Abstract
The present study addresses parasitic infestation in Macaca mulatta in free-ranging and captive areas of Shadhana Awshadhalaya factory area, Gandaria and National Zoo of Bangladesh, Mirpur, Dhaka. A total of 190 stool samples were collected randomly from Gandaria (free-ranging) and Zoo (captive). For the collection of cyst, egg and larvae of the parasites, the formalin-ether concentration technique was applied. Parasites belonging to 30 species were identified, 6 species of protozoa (Entamoiba coli, Eimeria sp., Isospora sp., Toxoplasma gondii, Chilomastix mesnili and Gregarina sp.), 5 species of cestodes (Taenia sp., Moniezia sp., Reillietina sp., Bertiella sp. and Amoebataenia sp.), 11 species of nematodes (Ascaris lumbricoides, Toxocara sp., Trichuris trichiura, Strongyloides sp., Ancylostoma sp., Ascarops sp., Gongylonema sp., Gnathostoma sp., Subulura sp., Enterobius sp. and Capillaria sp.), 7 species of trematodes (Neoglyphe sp., Watsonius watsoni, Schistosoma mansoni, Paragonimus sp., Clonorchis sinensis, Brachylaemus sp. and Gastrothylax sp.) and 1 species of pentastomida (Linguatula sp.).
This was the first record in Bangladesh on the species of gastrointestinal parasites affecting monkeys in both captivity and wild; and also their prevalence. While the average intensity of parasites was almost the same (35%) in both areas, the overall prevalence of parasites in Zoo (46.42%) was significantly higher than in Gandaria (39.76%). Nematoda was the most dominant parasite in both study areas. In Gandaria and Zoo (2014-2015), the highest prevalence of nematoda was 77.78% and 80% respectively and in 2015-2016, the prevalence in both areas was 72% and 74% accordingly. The estimated EPG was the highest for nematoda in both study areas and both study periods. Rhesus macaques (Macaca mulatta) constitute an integral part of biodiversity and the cognizable link between humans and nature. Therefore, this research will serve as a guide to establish the necessary preventive measures to favor the conservation of these species.
Keywords: Macaques; Gastrointestinal Parasites; Intensity; Prevalence; EPG
Introduction
Primates are noticed to be one of the severely threatened animals in the world, with approximately 50% of species currently at risk of extinction (IUCN [43]). More than 70% of primates are classified on the IUCN Red List as critically endangered, which in the near future they could disappear forever from Asia (IUNC [44]). Therefore, there is a growing recognition that it is very significant to better understand infectious disease dynamics in wild primate populations (Chapman, et al. [1,2]). Parasites play a significant role in the ecosystems, influencing the ecology and evolution of species interactions, host population growth and regulation (Hudson, et al. [3,4]) and even community biodiversity (Hudson [5]). Parasites and infectious diseases can trigger or accelerate population declines and have become a big alarm in conservation biology (Altizer, et al. [6]). Primates are susceptible to various protozoa and helminth parasites (Wanert, et al. [7]). Thus, parasites are an integral part of the natural history of mammals and are always of interest.
Natural infections of gastrointestinal protozoa and helminths have been exhibited in a variety of species of monkeys. Gastrointestinal parasitism in the colonies of non-human primates (NHPs) is often addressed as a research topic (Sano, et al. [8-10]). Strongyloides sp., Oesophagostomum sp., and Trichuris trichiura were previously considered among the most common pathogens causing poor development, anemia, and diarrhoea in macaques and other NHPs (Honjo, et al. [11-13]). Recently, captive NHPs were resulted pathogenic for their hosts and found that they can be frequently affected by several species of intestinal protozoans (Lee, et al. [14- 17]). Several studies have demonstrated that non-human primates may be carriers of human gastrointestinal parasites. Moreover, most parasites noted in NHPs represent a high zoonotic risk for researchers and caretakers in breeding centers (Loomis [18]).
