Abstract
Introduction: Tuberculosis is an important cause of child mortality in developing
countries. BCG vaccination of newborns has been shown to reduce the incidence and
mortality of children with TB. Tuberculin skin test is used to identify people with a
history of exposure to mycobacterial antigens. This test involves an intradermal
injection of tuberculin, which causes delayed hypersensitivity due to T lymphocytes
and occurs within 48 to 72 hours. In this study, we aimed to test the rate of positive
tuberculin test after vaccination Test for infants 4 months to 5 years old in Zahedan.
Materials and Methods: This study was a cross-sectional study that was
performed in 2020 in referral Hospital in Zahedan. In this study, 400 infants from 4
months to 5 years old children who were referred to the hospital were studied. Samples
were selected sequentially and PPD test was performed on them. The bulge diameter
between 48 and 72 hours after inoculation was assessed. Findings were analyzed and
reported.
Results: the average protrusion diameter after inoculation was calculated to be
4.09 mm. Also, 60.8% of children had a bulge of 0 to 5 mm in diameter and 39.2%
were more than 5 mm in diameter. Also, there was a statistically significant difference
between increasing age and the probability of a positive test. (P <0.05)
Discussion: PPD test is a valuable test for diagnosing tuberculosis. According to
the results obtained and other studies, the positive rate of the test can indicate a high
incidence of tuberculosis.
Keywords: Tuberculin Test; BCG Vaccine; Tuberculosis
Introduction
Tuberculosis is an important cause of child mortality in
developing countries. BCG vaccination of newborns has been
shown to reduce the incidence and mortality of children with TB.
Protection against TB by BG vaccination has been observed shortly
after birth in Canadian Indians (80%) and Chicago (75%) [1].
Bacillus Calmette Guerin (BCG) is a live bacterium of Mycobacterium
bovis that was first used by Calmette and Green to immunize
against tuberculosis and mycobacterial infections. The wax was
first used for the general population in 1921 for tuberculosis. Since
then, various vaccines against tuberculosis have been developed
and various models of this vaccine have been developed to enhance
the effectiveness of this vaccine [2,3]. The BCG vaccine is the most
commonly prescribed vaccine worldwide. The vaccine has been
administered to approximately 3 billion people worldwide to date
and is one of the major vaccines in immunizing newborns [4].
Virtually, such as mycobacterial infections, this vaccine provides
some degree of immunity against tuberculosis and, in some cases,
mycobacterial infections [2]. History of natural infection with
Mycobacterium tuberculosis and previous infection with nontuberculosis
mycobacteria provide immunity against tuberculosis
[5,6]. Previous infection with Mycobacterium tuberculosis has
been shown to increase immunity against recurrent infections in
healthy individuals and in HIV-infected individuals, as well as in
immunocompromised individuals who are susceptible to infection
with other types of tuberculosis [7-11].
The duration of immunization of the BCG vaccine against
tuberculosis is approximately 10 to 15 years. This period protects
childhood against tuberculosis. Of course, this period can be
different. A long-running clinical trial since 1930 of BCG vaccination
among Native Americans and Alaskan Indians has shown that
in some cases immunity may last up to 50 or 60 years [12]. The
degree of usefulness of the BCG vaccine depends on three factors:
the individual’s own immunity before vaccination, a history of
mycobacteria infection prior to vaccination, and the potential of the
species used in the BCG vaccine [13]. So far, no agreement has been
reached on which type of BCG is best for the vaccine, and different
types of BCG are used to make the vaccine. There has also been
no evidence that repeated BCG vaccination is more effective than
a single dose against tuberculosis [14]. The standard dose of BCG
vaccine is 0.1 microgram per milliliter. Other childhood vaccines
can be given at the same time as the BCG vaccine. The vaccine can
be given intravenously or multiple subcutaneously using the device.
WHO prefers the subcutaneous method [15]. Skin reactions at the
injection site are a common complication of vaccination. Other
common complications include osteitis, osteomyelitis and diffuse
infection. Factors that affect the incidence of complications include
the dose of the vaccine, the type of vaccine and how the vaccine is
administered [16].
In preterm infants, it is recommended not to inject the vaccine
because the risk of infection in this group is much higher than in term
infants [17]. Among the tests currently used in clinical medicine is
the PPD test, which has been developed in the last century. This long
history of using this test indicates that the interpretation of this
test is still controversial. However, it has been well shown that the
tuberculin skin test reaction and immunity are in fact independent
phenomena. The rate of tuberculin reaction is often consistent with
immune conditions [1]. Tuberculin skin test is used to identify
people with a history of exposure to mycobacterial antigens. The
test involves an intradermal injection of tuberculin, which causes
delayed hypersensitivity due to T lymphocytes and occurs within 48
to 72 hours. Tuberculin is actually purified protein derivative (PPD)
(the recommended dose in North America is 5 units of tuberculin
(0.1 ml)). The standard dose is 2 units of tuberculin [18]. The only
way to test for tuberculin skin is the Mantoux technique, which
involves injecting tuberculin intravenously into the forearm. To
read the test, the diameter of the swollen area should be recorded
and expressed in millimeters. This measurement should be done
within 48 to 72 hours [19]. In this study, we aimed to evaluate the
positive rate of tuberculin test after inoculation of BCG vaccine
in infants 4 months to 5 years old children in university referral
Hospital in Zahedan-Iran.
