40(5): 1285-1317.
- Sedlmeyer IL, Palmert MR (2002) Delayed puberty: Analysis of a large case series from an academic center. J Clin Endocrinol Metab 87(4): 1613-1620.

 Research Article
Research Article