Thus, people living in close proximity of such animals or individuals involved in game parks, animal orphanages or research stations may be at risk of acquiring pathogens from infected animals. Moreover, non-human primates are often valuable in studies of either human diseases processes or vaccine and drug development. It is therefore important that animals used in such studies should not harbour parasites since results obtained using parasitized animals may not reveal the true picture. Additionally, these animals play an important role in natural ecosystems; hence heavy parasite loads may have a negative impact on their population sizes. The rhesus macaque is well known to science due to its relatively easy upkeep in captivity, wide availability and closeness to humans anatomically and physiologically. It has been used extensively in medical and biological research on human and animal health-related topics. However, no previous works on endoparasitic infections of rhesus monkeys in Bangladesh were found in the literature. This article, therefore, highlights the exploratory and descriptive study on the different types of gastrointestinal parasites that affect populations of both wild and captive monkeys.
Materials and Methods
Sample Collection and Preparation
A total of 190 fecal samples were collected randomly from two study areas, the Shadhana Awshadhalaya factory area, Gandaria, Dhaka and the National Zoo of Bangladesh, Mirpur, Dhaka between March 2014 to February 2016. The monthly sampling covered the seasons of summer (March to June), fall (July to October) and winter (November to February). The samples of free-living monkeys were collected from Gandaria region and captive monkeys from Zoo with the permission of the authority. Only fresh, non-dry samples were collected and, whenever possible, when defecation was observed. As far as possible faecal samples were usually collected in the morning and then brought to the Parasitology laboratory of the Department of Zoology, University of Dhaka for microscopic examination.
Formalin-Ethyl Acetate Sedimentation Technique: Preserved faecal samples were processed using the formalin-ethyl acetate sedimentation technique (Cheesbrough [19]). Samples were collected into sealable, plastic bags immediately after defecation is observed. Within 8 h of collection, the collective sum of 2 g taken from each sample was transferred to 2-ml plastic tubes containing 0.75 ml 10% buffered formalin solution. Fixed faecal samples of Rhesus macaques were later transported to the Parasitological laboratory of the Department of Zoology, University of Dhaka, Bangladesh. All faecal samples were examined via a modified formalin–ether sedimentation protocol, substituting ethyl acetate for ether, as the former is less volatile (Hernandez, et al. [20]). Approximately 1 g of formalized faeces was diluted in 5 ml formalin, and centrifuged at 1,500 rpm for 3 min. The supernatant was then discarded, and the concentrated pellet was weighed (Muller-Graf, et al. [21]). To identify and quantify parasitic infection, an aliquot was removed from the homogenous suspension, placed in the chamber of a McMaster slide, and examined at 100x magnifications. It was counted all helminthes eggs and larvae were observed within the chamber’s 0.15-ml grid to calculate the number of eggs per gram of feces (EPG) from each sample.
Epidemiological Parameters: For each parasite, the prevalence and mean intensity were recorded. Prevalence was counted as the number of infected macaques per sampled subset of the population (Margolis, et al. [22-25]). Mean intensity was defined as the average number of infective stages (Table 1), among the infected members of each population.
Taxonomic Identification of the Parasites: Eggs of various gastrointestinal parasites were identified and the references and published articles such as Yamaguti [46-49], Cheng, et al. [26,27], other references, etc. were consulted. Representatives of each parasite were photographed by a digital camera.
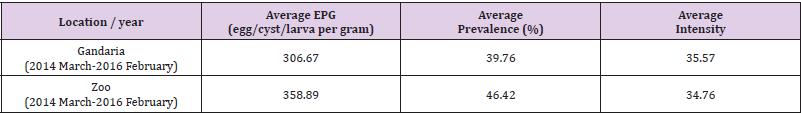
Table 1: Average prevalence, intensity and EPG (egg/cyst/larva per gram) of parasites by location from March 2014 to February 2016.
Statistical Analysis: All statistical tests were carried out using SPSS (Version 22) and R software (Version 3.1.3). A nonparametric repeated-measures analysis of variance (ANOVA) was conducted. The variance ratio with the p-value was used to compare the dispersion of parasites between the Gandaria and Zoo samples. For each case, it was considered the parametric (Welch) and nonparametric (Wilcoxon) tests to study if the two population means for each of the parasites was the same for the two study areas. It was also found the 95% confidence interval of the differences between the two population means for each of the parasites.