Methodology
The study was a descriptive study that included children between 4 months and 5 years old who were referred to Hospital in Zahedan who were vaccinated with BCG at birth. Inclusion criteria included having informed consent, age between 4 months to 5 years and history of BCG vaccination. Exclusion criteria also included congenital anomalies, cancer, immune disorders (acquired and congenital), history of infection during vaccination, palpable adenopathy, malnutrition, immunosuppressive drugs and concomitant viral diseases have been. At the beginning of the project, informed consent was obtained from all parents or legal guardians of the children. All children’s information is kept confidential. Sampling was performed through available and sequential samples. Then, for all these patients, a questionnaire form containing all the mentioned variables was filled out. Then PPD test was performed for all subjects and then after 48 hours to 72 hours later the bulge diameter was measured and then the data were entered into the computer and analyzed. To analyze and describe the data, descriptive statistics were used to describe the data including frequency - percentage - mean and standard deviation. Chi-square test was used to compare the frequency of qualitative variables between the two groups.
Results
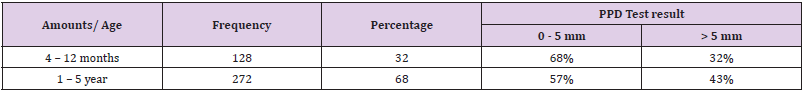
In this study, 400 infants from 4 months to 5 years old children referred to university referral Hospital were evaluated. 173 (43.25%) were girls and 227 (56.27) were boys. In the age study, 128 subjects (32%) were 4 months to 1 year old. Also, 272 people (68%) were 1 to 5 years old. According to the positive and negative criteria of the test mentioned at the Introduction, 243 people (60.8%) were 0 to 5 mm. 157 patients (39.2%) were more than 5 mm in diameter, which indicates that this test is positive. In terms of protrusion diameter after inoculation, the mean protrusion diameter was 4.09 mm with a standard deviation of 2.88. The lowest was 0 mm and the highest was 17 mm. Among people aged 4 months to 5 years, 68% were 0 to 5 mm in diameter and 32% were more than 5 mm in diameter. Also, between 1 and 5 years old, 57% were 0 to 5 mm, 43 more than 5 mm in diameter. (Table 1) Chi-square test was used to evaluate the relationship between age and the possibility of a positive PPD test. After statistical analysis, P valuation was calculated to be 0.026 and there is a statistically significant relationship between age and the probability of positive PPD test. (P<0.05).
Table 1: Frequency of positive PPD test after inoculation of BCG vaccine in 4-month-old infants to 5-year-old.
Discussion
In our study, the bulge diameter was reported to be 4.09 88 2.88 after inoculation. Also, 61% of people had a negative test result and 39% had a positive PPD test result. Another goal of this study was to investigate the relationship between age and the probability of positive PPD test. In data analysis, there was a significant relationship between age and the probability of positive test (P <0.05), ie with these interpretations with increasing age of the child. The probability of a positive PPD test increase. In different studies, the percentage of PPD test positive in different communities, different results have been reported. Which, of course, is related to different conditions such as the prevalence of tuberculosis, the percentage of vaccination coverage, and so on. In a meta-analysis conducted by Dr. Rezai et al. In 2017, which analyzed the last 14 studies, among 26,281 Iranian children (CI: 95%), 8.5% reacted more than 10 mm, 29.9% reacted between 5 They had 9 mm and 60% less than 5 mm. In this study, it was shown that with increasing age, the percentage of positive test decreases. On the contrary, in our study, the test was more positive with age, which is most likely due to the wider age range selected by Dr. Rezaei. On the other hand, in our study, the rate of positive tests was 39%, but in Dr. Rezaei’s study, only 8.5% of cases above 10 mm reacted [20]. In the study of Alavi SM et al. In 2001 in Ahvaz, the percentage of positive test results was reported to be 2%, which was about 1% less than the amount obtained in our study. However, in this study, 90% of people in the first week after birth They had received the BCG vaccine [21]. In a study by Sleiman R in Saudi Arabia in 2007, the positive rate was much higher than our study and Mr. Alavi’s study in Ahwaz, which was reported to be 8%. About 62% of people were also vaccinated with BCG [22]. In 2008, Araujo Z et al reported a positive rate of 28%, which is very high. This high percentage can be due to the reasons mentioned above [23]. In a study published in Uganda, this study also reported a very high rate of positive testing. In this study, the prevalence of positive PPD test was 32%, which is due to factors such as the prevalence of tuberculosis and the percentage of vaccination coverage, etc. As a result of this study.
References
- Shen CM, Soong WJ, Jeng JM, Hwang B (2007) Tuberculin response in infant six month after an intradermal bacille calmette - Guerin vaccinatuin. FJJM 5(3): 115-121.