Results
This paper aims to determine the parasite community of the host rhesus monkeys (Macaca mulatta), to estimate the prevalence and intensity of rhesus macaques in two study areas, the National Zoo (captive) and Shadhana Awshadhalaya factory area (freeranging) Dhaka, and to study the seasonal prevalence of helminth and protozoan parasitic infestation. Eggs, cysts and larva of parasites were identified based on their taxonomy, morphology and characteristics. Parasites belonging to 30 species were identified, 6 species of protozoa (Entamoeba coli, Eimeria sp., Isospora sp., Toxoplasma gondii, Chilomastix mesnili and Gregarina sp.), 5 species of cestodes (Taenia sp., Moniezia sp., Reillietina sp., Bertiella sp. and Amoebataenia sp.), 11 species of nematodes (Ascaris lumbricoides, Toxocara sp., Trichuris trichiura, Strongyloides sp., Ancylostoma sp., Ascarops sp., Gongylonema sp., Gnathostoma sp., Subulura sp., Enterobius sp. and Capillaria sp.), 7 species of trematodes (Neoglyphe sp., Watsonius watsoni, Schistosoma mansoni, Paragonimus sp., Clonorchis sinensis, Brachylaemus sp. and Gastrothylax sp.). The pentastomida (Linguatula sp.), was the first record in Bangladesh on the species of gastrointestinal parasites affecting monkeys in both captivity and the wild. This article compares the prevalence and intensity of parasites from two study areas which covered two study periods- the first study period (2014-2015) and the last study period (2015-2016).
Overall Estimate of Parasites Prevalence, Intensity and EPG (Egg/Cyst/Larva Per Gram): In the present study, from March 2014 to February 2016, the average prevalence and EPG of parasites were higher in Zoo than that in Gandaria. The average prevalence and EPG of parasites in Zoo were 46.42% and 358.89 respectively. The overall estimates of EPG prevalence and intensity of parasites for free-ranging and captive areas were averaged over the two study periods in both cases. It was observed that on average the number of eggs per gram of Gandaria samples was 306.67 and for that of Zoo, samples were 3888.89. While the average intensity of parasites was estimated to be around 35% in both the areas, the overall prevalence of parasites in Zoo (46.42%) was significantly higher (P-value <0.1) than Gandaria (39.76%) (Table 1).
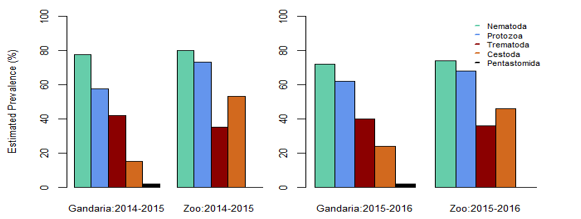
Estimated Prevalence, Mean Intensity and EPG (Egg/Cyst/ Larva Per Gram) of Parasite Groups by Location and Study Year: In the present study, it is evident that nematoda was the most dominant parasite. In both, the study areas, the prevalence of nematoda among others was the maximum during 2014-2015 and 2015-2016. In 2014-2015 samples showed the prevalence of nematoda was 77.78% in Gandaria and 80% in Zoo. In 2015-2016, the prevalence of nematoda in Gandaria and Zoo were 72% and 74% respectively. Unlike a very low prevalence (around 2%) of pentastomida was observed in Zoo (2014-2015). The study did not find any parasites of pentastomida in Gandaria. (Table 1, Figure 1). The study further observed the mean intensity of parasite groups among the two study periods and study areas. the estimated mean intensity of protozoa was maximum (76.06) in Zoo (2015-16). On the other hand, the mean intensity of pentastomida was absent in Zoo for both study years.
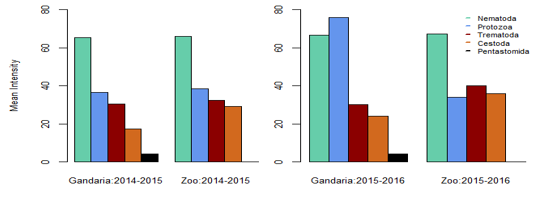
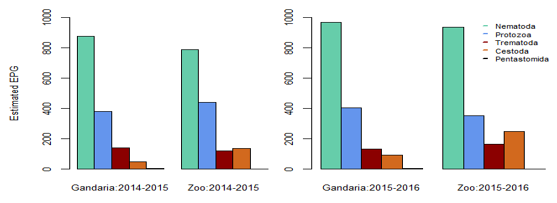
In Gandaria, the mean intensity of nematoda was 65.41 in 2014-2015 and that was 66.17 in Zoo at that time. In 2015-2016, the intensity of nematoda in both Gandaria and Zoo were 66.76 and 67.51 respectively. However, the mean intensity of trematoda and cestode (35.85) was found in Gandaria, Dhaka (2015-16) samples. (Table 1, Figure 2). The estimated EPG was the highest for nematoda in both study areas and both study periods. In 2014-2015, the EPG of Gandaria and Zoo were 879.17 and 791.67 respectively. Moreover, in 2015-2016 those were 970.83 and 937.50 accordingly. On the contrary, in 2014-15, the estimated EPG of trematoda was higher in Gandaria than that of Zoo and the opposite situation was seen in 2015-16. In the first study year, the EPG of cestoda in Gandaria was 50 and that was 138.89 in Zoo. On the other hand, in 2015-2016 those were 95.83 and 250 in Gandaria and Zoo respectively (Figure 3).
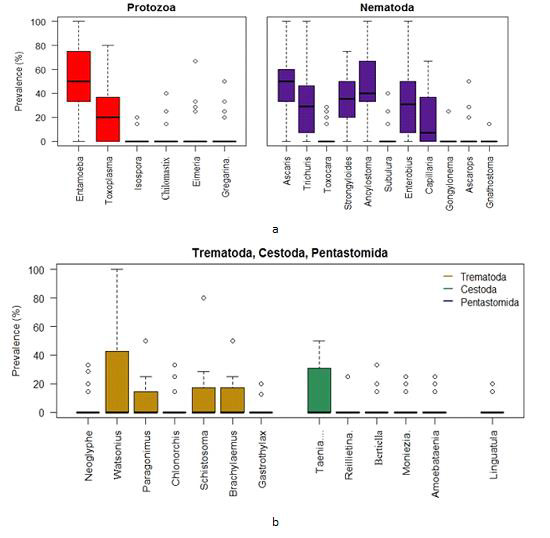
Box plots for the prevalence of parasites in the Gandaria samples (2 years data).
Using the combined data of Gandaria the Box plot for each of the parasites demonstrate that the prevalence of Entamoeba sp. was centered higher than all other parasites in the protozoa group. Both the Entamoeba sp. and Toxoplasma sp. showed higher spread and positive infections.The prevalence of Ascaris sp. was centered higher than other nematode parasites. Most of the parasites in this group were highly spread out. Four parasites in protozoa and five in nematoda group had very negligible prevalence (Figure 4a). The prevalence of most of the parasites of trematoda, cestoda and pentastomida were centered near zero. All most all the parasites occurred with less than 50% of prevalence in Gandaria. Only the Taenia sp. among the cestoda group had few non zero observations (Figure 4b).
Figure 4:
a. Box plots for the prevalence of parasites (protozoa & nematoda) in the Ganderia samples (2 years data).
b. Box plots for the prevalence of parasites (trematoda, cestoda & pentastomida) in the Gandaria samples (2 years data).
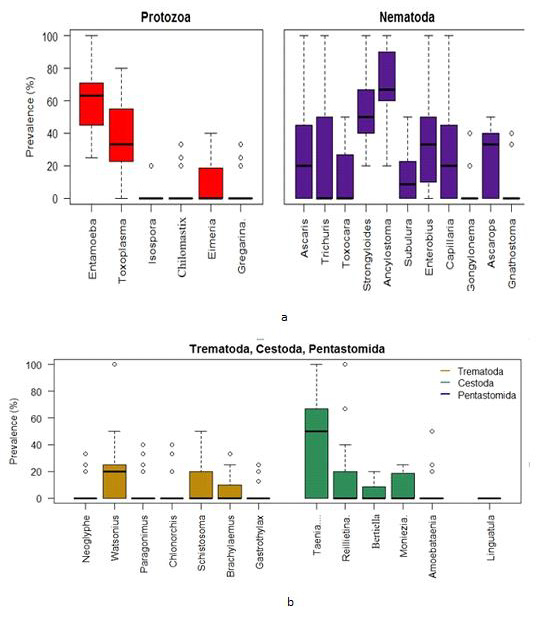
Using the combined Zoo data, the Box plots indicate that the prevalence of Entamoeba sp. was centered the highest followed by Toxoplasma sp. among the protozoa group. The Entamoeba sp. prevalence was negatively skewed while that of Toxoplasma sp. was positively skewed. The spread of Toxoplasma sp. prevalence was higher than other protozoan parasites. On the other hand, the prevalence of Ancylostoma sp. was centered the highest among the nematoda parasites. Most of the parasites in this group were highly spread out in terms of prevalence (Figure 5a).