- Zhou G, Luo Q, Luo S, Teng Z, Ji Z, et al. (2020) Interferon-γ release assays or tuberculin skin test for detection and management of latent tuberculosis infection: a systematic review and meta-analysis. Lancet Infect Dis 20(12): 1457-1469.
- Alam T, Abbas Z, Gillani S, Mustafa SH, Ali SM, et al. (2020) Comparison of Positivity of Tuberculin Skin Test with Diagnostic Bcg in Children Suffering from Tuberculosis. J Ayub Med Coll Abbottabad 32(2): 204-207.
- Khalid FA, Eldirdery MM, El-Gasim ME, Mukhtar MM (2020) Tuberculin reactivity in schoolchildren, Kassala State, Sudan. Int J Mycobacteriol 9(2): 200-204.
- Howard Jones AR, Marais BJ (2020) Tuberculosis in children: screening, diagnosis and management. Curr Opin Pediatr 32(3): 395-404.
- Nygaard U, Poulsen A, Yde Nielsen A, Ravn P, Hawwa Vissing N (2019) Borderline-Positive Quantiferon in Children. Pediatr Infect Dis J 38(11): e307-e309.
- Bjartveit K (2003) Olaf Scheel and Johannes Heimbeck: their contribution to understanding the pathogenesis and prevention of tuberculosis. Int J Tuberc Lung Dis 7(4): 306-11.
- Sudbury E, Villanueva P, Goetzinger F, Tebruegge M, Curtis N (2019) The impact of bacille Calmette-Guérin vaccination on tuberculin skin test results in young children. Int J Tuberc Lung Dis 23(12): 1335-1336.
- Verver S, Warren RM, Beyers N, Madalene Richardson, Gian Vander Spuy, et al. (2005) Rate of reinfection tuberculosis after successful treatment is higher than rate of new tuberculosis. Am J Respir Crit Care Med 171(12): 1430.
- Le Saux N (2019) Approaches to detecting tuberculosis in children and youth. Paediatr Child Health 24(1): 52-53.
- Aronson NE, Santosham M, Comstock GW, Robin S Howard, Lawrence H Moulton, et al. (2004) Long-term efficacy of BCG vaccine in American Indians and Alaska Natives: A 60-year follow-up study. JAMA 291(17): 2086.
- Kumar CM, Bedi N (2019) Tuberculin Conversion after BCG Vaccination. Indian Pediatr 56(2):141-142.
- Black GF, Weir RE, Floyd S, Lyn Bliss, David K Warndorff, et al. (2002) BCG-induced increase in interferon-gamma response to mycobacterial antigens and efficacy of BCG vaccination in Malawi and the UK: two randomised controlled studies. Lancet 359(9315): 1393-401.
- Hawkridge A, Hatherill M, Little F, Margaret Ann Goetz, Lew Barker, et al. (2008) Efficacy of percutaneous versus intradermal BCG in the prevention of tuberculosis in South African infants: randomised trial. BMJ 337: a2052.
- Volkman T, Graham H, Laemmle-Ruff I, Tosif S, Tebruegge M, et al. (2019) Interpretation and management of discordant tuberculin skin test and interferon-gamma release assays results in children. J Paediatr Child Health. 55(2): 247-248.
- Sedaghatian MR, Kardouni K (1993) Tuberculin response in preterm infant after BCG vaccination at birth. Arch dis child 69: 309-311.
- Comstock GW, Edwards LB, Philip RN, Winn WA (1964) A Comparison in the United States of America of Two Tuberculins, PPD-S and RT 23. Bull World Health Organ 31(2): 161–170.
- Primaturia C, Reniarti L, Nataprawira HMN (2020) Comparison between the Interferon γ Release Assay-QuantiFERON Gold Plus (QFT-Plus)-and Tuberculin Skin Test (TST) in the Detection of Tuberculosis Infection in Immunocompromised Children. Pulm Med: 7159485.
- Rezai MS, Abedi S, Afshari M, Moosazadeh M (2017) Estimating Tuberculin Skin Test Reactions among Children and Teenagers Who Received the Bacillus Calmette-Guerin Vaccination at Birth: A Meta-analysis. Osong Public Health Res Perspect 8(1): 3-10.
- Alavi SM, Sefidgaran GH (2008) Tuberculin survey among school-aged children in Ahvaz, Iran, 2006. Int J Infect Dis 12(4): 406-9.
- Soleiman R, Al-Tannir M, Dakdouki G, Ziade F, Assi NA, et al. (2007) Interpretation of the tuberculin skin test in bacille Calmette-Guérin vaccinated and nonvaccinated school children. Pediatr Infect Dis J 26(2): 134-8.
- Araujo Z, De Waard JH, De Larrea CF, Borges R, Convit J (2008) The effect of Bacille Calmette-Guérin vaccine on tuberculin reactivity in indigenous children from communities with high prevalence of tuberculosis. Vaccine 26(44): 5575-81.
- Mudido PM, Guwatudde D, Nakakeeto MK, Bukenya GB, Nsamba D, et al. (1999) The effect of bacille Calmette-Guérin vaccination at birth on tuberculin skin test reactivity in Ugandan children. Int J Tuberc Lung Dis 3(10): 891-5.

 Short Communication
Short Communication