Figure 5:
a. Box plots for the prevalence of parasites (protozoa & nematoda) in the Zoo samples (2 years data).
b. Box plots for the prevalence of parasites (trematoda, cestoda & pentastomida) in the Zoo samples (2 years data).
Discussion
The present analyses reveal significant richness and diversity of gastrointestinal parasites in the rhesus monkeys in Gandaria and Zoo. Parasites belonging to 30 species were identified, 6 species of protozoa (Entamoiba coli, Eimeria sp., Isospora sp., Toxoplasma gondii, Chilomastix mesnili and Gregarina sp.), 5 species of cestodes (Taenia sp., Moniezia sp., Reillietina sp., Bertiella sp. and Amoebataenia sp.), 11 species of nematodes (Ascaris lumbricoides, Toxocara sp., Trichuris trichiura, Strongyloides sp., Ancylostoma sp., Ascarops sp., Gongylonema sp., Gnathostoma sp., Subulura sp., Enterobius sp. and Capillaria sp.), 7 species of trematodes (Neoglyphe sp., Watsonius watsoni, Schistosoma mansoni, Paragonimus sp., Clonorchis sinensis, Brachylaemus sp. and Gastrothylax sp.) and 1 species of pentastomida (Linguatula sp.). This was the first record in Bangladesh on the species of gastrointestinal parasites affecting monkeys in both captivity and wild; and also their prevalence.
By comparison, 21 gastrointestinal parasites were identified in Kenya’s Tana River mangabey (Mbora, et al. [28]) and 14 parasite species were identified in monkeys of Uganda’s Kibale Forest (Gillespie, et al. [29]). Thirteen parasite species were found in Mahale National Park of Tanzania (Kooriyama, et al. [30]). Total 14 species were found in Rubondo Island National Park of Tanzania (Petrzelkova, et al. [45]) and a total of 23 gastrointestinal parasites were recorded in the Taï monkeys, African nonhuman primates (Kouassi, et al. [32]); recorded 23 gastrointestinal parasites with relatively high prevalence. However, no cestode species were recovered. According to the present work a total of 30 parasites with 5 species of cestodes were recorded. Jha, et al. [33] a prevalence survey of gastrointestinal parasites of Rhesus Monkeys was conducted in three temples Pashupatinath, Swyombhunath and Tripureshwor of Kathmandu. Total 121 fresh faecal samples, were collected randomly from these areas, and three species of protozoa and ten species of helminths were detected by microscopical examination of faecal samples.
A total of 30 gastrointestinal parasites were recorded in the present observation which represents the greatest parasite richness documented to date for Rhesus monkeys. Therefore, the diversity indices demonstrate considerable species diversity and equitable distribution of gastrointestinal parasites in monkeys. Faecal samples of monkeys (captive and free-living) were collected from the Shadhana Awshadhalaya factory area, Gandaria and the National Zoo of Bangladesh, Mirpur. Primates living in complex habitats are infected by a greater intensity and diversity of parasites (Poulin, et al. [34]; Nunn,et al. [35]; Nunn and Altizer [36]). Poor quality habitat and disturbed habitats are much more likely to harbor a greater intensity and diversity of parasites relative to host populations in optimum habitats (Poulin, et al. [34]; Nunn, et al. [35]; Nunn and Altizer [36]). In parasites with multi-stage life cycles, or with infective stages transmitted through the environment, the habitat of the host-parasite interaction plays a more direct role in the transmission of that parasite (Altizer [36]; Combes, et al. [38] and Grutter [39]).
Thus, the environmental conditions and management practices in the study areas (captive and free-living), such as food, cleanliness, overcrowding and deworming among others may influence the prevalence of parasitic infections and are responsible for the parasite richness in rhesus monkeys. In the present study, nematoda was the most dominant parasite in both study areas (captive and free-ranging). In contrast, the majority of the enteric parasites of captive non-human primates are protozoans, (Lane, et al. [40]; Ye,et al. [41]; da Silva Barbosa, et al. [10]). In this study, the average prevalence and EPG of parasites were higher in Zoo (captive) monkeys than that in Gandaria (free-living) monkeys. According to Kouassi, et al. [32], captive non-human primates are frequently infected with parasites having a direct life cycle and show a lower number of parasitic species in comparison with wild NHPs. Therefore, the environmental conditions of zoo and the life cycle pattern of parasites might be the reason for the higher prevalence of parasitic infections in Zoo than in Ganderia. According to Altizer, et al. [42], seasonality affects free-living stages of parasites through changes in temperature, humidity and rainfall, and host susceptibility and exposure through variations in physiology and behavior. Thus, environmental factors such as temperature, humidity and rainfall may influence the seasonal variation of parasites in Macaca mulatta.
Conclusion
This is the first report on endo-parasitic infection of the rhesus monkey in captive and free-ranging populations. The prevalence, EPG and intensity of helminth and protozoan parasites of the gastrointestinal tract in populations of free-ranging and captive monkeys were noted which provides baseline data on the gastrointestinal parasites in both study areas. The average prevalence of parasites was higher in captive monkeys than that in free-ranging monkeys. Seasonal fluctuation of parasitic infection was found from the captive and free-ranging monkeys which suggests environmental factors may affect the seasonal variation of parasites in monkeys. Rhesus macaques (Macaca mulatta) constitute an integral part of biodiversity and a cognizable link between humans and nature. Therefore, the findings of this study will serve as a guide to establish the necessary preventive measures to favor the conservation of this species.
Acknowledgement
We would like to thank the respective authority Zoo for the permission to collect stool samples from captive monkeys. Deep appreciation and thanks are extended to Bangabondhu Fellowship award on Science, Information and Communication Technology for providing financial support to successfully conduct this research. We are grateful to Department of Zoology, the University of Dhaka for their kind permission and providing necessary laboratory facilities. Our sincere thanks to the laboratory technician Mizanur Rahman for his technical assistance during the laboratory works.
References
- Gillespie TR, Greiner EC, Chapman CA (2005) Gastrointestinal parasites of the colobus monkeys of Uganda. J Parasitol 91(3): 569-573.
- Gillespie TR, Nunn CL, Leendertz FH (2008) Integrative approaches to the study of primate infectious disease: implications for biodiversity conservation and global health. Am J Phys Anthropol 51: 53-69.
- Hudson PJ, Dobson AP, Newborn D (1998) Prevention of population cycles by parasitic removal. Science 282: 2256-2258.
- Hochachka VW, Dhondt AA (2000) Density- dependent decline of host abundance resulting from a new infectious disease. Proc Nat Acad Sci 97: 5303-5306.
- Hudson PJ, Rizzoli A, Grenfell BT, Heesterbeek H, Dobson AP (2002) The Ecology of Wildlife Disease. Oxford UK: Oxford University Press, pp. 119- 138.
- Altizer S, Nunn CL, Lindenfors P (2007) Do threatened hosts have fewer parasites? A comparative study in primates. J Anim Ecol 76: 304-314.
- Wanert F, Vidal S (2006) Maladies, parasites et agents infectieux des Primates non Humains. Sciences et Technique de l’Animal de Laboratoire 1er trimestre 1: 59-65.
- Sano M Kino H, De Guzman TS, Ishii AI, Kino J, Tanaka T, et al. (1980) Studies on the examination of imported laboratory monkey, Macaca fascicularis for E. histolytica and other intestinal parasites. Int J Zoonoses 7(1): 34-39.
- Takano J Narita T, Tachibana H, Shimizu T, Komatsubara H, Terao K, et al. (2005) Entamoeba histolytica and Entamoeba dispar infections in cynomolgus monkeys imported into Japan for research. Parasitol Res 97: 255-257.
- Da Silva Barbosa, A Pissinatti, A Verdan Dib, L Perlingeiro de Siqueira, M Lessa Cardozo, et al. (1993) A Functional Biology of Parasitism: Ecological and Evolutionary Implications. London: Chapman and Hall, pp. 148-171.
- Honjo S, Muto K, Fujiwara T, Suzuki Y, Imaizumi K (1963) Significance of the natural infection of Oesophagostomum sp. in cynomolgus monkeys (Macaca irus) used as experimental animals. Jpn J Med Sci Biol 16: 225-227.
- Wong MM, Conrad HD (1978) Prevalence of metazoan parasite infections in five species of Asian macaques. Lab Anim Sci 28: 412-416.
- Abbott DP, Majeed SK, (1984) A survey of parasitic lesions in wild caught, laboratory-maintained primates: (rhesus, cynomolgus, and baboon). Vet Pathol 21(2): 198-207.
- Lee RV, Prowten AW, Anthone S, Satchidanand SK, Fisher JE, et al. (1990) Typhlitis due to Balantidium coli in captive lowland gorillas. Rev Infect Dis 12: 1052-1059.
- Muriuki SMK, Farah IO, Agwiria RM, Chai DC, Njamunge G, et al. (1997) The presence of Cryptosporidium oocysts in stools of clinically diarrhoeic and normal non-human primates in Kenya. Vet Parasitol 72 (2): 141-147.
- Vogel P, Zaucha G, Goodwin D, Kuehl K, Fritz D (1996) Rapid postmortem invasion of cecal mucosa of macaques by non pathogenic Entamoeba chattoni. Am J Trop Med Hyg 55: 595-602.
- Zanzani S, Epis S, Bandi C, Manfredi MT (2014) What is your diagnosis? Fecal smear stained with Lugol’s solution and Giemsa from a cynomolgus macaque (Macaca fascicularis) presenting with liquid diarrhea. Vet Clin Path 43(2): 293-294.
- Loomis M (1983) Hepatic and gastric amebiasis in black and white colobus monkeys. J Am Vet Med Assoc 183: 1188-1191.
- Cheesbrough M (1987) Medical laboratory manual for tropical countries. Blackworth co. publishers, pp. 570.
- Hernandez AD, Macintosh AJ, Huffman MA (2009) Primate parasite ecology: patterns and predictions from an on-going study of Japanese macaques. In: Huffman MA, Chapman CA (Eds.)., Primate parasite ecology: the dynamics and study of host parasite relationships.Cambridge University Press, Cambridge, pp. 387-402.
- Müller Graf CDM, Collins DA, Woolhouse MEJ (1996) Intestinal parasite burden in five troops of olive baboons (Papio cynocephalus anubis) in Gombe Stream National Park Tanzania. Parasitology 112: 489- 497
- Margolis L, Esch GW, Holmes JC, Kuris AM, Schad GA (1982) The use of ecological terms in parasitology (Report of an ad hoc committee of the American society of parasitologists). J Parasitol 68(1): 131-133.
- Rózsa L, Reiczigel J, Majoros G (2000) Quantifying parasites in samples of hosts. J Parasitol 86(2): 228-232.
- Chapman CA, Gillespie TR, Goldberg T L (2005) Primates and the ecology of their infectious diseases: how will anthropogenic change affect host–parasite interactions? Evol Anthropol 14: 134-144.
- Greiner EC, McIntosh A (2009) Collection methods and diagnostic procedures for primate parasitology. In: Huffman MA, Chapman CA (Eds.)., Primate Parasite Ecology: The Dynamics and Study of Host-Parasite Relationships. New York: Cambridge University Press, p. 3-28.
- Cheng TC (2012) General parasitology.
- Soulsby EJL (1982) Helminths, Arthropods and Protozoa of Domesticated Animals (7th)., London :Baillere Tindall and Cassell Ltd, pp. 366‐387.
- Mbora DN, Wieczkowski J, Munene E (2009) Links between habitat degradation, and social group size, ranging, fecundity, and parasite prevalence in the Tana River mangabey (Cercocebusgaleritus). Am J Phys Anthropol 140(3): 562-571.
- Gillespie TR, Greiner EC, Chapman CA (2004) Gastrointestinal parasites of the guenons of western Uganda. J Parasitol 90(6): 1356-1360.
- Kooriyama T, Hasegawa H, Shimozuru M, Tsubota T, Nishida T, et al. (2012) Parasitology of five primates in Mahale Mountains National Park Tanzania. Primates 53(4): 365-375.
- O Reis Amendoeira MR (2015) Balantidium coli and other gastrointestinal parasites in captives non-human primates of the Rio de Janeiro. Brazil J Med Primatol 44(2): 18-26.
- Kouassi RY W, Mcgraw SW, Yao1 PK, Abou-Bacar, A Brunet, et al. (2015) Diversity and prevalence of gastrointestinal parasites in seven non-human primates of the Taï National Park Côte d’Ivoire. Parasite 22(1): 1-12.
- Jha A, Chalise MK, Shrestha RM, Karki K (2011) Intestinal Parasitic Investigation in Temple Rhesus Monkeys of Kathmandu. The Initiation 4: 1-7.
- Poulin R (1997) Species richness of parasite assemblages: evolution and patterns. Annu Rev Ecol Syst 28: 341-358.
- Nunn CL, Altizer S, Jones KE, Sechrest W (2003) Comparative tests of parasite species richness in primates Am Nat 162: 597-614.
- Nunn CL, Altizer SM (2006) Infectious diseases in primates: behavior, ecology and evolution. Oxford, UK: Oxford University Press, pp. 384.
- Morand S (2000) Wormy world: comparative tests of theoretical hypotheses on parasite species richness. In: Poulin R, Morand S, Skorping A (Eds.)., Evolutionary biology of host–parasite relationships: theory meets reality. Elsevier Amsterdam.
- Combes C (1996) Parasites, biodiversity and ecosystem stability. Biodivers Conserv 5: 953-962.
- Grutter AS (1998) Habitat-related differences in the abundance of parasites from a coral reef fish: an indication of the movement patterns of Hemigymnusmelapterus. J Fish Biol 53: 49-57.
- Lane KE, Holley C, Hollocher H, Fuentes A (2011) The anthropogenic environment lessens the intensity and prevalence of gastrointestinal parasites in Balinese long-tailed macaques (Macaca fascicularis). Primates 52(2): 117-128.
- Ye J, Xiao L, Li J, Huang W, Amer SE, et al. (2014) Occurrence of human-pathogenic Enterocytozoonbieneusi, and Cryptosporidium genotypes in laboratory macaques in Guangxi, China. Parasitol Int 63(1): 132-137.
- Altizer S, Dobson A, Hosseini P, Hudson P, Pascual M, et al (2006) Seasonality and the dynamics of infectious diseases. Ecol Lett 9(4): 467-84.
- (2008) IUCN. The 2008 Review of the IUCN Red List of Threatened Species™.
- (2011) IUCN. Guidelines for the application of IUCN Red List categories and criteria. Version 9.0. Red List Standards and Petitions Subcommittee of the Species Survival Commission, IUCN: Gland.
- Petrželkova KJ, Hasegawa H, Appleton CC, Huffman M, Archer, et al. (2010) Gastrointestinal parasites of the chimpanzee population introduced onto Rubondo Island National Park Tanzania. Am J Primatol 72(4): 307-316.
- Sanchez VVV, Patino AS, Segundo VJP, Sandova JAC, Esquivel CVC, et al. (2009) Prevalence of Gastrointestinal Parasites among Captive Primates in Panama. J Anim Vet Adv 8(12): 2644-2649.
- Yamaguti S (1958) Systema Helminthum Vol. I. The digenetic trematodes of vertebrates, Intersciece, New York. Druzno lake Part-I Acta Parasite Pole 6: 1-64.
- Yamaguti S (1959) Systema Helminthum. Vol. II. Cestodes of Vertebrates. Inter Science Publisher Inc. New York, pp. 860.
- Yamaguti S (1961) Systema Helminthum. Vol. III. Nematodes of Vertebrates. Inter Science Publisher Inc New York, pp. 1261.
- Yamaguti S (1963) Systima Helminthum Vol. 5: Acanthocephala. Inter science Publish New York London, pp. 1-423.

 Research Article
Research Article